Home > Press > Researcher suggests new memory storage mineral
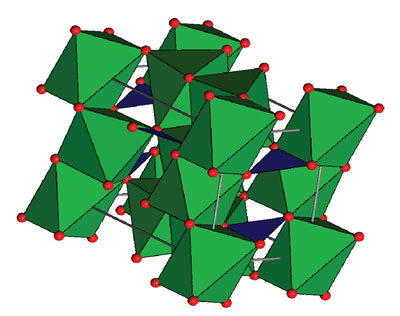 |
| A representation of the mineral kotoite's crystal structure. The oxygen atoms are red, and the magnesium atoms are located at the centers of the green octahedra. The boron atoms are located at the centers of the blue triangles connecting the oxygen atoms. Derek Stewart. |
Abstract:
Breakthroughs in electronics often are the result of finding just the right material for a device -- like the tungsten in light bulbs or the silicon in transistors. Now, a Cornell scientist believes that the mineral kotoite could be an ideal insulator for memory storage devices called magnetic tunnel junctions, found in computers, cell phones and magnetic field sensors.
Researcher suggests new memory storage mineral
Ithaca, NY | Posted on January 21st, 2010The work, building on previous research by other Cornell scientists, is published by Derek Stewart, the Cornell NanoScale Science and Technology Facility's computational research associate, in the Dec. 17 online edition of Nano Letters (to appear later in print).
Magnetic tunnel junctions are made of a sandwich of two magnets, typically iron-based, with an oxide in the middle only nanometers thick. (A nanometer is one-billionth of a meter.) Electrons "tunnel" between the two magnets, and the oxide filters information from the electrons' spin states to create what is called nonvolatile memory, which doesn't require electricity to store information. These junctions are also used as very sensitive magnetic sensors or read heads for hard drives, since the device currents depend on the relative orientation of the iron layers' magnetic poles.
Cornell researchers, including Robert Buhrman, the John Edson Sweet Professor of Engineering, and Dan Ralph, the Horace White Professor of Physics, have been on the leading edge of this technology for several years.
In industry today, most magnetic tunnel junctions use aluminum oxide as the insulator. But in labs across the world, magnesium oxide is being tested as a next-generation insulator, because its cubic crystal structure matches well with the metallic leads, allowing more efficient filtering of electrons. John Read, a former graduate student in Buhrman's lab (now a postdoctoral associate at the National Institute of Standards and Technology), discovered by accident that the element boron, which he had used at Cornell while fabricating magnetic tunnel junctions to help smooth the material interfaces, was leaking into the insulators and forming a crystal, rather than diffusing away as intended. Yet the devices still worked.
Curious, the team tapped Stewart's computational expertise to work backward and figure out what specific material may have been inadvertently created between the two magnets as a result of the boron contamination.
Density functional calculations brought Stewart to kotoite (Mg3B2O6), a magnesium oxide that also has two boron atoms, which matches well with the magnets' chemistry, allows good electron filtering, and has a slightly different crystal shape than plain magnesium oxide (MgO). He also demonstrated that the mineral's crystal shape -- orthorhombic, as opposed to magnesium oxide's cubic symmetry -- could lead to even better electron spin filtering.
"Derek did a beautiful job of demonstrating that the symmetry arguments that one makes for magnesium oxide can be demonstrated for [kotoite]," Read said.
Calculations were done on the Intel Cluster at CNF, which is funded by the National Science Foundation.
####
About Cornell University
Once called "the first American university" by educational historian Frederick Rudolph, Cornell University represents a distinctive mix of eminent scholarship and democratic ideals. Adding practical subjects to the classics and admitting qualified students regardless of nationality, race, social circumstance, gender, or religion was quite a departure when Cornell was founded in 1865.
Today's Cornell reflects this heritage of egalitarian excellence. It is home to the nation's first colleges devoted to hotel administration, industrial and labor relations, and veterinary medicine. Both a private university and the land-grant institution of New York State, Cornell University is the most educationally diverse member of the Ivy League.
On the Ithaca campus alone nearly 20,000 students representing every state and 120 countries choose from among 4,000 courses in 11 undergraduate, graduate, and professional schools. Many undergraduates participate in a wide range of interdisciplinary programs, play meaningful roles in original research, and study in Cornell programs in Washington, New York City, and the world over.
