Home > Press > Inexpensive “Nanoglue” Can Bond Nearly Anything Together
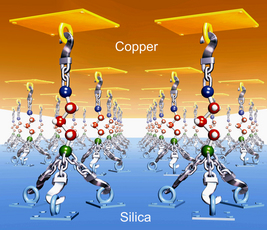 |
| Rensselaer/G. Ramanath A new method allows a self-assembled molecular nanolayer to become a powerful nanoglue by "hooking" together any two surfaces that normally don't stick well. Unprotected, a nanolayer (green ball: silicon, blue: sulphur, red: carbon, white: hydrogen) would degrade or detach from a surface when heated to 400 degrees Celsius. But when topped with a thin copper film that binds strongly with the nanolayer, heat causes the nanolayer to form strong chemical bonds to the silica underlayer -- hooking or gluing the copper-silica "sandwich" together. This technique produces a sevenfold increase of the thin film sandwich's adhesion strength and allows the nanolayer to withstand temperatures of at least 700 degrees Celsius. Both features are unexpected and unprecedented. This new ability to bond together nearly any two surfaces using nanolayers will benefit nanoelectronics and computer chip manufacturing. Other envisioned applications include coatings for turbines and jet engines, and adhesives for high-heat environments. |
Abstract:
Researchers at Rensselaer Polytechnic Institute have developed a new method to bond materials that don't normally stick together. The team's adhesive, which is based on self-assembling nanoscale chains, could impact everything from next-generation computer chip manufacturing to energy production.
Inexpensive “Nanoglue” Can Bond Nearly Anything Together
Troy, NY | Posted on May 16th, 2007Researchers at Rensselaer Polytechnic Institute have developed a new method to bond materials that don't normally stick together. The team's adhesive, which is based on self-assembling nanoscale chains, could impact everything from next-generation computer chip manufacturing to energy production.
Less than a nanometer - or one billionth of a meter - thick, the nanoglue is inexpensive to make and can withstand temperatures far higher than what was previously envisioned. In fact, the adhesive's molecular bonds strengthen when exposed to heat.
The glue material is already commercially available, but the research team's method of treating the glue to dramatically enhance its "stickiness" and heat resistance is completely new. The project, led by Rensselaer materials science and engineering professor Ganapathiraman Ramanath, is featured in the May 17 issue of the journal Nature.
Like many key scientific discoveries, Ramanath and his team happened upon the novel, heat-hardened nanoglue by accident.
For years Ramanath has investigated ways of assembling layers of molecular chains between two different materials to enhance the structural integrity, efficiency, and reliability of semiconductor devices in computer chips. His team has shown that molecular chains with a carbon backbone - ending with appropriate elements such as silicon, oxygen, or sulfur - can improve adhesion and prevent heat-triggered mixing of atoms of the adjoining substances. Recently, Ramanath's group and other researchers have found these nanolayers to be useful for creating adhesives, lubricants, and protective surface coatings.
The nanolayers, however, are extremely susceptible to heat and begin to degrade or simply detach from a surface when exposed to temperatures above 400 degrees Celsius. This severe limitation has precluded more widespread use of the nanolayers.
Ramanath's team decided to sandwich a nanolayer between a thin film of copper and silica, thinking the extra support would help strengthen the nanolayer's bonds and boost its adhesive properties. It proved to be an insightful venture, and the research team found more than it bargained for.
When exposed to heat, the middle layer of the "nanosandwich" did not break down or fall off - as it had nowhere to go. But that was not the only good news. The nanolayer's bonds grew stronger and more adhesive when exposed to temperatures above 400 degrees Celsius. Constrained between the copper and silica, the nanolayer's molecules hooked onto an adjoining surface with unexpectedly strong chemical bonds.
"The higher you heat it, the stronger the bonds are," Ramanath said. "When we first started out, I had not imagined the molecules behaving this way."
To make sure it wasn't a fluke, his team recreated the test more than 50 times over the past two years. The results have been consistent, and show heating up the sandwiched nanolayer increases its interface toughness - or "stickiness" - by five to seven times. Similar toughness has been demonstrated using micrometer-thick layers, but never before with a nanometer-thick layer. A nanometer is 1,000 times smaller than a micrometer.
Because of their small size, these enhanced nanolayers will likely be useful as adhesives in a wide assortment of micro- and nanoelectronic devices where thicker adhesive layers just won't fit.
Another unprecedented aspect of Ramanath's discovery is that the sandwiched nanolayers continue to strengthen up to temperatures as high as 700 degrees Celsius. The ability of these adhesive nanolayers to withstand and grow stronger with heat could have novel industrial uses, such as holding paint on hot surfaces like the inside of a jet engine or a huge power plant turbine.
Along with nanoscale and high heat situations, Ramanath is confident the new nanoglue will have other unforeseen uses.
"This could be a versatile and inexpensive solution to connect any two materials that don't bond well with each other," Ramanath said. "Although the concept is not intuitive at first, it is simple, and could be implemented for a wide variety of potential commercial applications.
"The molecular glue is inexpensive - 100 grams cost about $35 - and already commercially available, which makes our method well-suited to today's marketplace. Our method can definitely be scaled up to meet the low-cost demands of a large manufacturer," he said.
Ramanath and his team have filed a disclosure on their findings and are moving forward toward a patent, which will complement the robust portfolio of other intellectual property they hold in this field. The team is also exploring what happens when certain variables of the nanoglue are tweaked, such as making taller nanolayers or sandwiching the layers between substances other than copper and silica.
