Home > Press > Composite nanofibers developed by Penn scientists next chapter in orthopaedic biomaterials
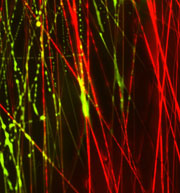 |
| Dynamic transition in a fibrous biomaterial composed of tunable fractions of structural (red) and water-soluble, sacrificial (green) electrospun polymeric nanofibers. The image was captured as fluid entered from right to left, dissolving sacrificial fibers and creating a more open fibrous network. Credit: Brendon M. Baker, PhD; Perelman School of Medicine, University of Pennsylvania. |
Abstract:
Bioengineered replacements for tendons, ligaments, the meniscus of the knee, and other tissues require re-creation of the exquisite architecture of these tissues in three dimensions. These fibrous, collagen-based tissues located throughout the body have an ordered structure that gives them their robust ability to bear extreme mechanical loading.
Composite nanofibers developed by Penn scientists next chapter in orthopaedic biomaterials
Philadelphia, PA | Posted on August 7th, 2012Many labs have been designing treatments for ACL and meniscus tears of the knee, rotator cuff injuries, and Achilles tendon ruptures for patients ranging from the weekend warrior to the elite Olympian. One popular approach has involved the use of scaffolds made from nano-sized fibers, which can guide tissue to grow in an organized way. Unfortunately, the fibers' widespread application in orthopaedics has been slowed because cells do not readily colonize the scaffolds if fibers are too tightly packed.
Robert L. Mauck, PhD, professor of Orthopaedic Surgery and Bioengineering, and Brendon M. Baker, PhD, previously a graduate student in the Mauck lab at the Perelman School of Medicine, University of Pennsylvania, have developed and validated a new technology in which composite nanofibrous scaffolds provide a loose enough structure for cells to colonize without impediment, but still can instruct cells how to lay down new tissue. Their findings appear online this week in the Proceedings of the National Academy of Sciences.
"These are tiny fibers with a huge potential that can be unlocked by including a temporary, space-holding element," says Mauck. The fibers are on the order of nanometers in diameter. A nanometer is a billionth of a meter.
Using a method that has been around since the 1930s called electrospinning, the team made composites containing two distinct fiber types: a slow-degrading polymer and a water-soluble polymer that can be selectively removed to increase or decrease the spacing between fibers. The fibers are made by electrically charging solutions of dissolved polymers, causing the solution to erupt as a fine spray of fibers which fall like snow onto a rotating drum and collect as a stretchable fabric. This textile can then be shaped for medical applications and cells can be added, or it can be implanted directly -- as a patch of sorts -- into damaged tissue for neighboring cells to colonize.
Increasing the proportion of the dissolving fibers enhanced the ability of host cells to colonize the nanofiber mesh and eventually migrate to achieve a uniform distribution and form a truly three- dimensional tissue. Despite the removal of more than 50 percent of the initial fibers, the remaining scaffold was a sufficient architecture to align cells and direct the formation of a highly organized extracellular matrix by collagen-producing cells. This, in turn, led to a biologic material with tensile properties nearly matching human meniscus tissue, in lab tests of tissue mechanics.
"This approach transforms what was once an interesting biomaterials phenomenon -- cells on the surface of nanofibrous mats -- into a method by which functional, three-dimensional tissues can be formed," says Mauck.
It is a marked step forward in the engineering of load-bearing fibrous tissues, and will eventually find widespread applications in regenerative medicine, say the authors.
Mauck and his team are currently testing these novel materials in a large animal model of meniscus repair and for other orthopaedic applications.
Co-authors are Roshan P. Shah, Amy M. Silverstein, and Jason A. Burdick, all from Penn, and John L. Esterhai, from the Philadelphia VA Medical Center.
This work was supported by National Institutes of Health Grant R01 AR056624 from the National Institute of Arthritis and Musculoskeletal and Skin Diseases and a Department of Veterans Affairs Grant I01 RX000174.
####
About University of Pennsylvania School of Medicine
Penn Medicine is one of the world's leading academic medical centers, dedicated to the related missions of medical education, biomedical research, and excellence in patient care. Penn Medicine consists of the Raymond and Ruth Perelman School of Medicine at the University of Pennsylvania (founded in 1765 as the nation's first medical school) and the University of Pennsylvania Health System, which together form a $4.3 billion enterprise.
The Perelman School of Medicine is currently ranked #2 in U.S. News & World Report's survey of research-oriented medical schools. The School is consistently among the nation's top recipients of funding from the National Institutes of Health, with $479.3 million awarded in the 2011 fiscal year.