For more information, please click here
Contacts:
Media Contact:
Blaine Friedlander
(607) 254-8093
Cornell Chronicle:
Anne Ju
(607) 255-9735
Related Information
Copyright © Cornell University
If you have a comment, please Contact us.Issuers of news releases, not 7th Wave, Inc. or Nanotechnology Now, are solely responsible for the accuracy of the content.
| Related News Press |
Physics
![]() Simulating magnetization in a Heisenberg quantum spin chain April 5th, 2024
Simulating magnetization in a Heisenberg quantum spin chain April 5th, 2024
News and information
![]() Simulating magnetization in a Heisenberg quantum spin chain April 5th, 2024
Simulating magnetization in a Heisenberg quantum spin chain April 5th, 2024
![]() NRL charters Navy�s quantum inertial navigation path to reduce drift April 5th, 2024
NRL charters Navy�s quantum inertial navigation path to reduce drift April 5th, 2024
![]() Discovery points path to flash-like memory for storing qubits: Rice find could hasten development of nonvolatile quantum memory April 5th, 2024
Discovery points path to flash-like memory for storing qubits: Rice find could hasten development of nonvolatile quantum memory April 5th, 2024
Govt.-Legislation/Regulation/Funding/Policy
![]() NRL charters Navy�s quantum inertial navigation path to reduce drift April 5th, 2024
NRL charters Navy�s quantum inertial navigation path to reduce drift April 5th, 2024
![]() Discovery points path to flash-like memory for storing qubits: Rice find could hasten development of nonvolatile quantum memory April 5th, 2024
Discovery points path to flash-like memory for storing qubits: Rice find could hasten development of nonvolatile quantum memory April 5th, 2024
![]() Chemical reactions can scramble quantum information as well as black holes April 5th, 2024
Chemical reactions can scramble quantum information as well as black holes April 5th, 2024
Possible Futures
![]() Discovery points path to flash-like memory for storing qubits: Rice find could hasten development of nonvolatile quantum memory April 5th, 2024
Discovery points path to flash-like memory for storing qubits: Rice find could hasten development of nonvolatile quantum memory April 5th, 2024
![]() With VECSELs towards the quantum internet Fraunhofer: IAF achieves record output power with VECSEL for quantum frequency converters April 5th, 2024
With VECSELs towards the quantum internet Fraunhofer: IAF achieves record output power with VECSEL for quantum frequency converters April 5th, 2024
Memory Technology
![]() Utilizing palladium for addressing contact issues of buried oxide thin film transistors April 5th, 2024
Utilizing palladium for addressing contact issues of buried oxide thin film transistors April 5th, 2024
![]() Interdisciplinary: Rice team tackles the future of semiconductors Multiferroics could be the key to ultralow-energy computing October 6th, 2023
Interdisciplinary: Rice team tackles the future of semiconductors Multiferroics could be the key to ultralow-energy computing October 6th, 2023
![]() Researchers discover materials exhibiting huge magnetoresistance June 9th, 2023
Researchers discover materials exhibiting huge magnetoresistance June 9th, 2023
Nanoelectronics
![]() Interdisciplinary: Rice team tackles the future of semiconductors Multiferroics could be the key to ultralow-energy computing October 6th, 2023
Interdisciplinary: Rice team tackles the future of semiconductors Multiferroics could be the key to ultralow-energy computing October 6th, 2023
![]() Key element for a scalable quantum computer: Physicists from Forschungszentrum J�lich and RWTH Aachen University demonstrate electron transport on a quantum chip September 23rd, 2022
Key element for a scalable quantum computer: Physicists from Forschungszentrum J�lich and RWTH Aachen University demonstrate electron transport on a quantum chip September 23rd, 2022
![]() Reduced power consumption in semiconductor devices September 23rd, 2022
Reduced power consumption in semiconductor devices September 23rd, 2022
![]() Atomic level deposition to extend Moore�s law and beyond July 15th, 2022
Atomic level deposition to extend Moore�s law and beyond July 15th, 2022
Discoveries
![]() Chemical reactions can scramble quantum information as well as black holes April 5th, 2024
Chemical reactions can scramble quantum information as well as black holes April 5th, 2024
![]() New micromaterial releases nanoparticles that selectively destroy cancer cells April 5th, 2024
New micromaterial releases nanoparticles that selectively destroy cancer cells April 5th, 2024
![]() Utilizing palladium for addressing contact issues of buried oxide thin film transistors April 5th, 2024
Utilizing palladium for addressing contact issues of buried oxide thin film transistors April 5th, 2024
Announcements
![]() NRL charters Navy�s quantum inertial navigation path to reduce drift April 5th, 2024
NRL charters Navy�s quantum inertial navigation path to reduce drift April 5th, 2024
![]() Discovery points path to flash-like memory for storing qubits: Rice find could hasten development of nonvolatile quantum memory April 5th, 2024
Discovery points path to flash-like memory for storing qubits: Rice find could hasten development of nonvolatile quantum memory April 5th, 2024
|
|
||
|
|
||
| The latest news from around the world, FREE | ||
|
|
||
|
|
||
| Premium Products | ||
|
|
||
|
Only the news you want to read!
Learn More |
||
|
|
||
|
Full-service, expert consulting
Learn More |
||
|
|
||








