Home > Press > 'Nanochains' could increase battery capacity, cut charging time
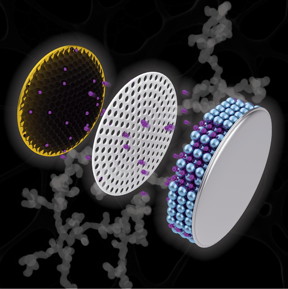 |
| Artistic depiction of a coin cell battery with a copper electrode (left) containing a black nanochain structure, which researchers have discovered could increase the capacity of a battery and cut charging time. CREDIT Purdue University illustration/Henry Hamann |
Abstract:
Three-Dimensional Antimony Nanochains for Lithium-Ion Storage
Jassiel R. Rodriguez, Henry J. Hamann, Garrett M. Mitchell, Volkan Ortalan, Vilas G. Pol, P. Veeraraghavan Ramachandran
Purdue University, West Lafayette, IN, USA
DOI: 10.1021/acsanm.9b01316
Three-dimensional antimony nanochain architectures with a rhombohedral phase, possessing particle sizes of ~30 nm, have been prepared via ammonia-borane reduction of SbCl3 in an aqueous medium, followed by nucleation and capping processes. These offer adequate space to abate the large volumetric expansion during lithiation. Lithium-ion batteries fabricated with these antimony nanochains exhibited a stable specific charge capacity of 523 mAh g-1 at a C rate of 0.5 C with a Coulombic efficiency of 99.8% and a capacity retention of 92% after 100 cycles.
'Nanochains' could increase battery capacity, cut charging time
West Lafayette, IN | Posted on September 20th, 2019How long the battery of your phone or computer lasts depends on how many lithium ions can be stored in the battery's negative electrode material. If the battery runs out of these ions, it can't generate an electrical current to run a device and ultimately fails.
Materials with a higher lithium ion storage capacity are either too heavy or the wrong shape to replace graphite, the electrode material currently used in today's batteries.
Purdue University scientists and engineers have introduced a potential way that these materials could be restructured into a new electrode design that would allow them to increase a battery's lifespan, make it more stable and shorten its charging time.
The study, appearing as the cover of the September issue of Applied Nano Materials, created a net-like structure, called a "nanochain," of antimony, a metalloid known to enhance lithium ion charge capacity in batteries.
The researchers compared the nanochain electrodes to graphite electrodes, finding that when coin cell batteries with the nanochain electrode were only charged for 30 minutes, they achieved double the lithium-ion capacity for 100 charge-discharge cycles.
Some types of commercial batteries already use carbon-metal composites similar to antimony metal negative electrodes, but the material tends to expand up to three times as it takes in lithium ions, causing it to become a safety hazard as the battery charges.
"You want to accommodate that type of expansion in your smartphone batteries. That way you're not carrying around something unsafe," said Vilas Pol, a Purdue associate professor of chemical engineering.
Through applying chemical compounds - a reducing agent and a nucleating agent - Purdue scientists connected the tiny antimony particles into a nanochain shape that would accommodate the required expansion. The particular reducing agent the team used, ammonia-borane, is responsible for creating the empty spaces - the pores inside the nanochain - that accommodate expansion and suppress electrode failure.
The team applied ammonia-borane to several different compounds of antimony, finding that only antimony-chloride produced the nanochain structure.
"Our procedure to make the nanoparticles consistently provides the chain structures," said P. V. Ramachandran, a professor of organic chemistry at Purdue.
The nanochain also keeps lithium ion capacity stable for at least 100 charging-discharging cycles. "There's essentially no change from cycle 1 to cycle 100, so we have no reason to think that cycle 102 won't be the same," Pol said.
Henry Hamann, a chemistry graduate student at Purdue, synthesized the antimony nanochain structure and Jassiel Rodriguez, a Purdue chemical engineering postdoctoral candidate, tested the electrochemical battery performance.
The electrode design has the potential to be scalable for larger batteries, the researchers say. The team plans to test the design in pouch cell batteries next.
###
This work was financially supported by the Herbert C. Brown Center for Borane Research.
