 |
Abstract:
Photoluminescent liquid crystals based on metal clusters
An Odd Couple
Weinheim, Germany | Posted on April 30th, 2010Combine liquid crystals (mesogens) and metal clusters and you get clustomesogens -- a new class of compounds, the first examples of which have now been produced by scientists at the Universities of Rennes (France) and Bucharest (Rumania). As the team headed by Yann Molard and Stephane Cordier reports in the journal Angewandte Chemie, when irradiated, the material glows intensely in the red and infrared range.
Liquid crystals are familiar from LC displays. These materials behave like something between a liquid and a solid. On one hand, their molecules are oriented and partially ordered like in a crystal; on the other, they are not rigid, but truly mobile, like in a liquid. When a voltage is applied, liquid crystals can be switched between different states.
Another class of materials of interest to the optoelectronics field is metal clusters. Clusters are aggregates of a few atoms. Such clusters of metal atoms show unusual electronic, magnetic, and optical properties that arise from the metal-metal bonds in the cluster, and the properties are completely different from those of macroscopic metal particles. The team was able to unite the interesting characteristics of both types of material in the form of a new class of materials called clustomesogens, which contain metal clusters in a liquid-crystalline phase.
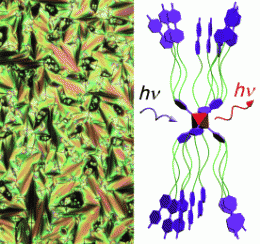
Liquid crystals with metal-metal bonds have been rare and limited to species with only two connected metal atoms. The scientists have now constructed a liquid crystal that contains octahedral clusters made of six molybdenum atoms. For stabilization, there are eight bromide ions coordinated as ligands above the eight surfaces of the octahedron. The researchers attached special organic ligands to the six corners of the octahedron. These ligands are aromatic rings, each equipped with three long hydrocarbon chains, the ends of which are also made of two aromatic rings. Simple warming triggers a self-organization process by which the clusters stretch out to form long, narrow units (see figure) arranged in a lamellar structure. The flat rings at the ends of the ligands of neighboring layers are interleaved; this structure has liquid-crystalline properties.
The material shows strong luminescence in the red/near infrared range when excited over a broad range of wavelengths, and this type of material may be useful for the production of red displays and infrared signals. The new class of compounds has been patented in the USA in November 2009 as no. 61/264888.
Author: Yann Molard, Universit� de Rennes 1 (France),
scienceschimiques.univ-rennes1.fr/csm/personnel/y_molard.html
Title: Clustomesogens: Liquid Crystal Materials Containing Transition-Metal Clusters
Angewandte Chemie International Edition 2010, 49, No. 19, 3351-3355, Permalink to the article: dx.doi.org/10.1002/anie.201000325
####
About Wiley InterScience
Wiley InterScience (www.interscience.wiley.com) provides access to over 3 million articles across nearly 1500 journals and 7000 Online Books and major reference works. It also holds industry leading databases such as The Cochrane Library, chemistry databases and the acclaimed Current Protocols laboratory manuals.
Wiley InterScience is one of the world's premiere resources for study, teaching and advanced research.
For more information, please click here
Contacts:
Editorial office
Amy Molnar (US)
Jennifer Beal (UK)
Alina Boey (Asia)
Copyright © Wiley InterScience
If you have a comment, please Contact us.Issuers of news releases, not 7th Wave, Inc. or Nanotechnology Now, are solely responsible for the accuracy of the content.
| Related News Press |
News and information
![]() Simulating magnetization in a Heisenberg quantum spin chain April 5th, 2024
Simulating magnetization in a Heisenberg quantum spin chain April 5th, 2024
![]() NRL charters Navy�s quantum inertial navigation path to reduce drift April 5th, 2024
NRL charters Navy�s quantum inertial navigation path to reduce drift April 5th, 2024
![]() Discovery points path to flash-like memory for storing qubits: Rice find could hasten development of nonvolatile quantum memory April 5th, 2024
Discovery points path to flash-like memory for storing qubits: Rice find could hasten development of nonvolatile quantum memory April 5th, 2024
![]() Good as gold - improving infectious disease testing with gold nanoparticles April 5th, 2024
Good as gold - improving infectious disease testing with gold nanoparticles April 5th, 2024
Display technology/LEDs/SS Lighting/OLEDs
![]() Utilizing palladium for addressing contact issues of buried oxide thin film transistors April 5th, 2024
Utilizing palladium for addressing contact issues of buried oxide thin film transistors April 5th, 2024
![]() Light guide plate based on perovskite nanocomposites November 3rd, 2023
Light guide plate based on perovskite nanocomposites November 3rd, 2023
![]() Simple ballpoint pen can write custom LEDs August 11th, 2023
Simple ballpoint pen can write custom LEDs August 11th, 2023
Possible Futures
![]() Discovery points path to flash-like memory for storing qubits: Rice find could hasten development of nonvolatile quantum memory April 5th, 2024
Discovery points path to flash-like memory for storing qubits: Rice find could hasten development of nonvolatile quantum memory April 5th, 2024
![]() With VECSELs towards the quantum internet Fraunhofer: IAF achieves record output power with VECSEL for quantum frequency converters April 5th, 2024
With VECSELs towards the quantum internet Fraunhofer: IAF achieves record output power with VECSEL for quantum frequency converters April 5th, 2024
Materials/Metamaterials/Magnetoresistance
![]() Nanoscale CL thermometry with lanthanide-doped heavy-metal oxide in TEM March 8th, 2024
Nanoscale CL thermometry with lanthanide-doped heavy-metal oxide in TEM March 8th, 2024
![]() Focused ion beam technology: A single tool for a wide range of applications January 12th, 2024
Focused ion beam technology: A single tool for a wide range of applications January 12th, 2024
Announcements
![]() NRL charters Navy�s quantum inertial navigation path to reduce drift April 5th, 2024
NRL charters Navy�s quantum inertial navigation path to reduce drift April 5th, 2024
![]() Discovery points path to flash-like memory for storing qubits: Rice find could hasten development of nonvolatile quantum memory April 5th, 2024
Discovery points path to flash-like memory for storing qubits: Rice find could hasten development of nonvolatile quantum memory April 5th, 2024
Patents/IP/Tech Transfer/Licensing
![]() Getting drugs across the blood-brain barrier using nanoparticles March 3rd, 2023
Getting drugs across the blood-brain barrier using nanoparticles March 3rd, 2023
![]() Metasurfaces control polarized light at will: New research unlocks the hidden potential of metasurfaces August 13th, 2021
Metasurfaces control polarized light at will: New research unlocks the hidden potential of metasurfaces August 13th, 2021
![]() Arrowhead Pharmaceuticals Announces Closing of Agreement with Takeda November 27th, 2020
Arrowhead Pharmaceuticals Announces Closing of Agreement with Takeda November 27th, 2020
|
|
||
|
|
||
| The latest news from around the world, FREE | ||
|
|
||
|
|
||
| Premium Products | ||
|
|
||
|
Only the news you want to read!
Learn More |
||
|
|
||
|
Full-service, expert consulting
Learn More |
||
|
|
||








