Home > Press > Biting its Own Tail
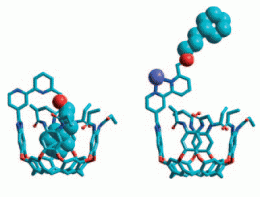 |
| Molecular switch: The ouroborand coordinates an internal side chain in its cavity, just as it were swallowing its own tail. The presence or absence of zinc(II) in solution switches the cavity between open and closed states to external guests (see scheme: deep blue sphere: Zn). |
Abstract:
Nanocontainer with an integrated switch
Biting its Own Tail
Weinheim, Germany | Posted on April 20th, 2010The ouroboros (ancient Greek for "tail devourer") is a motif found in many cultures: a snake biting its own tail, it symbolizes eternity and cycles. Julius Rebek, Jr. and Fabien Durola (The Scripps Research Institute, La Jolla, USA) have now constructed a molecular tail devourer, and have named this new class of compound "ouroborand". As the researchers report in the journal Angewandte Chemie, their tail-biter is a molecular "machine", which functions as a nanocontainer with a built-in switch that regulates access to the cavity.
Molecular machines and nanoscopic components imitate�at least theoretically�the functions of their macroscopic analogues. For example, nanoscopic capsules can act as reaction vessels, molecules with parts that rotate relative to each other to imitate rotors, and various types of on/off switches.
The ouroborand made by the American research duo is a molecule consisting of multiple parts. A cavity that can take up guest molecules serves as a container. At its edge, the container has a switchable rotor (a bipyridyl unit) to which an intramolecular guest is attached like a hand at the end of a coupling arm of appropriate length. The rotor is turned so that the hand at the end of the arm sits inside the container. The container is thus blocked and not accessible to other molecules; it is switched to closed. In this conformation it is reminiscent of a snake that is swallowing its own tail, the ouroboros.
If zinc ions are added to the solution, they trigger a switching mechanism: the rotor has two binding sites for zinc ions. In order for both to bind an ion, the rotor must make a half-turn. The coupling arm turns with it, which causes the hand to be pulled out of the container. The vessel is now free and accessible to other molecules; it is switched to open. If the zinc ions are taken back out of the solution, the rotor then turns back to the starting position and the hand throws the foreign molecule back out of the container.
Author: Julius Rebek, Jr., Scripps Research Institute, La Jolla (USA), www.scripps.edu/skaggs/rebek/
Title: The Ouroborand: A Cavitand with a Coordination-Driven Switching Device
Angewandte Chemie International Edition 2010, 49, No. 18, 3189-3191, Permalink to the article: dx.doi.org/10.1002/anie.200906753
####
About Angewandte Chemie
Wiley InterScience (www.interscience.wiley.com) provides access to over 3 million articles across nearly 1500 journals and 7000 Online Books and major reference works. It also holds industry leading databases such as The Cochrane Library, chemistry databases and the acclaimed Current Protocols laboratory manuals.
For more information, please click here
Contacts:
Editorial office
Amy Molnar (US)
Jennifer Beal (UK)
Alina Boey (Asia)
Copyright © Angewandte Chemie
If you have a comment, please Contact us.Issuers of news releases, not 7th Wave, Inc. or Nanotechnology Now, are solely responsible for the accuracy of the content.
| Related News Press |
News and information
![]() Simulating magnetization in a Heisenberg quantum spin chain April 5th, 2024
Simulating magnetization in a Heisenberg quantum spin chain April 5th, 2024
![]() NRL charters Navy�s quantum inertial navigation path to reduce drift April 5th, 2024
NRL charters Navy�s quantum inertial navigation path to reduce drift April 5th, 2024
![]() Discovery points path to flash-like memory for storing qubits: Rice find could hasten development of nonvolatile quantum memory April 5th, 2024
Discovery points path to flash-like memory for storing qubits: Rice find could hasten development of nonvolatile quantum memory April 5th, 2024
Molecular Machines
![]() First electric nanomotor made from DNA material: Synthetic rotary motors at the nanoscale perform mechanical work July 22nd, 2022
First electric nanomotor made from DNA material: Synthetic rotary motors at the nanoscale perform mechanical work July 22nd, 2022
![]() Nanotech scientists create world's smallest origami bird March 17th, 2021
Nanotech scientists create world's smallest origami bird March 17th, 2021
![]() Giant nanomachine aids the immune system: Theoretical chemistry August 28th, 2020
Giant nanomachine aids the immune system: Theoretical chemistry August 28th, 2020
Announcements
![]() NRL charters Navy�s quantum inertial navigation path to reduce drift April 5th, 2024
NRL charters Navy�s quantum inertial navigation path to reduce drift April 5th, 2024
![]() Discovery points path to flash-like memory for storing qubits: Rice find could hasten development of nonvolatile quantum memory April 5th, 2024
Discovery points path to flash-like memory for storing qubits: Rice find could hasten development of nonvolatile quantum memory April 5th, 2024
|
|
||
|
|
||
| The latest news from around the world, FREE | ||
|
|
||
|
|
||
| Premium Products | ||
|
|
||
|
Only the news you want to read!
Learn More |
||
|
|
||
|
Full-service, expert consulting
Learn More |
||
|
|
||