####
For more information, please click here
Contacts:
Kayla Wiles
765-494-2432
Copyright © Purdue University
If you have a comment, please Contact us.Issuers of news releases, not 7th Wave, Inc. or Nanotechnology Now, are solely responsible for the accuracy of the content.
| Related Links |
| Related News Press |
News and information
![]() Simulating magnetization in a Heisenberg quantum spin chain April 5th, 2024
Simulating magnetization in a Heisenberg quantum spin chain April 5th, 2024
![]() NRL charters Navy�s quantum inertial navigation path to reduce drift April 5th, 2024
NRL charters Navy�s quantum inertial navigation path to reduce drift April 5th, 2024
![]() Discovery points path to flash-like memory for storing qubits: Rice find could hasten development of nonvolatile quantum memory April 5th, 2024
Discovery points path to flash-like memory for storing qubits: Rice find could hasten development of nonvolatile quantum memory April 5th, 2024
Possible Futures
![]() Discovery points path to flash-like memory for storing qubits: Rice find could hasten development of nonvolatile quantum memory April 5th, 2024
Discovery points path to flash-like memory for storing qubits: Rice find could hasten development of nonvolatile quantum memory April 5th, 2024
![]() With VECSELs towards the quantum internet Fraunhofer: IAF achieves record output power with VECSEL for quantum frequency converters April 5th, 2024
With VECSELs towards the quantum internet Fraunhofer: IAF achieves record output power with VECSEL for quantum frequency converters April 5th, 2024
Discoveries
![]() Chemical reactions can scramble quantum information as well as black holes April 5th, 2024
Chemical reactions can scramble quantum information as well as black holes April 5th, 2024
![]() New micromaterial releases nanoparticles that selectively destroy cancer cells April 5th, 2024
New micromaterial releases nanoparticles that selectively destroy cancer cells April 5th, 2024
![]() Utilizing palladium for addressing contact issues of buried oxide thin film transistors April 5th, 2024
Utilizing palladium for addressing contact issues of buried oxide thin film transistors April 5th, 2024
Announcements
![]() NRL charters Navy�s quantum inertial navigation path to reduce drift April 5th, 2024
NRL charters Navy�s quantum inertial navigation path to reduce drift April 5th, 2024
![]() Discovery points path to flash-like memory for storing qubits: Rice find could hasten development of nonvolatile quantum memory April 5th, 2024
Discovery points path to flash-like memory for storing qubits: Rice find could hasten development of nonvolatile quantum memory April 5th, 2024
Interviews/Book Reviews/Essays/Reports/Podcasts/Journals/White papers/Posters
![]() Simulating magnetization in a Heisenberg quantum spin chain April 5th, 2024
Simulating magnetization in a Heisenberg quantum spin chain April 5th, 2024
![]() Discovery points path to flash-like memory for storing qubits: Rice find could hasten development of nonvolatile quantum memory April 5th, 2024
Discovery points path to flash-like memory for storing qubits: Rice find could hasten development of nonvolatile quantum memory April 5th, 2024
Battery Technology/Capacitors/Generators/Piezoelectrics/Thermoelectrics/Energy storage
![]() What heat can tell us about battery chemistry: using the Peltier effect to study lithium-ion cells March 8th, 2024
What heat can tell us about battery chemistry: using the Peltier effect to study lithium-ion cells March 8th, 2024
![]() A battery�s hopping ions remember where they�ve been: Seen in atomic detail, the seemingly smooth flow of ions through a battery�s electrolyte is surprisingly complicated February 16th, 2024
A battery�s hopping ions remember where they�ve been: Seen in atomic detail, the seemingly smooth flow of ions through a battery�s electrolyte is surprisingly complicated February 16th, 2024
Grants/Sponsored Research/Awards/Scholarships/Gifts/Contests/Honors/Records
![]() Discovery points path to flash-like memory for storing qubits: Rice find could hasten development of nonvolatile quantum memory April 5th, 2024
Discovery points path to flash-like memory for storing qubits: Rice find could hasten development of nonvolatile quantum memory April 5th, 2024
![]() Chemical reactions can scramble quantum information as well as black holes April 5th, 2024
Chemical reactions can scramble quantum information as well as black holes April 5th, 2024
|
|
||
|
|
||
| The latest news from around the world, FREE | ||
|
|
||
|
|
||
| Premium Products | ||
|
|
||
|
Only the news you want to read!
Learn More |
||
|
|
||
|
Full-service, expert consulting
Learn More |
||
|
|
||