Along with Ramanath, Rensselaer materials science and engineering graduate students Darshan Gandhi and Amit Singh contributed to the paper. Other co-authors include Rensselaer physics professor Saroj Nayak and graduate student Yu Zhou, IBM researcher Michael Lane at the T.J. Watson Research Center in Yorktown Heights, N.Y., and Ulrike Tisch and Moshe Eizenberg of the Technion-Israel Institute of Technology.
Ramanath's ongoing research is supported by the National Science Foundation, the U.S.-Israel Binational Science Foundation, the Alexander von Humboldt Foundation, and New York state through the Interconnect Focus Center.
LaVerne Hess, the NSF program official most familiar with Ramanath's work, applauded the interdisciplinary nature and strong technical relevance of the nanoglue project.
"It's a good example of basic materials science research motivated by an understanding of engineering needs in the electronics field, and involving fundamental chemistry concepts to create new materials capabilities to enable progress in a field important to U.S. competitiveness," Hess said.
####
About Rensselaer Polytechnic Institute
Rensselaer Polytechnic Institute, founded in 1824, is the nation’s oldest technological university. The university offers bachelor’s, master’s, and doctoral degrees in engineering, the sciences, information technology, architecture, management, and the humanities and social sciences. Institute programs serve undergraduates, graduate students, and working professionals around the world. Rensselaer faculty are known for pre-eminence in research conducted in a wide range of fields, with particular emphasis in biotechnology, nanotechnology, information technology, and the media arts and technology. The Institute is well known for its success in the transfer of technology from the laboratory to the marketplace so that new discoveries and inventions benefit human life, protect the environment, and strengthen economic development.
For more information, please click here
Contacts:
Michael Mullaney
Rensselaer Polytechnic Institute
Office of Strategic Communications and External Relations
518-276-6161
518-698-6336 (cell)
Copyright © Newswise
If you have a comment, please Contact us.Issuers of news releases, not 7th Wave, Inc. or Nanotechnology Now, are solely responsible for the accuracy of the content.
| Related News Press |
Chip Technology
![]() A reusable chip for particulate matter sensing April 17th, 2026
A reusable chip for particulate matter sensing April 17th, 2026
![]() Metasurfaces smooth light to boost magnetic sensing precision January 30th, 2026
Metasurfaces smooth light to boost magnetic sensing precision January 30th, 2026
Discoveries
![]() Quantum computer improves AI predictions April 17th, 2026
Quantum computer improves AI predictions April 17th, 2026
![]() Flexible sensor gains sensitivity under pressure April 17th, 2026
Flexible sensor gains sensitivity under pressure April 17th, 2026
![]() A reusable chip for particulate matter sensing April 17th, 2026
A reusable chip for particulate matter sensing April 17th, 2026
![]() Detecting vibrational quantum beating in the predissociation dynamics of SF6 using time-resolved photoelectron spectroscopy April 17th, 2026
Detecting vibrational quantum beating in the predissociation dynamics of SF6 using time-resolved photoelectron spectroscopy April 17th, 2026
Materials/Metamaterials/Magnetoresistance
![]() First real-time observation of two-dimensional melting process: Researchers at Mainz University unveil new insights into magnetic vortex structures August 8th, 2025
First real-time observation of two-dimensional melting process: Researchers at Mainz University unveil new insights into magnetic vortex structures August 8th, 2025
![]() Researchers unveil a groundbreaking clay-based solution to capture carbon dioxide and combat climate change June 6th, 2025
Researchers unveil a groundbreaking clay-based solution to capture carbon dioxide and combat climate change June 6th, 2025
![]() A 1960s idea inspires NBI researchers to study hitherto inaccessible quantum states June 6th, 2025
A 1960s idea inspires NBI researchers to study hitherto inaccessible quantum states June 6th, 2025
![]() Institute for Nanoscience hosts annual proposal planning meeting May 16th, 2025
Institute for Nanoscience hosts annual proposal planning meeting May 16th, 2025
Announcements
![]() A fundamentally new therapeutic approach to cystic fibrosis: Nanobody repairs cellular defect April 17th, 2026
A fundamentally new therapeutic approach to cystic fibrosis: Nanobody repairs cellular defect April 17th, 2026
![]() UC Irvine physicists discover method to reverse ‘quantum scrambling’ : The work addresses the problem of information loss in quantum computing system April 17th, 2026
UC Irvine physicists discover method to reverse ‘quantum scrambling’ : The work addresses the problem of information loss in quantum computing system April 17th, 2026
Energy
![]() Sensors innovations for smart lithium-based batteries: advancements, opportunities, and potential challenges August 8th, 2025
Sensors innovations for smart lithium-based batteries: advancements, opportunities, and potential challenges August 8th, 2025
![]() Simple algorithm paired with standard imaging tool could predict failure in lithium metal batteries August 8th, 2025
Simple algorithm paired with standard imaging tool could predict failure in lithium metal batteries August 8th, 2025
|
|
||
|
|
||
| The latest news from around the world, FREE | ||
|
|
||
|
|
||
| Premium Products | ||
|
|
||
|
Only the news you want to read!
Learn More |
||
|
|
||
|
Full-service, expert consulting
Learn More |
||
|
|
||








