 |
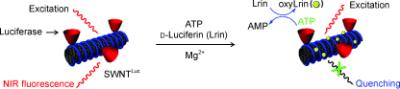
| Take the tube: The title conjugate provides spatiotemporal information of ATP in living cells by a two-step fluorescence quenching mechanism. OxyLrin, which is generated enzymatically in the reaction between ATP and D-luciferin (Lrin), is adsorbed onto the nanotubes and quenches their fluorescence (see picture). The sensor is highly selective toward ATP. |
Abstract:
Sensitive, selective, and resolved in time and space: ATP detection in living cells with carbon nanotubes and luciferase
Switched Off
Weinheim, Germany | Posted on February 12th, 2010All living cells require a fuel to function: adenosine triphosphate (ATP), the cell "gasoline". Detecting ATP within cells can help researchers observe energetic physiological processes, such as signal cascades or transport processes. Furthermore, ATP depletion is related to certain diseases, such as Parkinson's disease and ischemia (restricted blood flow within tissues). A team led by Michael S. Strano at the Massachusetts Institute of Technology in Cambridge (USA) has now developed a more sensitive, higher-resolution, and more robust method for the detection of ATP. As the scientists report in the journal Angewandte Chemie, the method is based on carbon nanotubes.
ATP is usually detected by means of the luciferase assay. Luciferases are enzymes that are used in fireflies and other bioluminescent organisms to produce light. They use oxygen to convert a substrate called luciferin into oxyluciferin, which then reacts further to produce light. Certain luciferases use ATP for their reactions. The luciferase assay currently in use is complex, time-consuming, and suffers from a poor signal-to-noise ratio.
The MIT team has now developed a variation of the luciferase protocol: They attached the luciferase to carbon nanotubes. In this form the enzyme is easily taken up by cells. In the presence of luciferin and ATP, oxyluciferin is formed as usual, which causes fluorescence. What is interesting in this case is that carbon nanotubes normally fluoresce in the near infrared (nIR) spectral region; however this is proportionately extinguished by the addition of ATP to the luciferase reaction. Why? "As it is formed, the product oxyluciferin attaches itself firmly to the nanotube," explains Strano. "Electrons are transferred from the nanotube to the oxyluciferin so that the carbon nanotube itself can no longer fluoresce." The reduction in nIR fluorescence is easy to detect and serves as an indicator of the ATP concentration.
"Our new sensor is very selective for ATP," continues Strano. "We were able to use it to observe the change in ATP concentration over time and space in a cell culture."
Author: Michael S. Strano, Massachusetts Institute of Technology, Cambridge (USA), web.mit.edu/stranogroup/
Title: A Luciferase/Single-walled Carbon Nanotube Conjugate for Near-Infrared Fluorescent Detection of Cellular ATP
Angewandte Chemie International Edition 2010, 49, No. 8, 1456-1459, Permalink: dx.doi.org/10.1002/anie.200906251
####
About Wiley InterScience
Wiley InterScience (www.interscience.wiley.com) provides access to over 3 million articles across nearly 1500 journals and 7000 Online Books and major reference works. It also holds industry leading databases such as The Cochrane Library, chemistry databases and the acclaimed Current Protocols laboratory manuals.
Wiley InterScience is one of the world's premiere resources for study, teaching and advanced research.
For more information, please click here
Contacts:
Editorial office
Amy Molnar (US)
Jennifer Beal (UK)
Alina Boey (Asia)
Copyright © Wiley InterScience
If you have a comment, please Contact us.Issuers of news releases, not 7th Wave, Inc. or Nanotechnology Now, are solely responsible for the accuracy of the content.
| Related News Press |
News and information
![]() Simulating magnetization in a Heisenberg quantum spin chain April 5th, 2024
Simulating magnetization in a Heisenberg quantum spin chain April 5th, 2024
![]() NRL charters Navy�s quantum inertial navigation path to reduce drift April 5th, 2024
NRL charters Navy�s quantum inertial navigation path to reduce drift April 5th, 2024
![]() Discovery points path to flash-like memory for storing qubits: Rice find could hasten development of nonvolatile quantum memory April 5th, 2024
Discovery points path to flash-like memory for storing qubits: Rice find could hasten development of nonvolatile quantum memory April 5th, 2024
Nanotubes/Buckyballs/Fullerenes/Nanorods/Nanostrings
![]() Tests find no free-standing nanotubes released from tire tread wear September 8th, 2023
Tests find no free-standing nanotubes released from tire tread wear September 8th, 2023
![]() Detection of bacteria and viruses with fluorescent nanotubes July 21st, 2023
Detection of bacteria and viruses with fluorescent nanotubes July 21st, 2023
Nanomedicine
![]() New micromaterial releases nanoparticles that selectively destroy cancer cells April 5th, 2024
New micromaterial releases nanoparticles that selectively destroy cancer cells April 5th, 2024
![]() Good as gold - improving infectious disease testing with gold nanoparticles April 5th, 2024
Good as gold - improving infectious disease testing with gold nanoparticles April 5th, 2024
![]() Researchers develop artificial building blocks of life March 8th, 2024
Researchers develop artificial building blocks of life March 8th, 2024
Sensors
Announcements
![]() NRL charters Navy�s quantum inertial navigation path to reduce drift April 5th, 2024
NRL charters Navy�s quantum inertial navigation path to reduce drift April 5th, 2024
![]() Discovery points path to flash-like memory for storing qubits: Rice find could hasten development of nonvolatile quantum memory April 5th, 2024
Discovery points path to flash-like memory for storing qubits: Rice find could hasten development of nonvolatile quantum memory April 5th, 2024
Nanobiotechnology
![]() New micromaterial releases nanoparticles that selectively destroy cancer cells April 5th, 2024
New micromaterial releases nanoparticles that selectively destroy cancer cells April 5th, 2024
![]() Good as gold - improving infectious disease testing with gold nanoparticles April 5th, 2024
Good as gold - improving infectious disease testing with gold nanoparticles April 5th, 2024
![]() Researchers develop artificial building blocks of life March 8th, 2024
Researchers develop artificial building blocks of life March 8th, 2024
|
|
||
|
|
||
| The latest news from around the world, FREE | ||
|
|
||
|
|
||
| Premium Products | ||
|
|
||
|
Only the news you want to read!
Learn More |
||
|
|
||
|
Full-service, expert consulting
Learn More |
||
|
|
||