The University of Pennsylvania Health System's patient care facilities include: The Hospital of the University of Pennsylvania -- recognized as one of the nation's top "Honor Roll" hospitals by U.S. News & World Report; Penn Presbyterian Medical Center; and Pennsylvania Hospital - the nation's first hospital, founded in 1751. Penn Medicine also includes additional patient care facilities and services throughout the Philadelphia region.
Penn Medicine is committed to improving lives and health through a variety of community-based programs and activities. In fiscal year 2011, Penn Medicine provided $854 million to benefit our community.
For more information, please click here
Contacts:
Karen Kreeger
215-349-5658
Copyright © University of Pennsylvania School of Medicine
If you have a comment, please Contact us.Issuers of news releases, not 7th Wave, Inc. or Nanotechnology Now, are solely responsible for the accuracy of the content.
| Related News Press |
News and information
![]() Simulating magnetization in a Heisenberg quantum spin chain April 5th, 2024
Simulating magnetization in a Heisenberg quantum spin chain April 5th, 2024
![]() NRL charters Navy�s quantum inertial navigation path to reduce drift April 5th, 2024
NRL charters Navy�s quantum inertial navigation path to reduce drift April 5th, 2024
![]() Discovery points path to flash-like memory for storing qubits: Rice find could hasten development of nonvolatile quantum memory April 5th, 2024
Discovery points path to flash-like memory for storing qubits: Rice find could hasten development of nonvolatile quantum memory April 5th, 2024
Govt.-Legislation/Regulation/Funding/Policy
![]() NRL charters Navy�s quantum inertial navigation path to reduce drift April 5th, 2024
NRL charters Navy�s quantum inertial navigation path to reduce drift April 5th, 2024
![]() Discovery points path to flash-like memory for storing qubits: Rice find could hasten development of nonvolatile quantum memory April 5th, 2024
Discovery points path to flash-like memory for storing qubits: Rice find could hasten development of nonvolatile quantum memory April 5th, 2024
![]() Chemical reactions can scramble quantum information as well as black holes April 5th, 2024
Chemical reactions can scramble quantum information as well as black holes April 5th, 2024
Nanomedicine
![]() New micromaterial releases nanoparticles that selectively destroy cancer cells April 5th, 2024
New micromaterial releases nanoparticles that selectively destroy cancer cells April 5th, 2024
![]() Good as gold - improving infectious disease testing with gold nanoparticles April 5th, 2024
Good as gold - improving infectious disease testing with gold nanoparticles April 5th, 2024
![]() Researchers develop artificial building blocks of life March 8th, 2024
Researchers develop artificial building blocks of life March 8th, 2024
Discoveries
![]() Chemical reactions can scramble quantum information as well as black holes April 5th, 2024
Chemical reactions can scramble quantum information as well as black holes April 5th, 2024
![]() New micromaterial releases nanoparticles that selectively destroy cancer cells April 5th, 2024
New micromaterial releases nanoparticles that selectively destroy cancer cells April 5th, 2024
![]() Utilizing palladium for addressing contact issues of buried oxide thin film transistors April 5th, 2024
Utilizing palladium for addressing contact issues of buried oxide thin film transistors April 5th, 2024
Materials/Metamaterials/Magnetoresistance
![]() Nanoscale CL thermometry with lanthanide-doped heavy-metal oxide in TEM March 8th, 2024
Nanoscale CL thermometry with lanthanide-doped heavy-metal oxide in TEM March 8th, 2024
![]() Focused ion beam technology: A single tool for a wide range of applications January 12th, 2024
Focused ion beam technology: A single tool for a wide range of applications January 12th, 2024
Announcements
![]() NRL charters Navy�s quantum inertial navigation path to reduce drift April 5th, 2024
NRL charters Navy�s quantum inertial navigation path to reduce drift April 5th, 2024
![]() Discovery points path to flash-like memory for storing qubits: Rice find could hasten development of nonvolatile quantum memory April 5th, 2024
Discovery points path to flash-like memory for storing qubits: Rice find could hasten development of nonvolatile quantum memory April 5th, 2024
Military
![]() NRL charters Navy�s quantum inertial navigation path to reduce drift April 5th, 2024
NRL charters Navy�s quantum inertial navigation path to reduce drift April 5th, 2024
![]() What heat can tell us about battery chemistry: using the Peltier effect to study lithium-ion cells March 8th, 2024
What heat can tell us about battery chemistry: using the Peltier effect to study lithium-ion cells March 8th, 2024
![]() New chip opens door to AI computing at light speed February 16th, 2024
New chip opens door to AI computing at light speed February 16th, 2024
|
|
||
|
|
||
| The latest news from around the world, FREE | ||
|
|
||
|
|
||
| Premium Products | ||
|
|
||
|
Only the news you want to read!
Learn More |
||
|
|
||
|
Full-service, expert consulting
Learn More |
||
|
|
||








