Home > Press > Porous material sized up
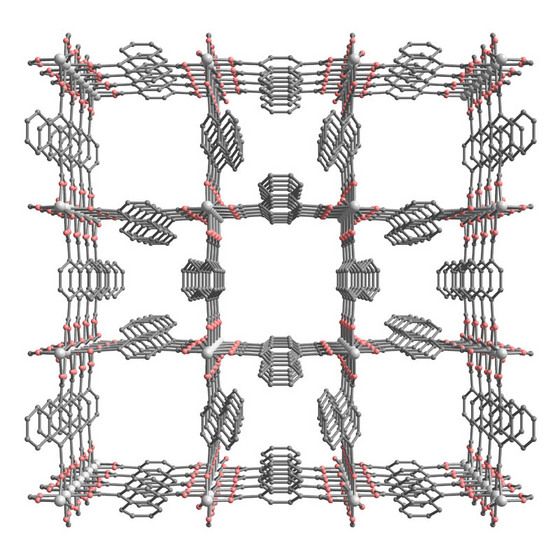 |
| Figure 1: A schematic of the metal-organic framework, which contains a highly ordered network of small and large nanopores. |
Abstract:
A metal-organic framework that contains ordered channels of two different sizes can separate different gases
Porous material sized up
Japan | Posted on February 26th, 2009The material's structure is based on a three-dimensional lattice of aluminum atoms, surrounded by oxygen atoms. These clusters are connected by a carbon-based molecule called 1, 4-naphthalene dicarboxylate (1, 4-NDC). Such metal-organic frameworks (MOFs) are finding uses in separating liquids or gases, acting as catalysts to speed up chemical reactions, or for safely storing large volumes of gas.
The international team of scientists that made the material, including Masaki Takata of RIKEN's Harima Institute, used x-rays to reveal that it contains ordered channels of two different sizes1. The smaller channels, crowded by napthalene units, are just 0.3 nanometers (billionths of a meter) across, while the larger channels are 0.77 nanometers across (Fig. 1).
"We expected the porous structure to some extent," says Satoshi Horike, now at the University of California, Berkeley, who was part of the team. "But the existence of two kinds of pores in our system is kind of surprising."
The team then used nuclear magnetic resonance (NMR), a technique which measures the magnetic behavior of atomic nuclei, to build up a more detailed three-dimensional picture of the MOF.
The scientists analyzed a sample of the inert gas xenon as it diffused through the MOF's pores. This experiment confirmed that the material has parallel, yet independent, pores. Xenon atoms have a diameter of about 0.44 nanometers, and did not fit down the smaller channels. However, it took mere milliseconds for the xenon to diffuse through the larger channels, which were not blocked by any other molecules, such as water.
Carbon dioxide could also seep through the material. The scientists calculate that each gram of the MOF can bind carbon dioxide to an internal surface area which is greater than 500 m2, roughly the size of two tennis courts.
And while the small organic molecules methanol and acetone were easily absorbed, more pressure was required to squeeze water into the pores because of high hydrophobicity. This could make the material useful for separating water from alcohols, suggests Horike. "Gas separation between water and ethanol is important for obtaining liquid fuel from biomass sources," he says.
The smaller pores could also be useful for storing hydrogen gas, a potentially environmentally friendly fuel. "Hydrogen has a really small interaction with pore surfaces," explains Horike, "and the small pore would be able to bind hydrogen gas more tightly than the large one."
Reference
1. Comotti, A., Bracco, S., Sozzani, P., Horike, S., Matsuda, R., Chen, J., Takata, M., Kubota, Y. & Kitagawa, S. Nanochannels of two distinct cross-sections in a porous Al-based coordination polymer. Journal of the American Chemical Society 130, 13664-13672 (2008).
The corresponding author for this highlight is based at the RIKEN Structural Materials Science Laboratory
####
For more information, please click here
Copyright © Riken
If you have a comment, please Contact us.Issuers of news releases, not 7th Wave, Inc. or Nanotechnology Now, are solely responsible for the accuracy of the content.
| Related Links |
| Related News Press |
News and information
![]() Quantum computer improves AI predictions April 17th, 2026
Quantum computer improves AI predictions April 17th, 2026
![]() Flexible sensor gains sensitivity under pressure April 17th, 2026
Flexible sensor gains sensitivity under pressure April 17th, 2026
![]() A reusable chip for particulate matter sensing April 17th, 2026
A reusable chip for particulate matter sensing April 17th, 2026
![]() Detecting vibrational quantum beating in the predissociation dynamics of SF6 using time-resolved photoelectron spectroscopy April 17th, 2026
Detecting vibrational quantum beating in the predissociation dynamics of SF6 using time-resolved photoelectron spectroscopy April 17th, 2026
Chemistry
![]() Projecting light to dispense liquids: A new route to ultra-precise microdroplets January 30th, 2026
Projecting light to dispense liquids: A new route to ultra-precise microdroplets January 30th, 2026
![]() From sensors to smart systems: the rise of AI-driven photonic noses January 30th, 2026
From sensors to smart systems: the rise of AI-driven photonic noses January 30th, 2026
![]() "Nanoreactor" cage uses visible light for catalytic and ultra-selective cross-cycloadditions October 3rd, 2025
"Nanoreactor" cage uses visible light for catalytic and ultra-selective cross-cycloadditions October 3rd, 2025
![]() Cambridge chemists discover simple way to build bigger molecules � one carbon at a time June 6th, 2025
Cambridge chemists discover simple way to build bigger molecules � one carbon at a time June 6th, 2025
Discoveries
![]() Quantum computer improves AI predictions April 17th, 2026
Quantum computer improves AI predictions April 17th, 2026
![]() Flexible sensor gains sensitivity under pressure April 17th, 2026
Flexible sensor gains sensitivity under pressure April 17th, 2026
![]() A reusable chip for particulate matter sensing April 17th, 2026
A reusable chip for particulate matter sensing April 17th, 2026
![]() Detecting vibrational quantum beating in the predissociation dynamics of SF6 using time-resolved photoelectron spectroscopy April 17th, 2026
Detecting vibrational quantum beating in the predissociation dynamics of SF6 using time-resolved photoelectron spectroscopy April 17th, 2026
Announcements
![]() A fundamentally new therapeutic approach to cystic fibrosis: Nanobody repairs cellular defect April 17th, 2026
A fundamentally new therapeutic approach to cystic fibrosis: Nanobody repairs cellular defect April 17th, 2026
![]() UC Irvine physicists discover method to reverse �quantum scrambling� : The work addresses the problem of information loss in quantum computing system April 17th, 2026
UC Irvine physicists discover method to reverse �quantum scrambling� : The work addresses the problem of information loss in quantum computing system April 17th, 2026
|
|
||
|
|
||
| The latest news from around the world, FREE | ||
|
|
||
|
|
||
| Premium Products | ||
|
|
||
|
Only the news you want to read!
Learn More |
||
|
|
||
|
Full-service, expert consulting
Learn More |
||
|
|
||








