Home > Press > Chemical interactions between silver nanoparticles and thiols: A comparison of mercaptohexanol again
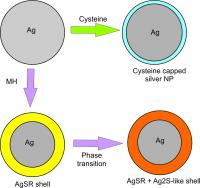 |
| This figure shows the interaction between silver nanoparticles and the two thiols of 6-mercaptohexanol (MH) and cysteine.
Credit: �Science China Press |
Abstract:
Silver nanoparticles are well known for their anti-bacteria properties[1-4]. One of the main routes by which they may act as an anti-bacteria agent, is through attaching themselves to the thiol group present on the cellular membrane surface and hence disrupting the membrane's function[5]. Hence, it is crucial to gain a greater understanding of this complex silver-thiol interaction to determine silver nanoparticles' role in biological systems. With thiols, silver nanoparticles have been proposed to form various types of compounds with different structures[6-8]. One of the plausible reaction routes suggested for organothiols is[6,7]: 4 RSH + 4 Ag + O2 4 AgSR + 2 H2O (1)
Chemical interactions between silver nanoparticles and thiols: A comparison of mercaptohexanol again
Beijing, China | Posted on September 30th, 2014It has been shown through scanning electron microscopy that AgSR remained on the surface, forming a shell[6]. Battocchio et al. suggested a shell of more than one species of silver thiolates - a mix of AgSR and Ag2S-like complexes were formed when aromatic organothiols were added during silver nanoparticle synthesis[8]. From the above, it known that the interaction between silver nanoparticles and thiol is complicated. Therefore, in this study, we presented a comparative view of silver-thiol interaction for MH and cysteine through voltammetric and spectroscopy experiments.
Cyclic voltammograms were performed in the presence of MH or cysteine to ascertain the changes in the ease and extent of oxidation of silver nanoparticles to silver(I) ions. The UV-vis spectra of silver nanoparticles were recorded to examine the changes in the nanoparticles' surface properties in the presence of thiols. From the experimental data, it was concluded that MH interacts with silver nanoparticles to give a sparingly soluble silver(I) thiolate complex AgSRm (Rm = -(CH2)6OH) on the surface. It was also inferred that the AgSRm complex on the nanoparticle surface undergoes a phase transition to give a mixture of AgSRm and Ag2S-like complexes due to the presence of two signals in both electrochemical voltammograms and UV-vis spectrums[8]. In contrast, when silver nanoparticles were exposed to cysteine, the citrate capping agent on the silver nanoparticles was replaced by cysteine to give cysteine capped nanoparticles. As cysteine capped nanoparticles form, the electrochemical data displayed a decrease in oxidative peak charge but the UV-vis spectra showed a constant signal. Therefore, cysteine capped nanoparticles were suggested to have either inactivated the silver surface or else promoted detachment from the electrode surface. Figure 1 shows schematically the route which silver nanoparticles interact with MH and cysteine.
It may be concluded that no general mechanism for the interactions of thiol with silver nanoparticles exist and that each thiol must be treated individually.
References
[1]R. K. Kunkalekar, M. M. Naik, S. K. Dubey, A. V. Salker, J. Chem. Technol. Biotechnol. 2013, 88, 873-877.
[2]Y. Park, H. J. Noh, L. Han, H.-S. Kim, Y.-J. Kim, J. S. Choi, C.-K. Kim, Y. S. Kim, S. Cho, J. Nanosci. Nanotechnol. 2012, 12, 7087-7095.
[3]M. Sureshkumar, D. Y. Siswanto, C.-K. Lee, J. Mater. Chem. 2010, 20, 6948.
[4]C. Marambio-Jones, E. M. V. Hoek, J.Nanopart. Res. 2010, 12, 1531-1551.
[5]A. Lapresta-Fern�ndez, A. Fern�ndez, J. Blasco, TrAC, 2012, 32, 40-59.
[6]S. M. Ansar, G. S. Perera, P. Gomez, G. Salomon, E. S. Vasquez, I. W. Chu, S. Zou, C. U. Pittman, K. B. Walters, D. Zhang, J. Phys. Chem. C 2013, 117, 27146-27154.
[7]A. Andrieux-Ledier, B. Tremblay, A. Courty, Langmuir 2013, 29, 13140-13145.
[8]C. Battocchio, C. Meneghini, I. Fratoddi, I. Venditti, M. V. Russo, G. Aquilanti, C. Maurizio, F. Bondino, R. Matassa, M. Rossi, S. Mobilio, G. Polzonetti, J. Phys. Chem. C 2012, 116, 19571-19578.
####
For more information, please click here
Contacts:
Richard G. Compton
Copyright © Science China Press
If you have a comment, please Contact us.Issuers of news releases, not 7th Wave, Inc. or Nanotechnology Now, are solely responsible for the accuracy of the content.
| Related Links |
| Related News Press |
News and information
![]() Simulating magnetization in a Heisenberg quantum spin chain April 5th, 2024
Simulating magnetization in a Heisenberg quantum spin chain April 5th, 2024
![]() NRL charters Navy�s quantum inertial navigation path to reduce drift April 5th, 2024
NRL charters Navy�s quantum inertial navigation path to reduce drift April 5th, 2024
![]() Discovery points path to flash-like memory for storing qubits: Rice find could hasten development of nonvolatile quantum memory April 5th, 2024
Discovery points path to flash-like memory for storing qubits: Rice find could hasten development of nonvolatile quantum memory April 5th, 2024
Chemistry
![]() What heat can tell us about battery chemistry: using the Peltier effect to study lithium-ion cells March 8th, 2024
What heat can tell us about battery chemistry: using the Peltier effect to study lithium-ion cells March 8th, 2024
![]() Nanoscale CL thermometry with lanthanide-doped heavy-metal oxide in TEM March 8th, 2024
Nanoscale CL thermometry with lanthanide-doped heavy-metal oxide in TEM March 8th, 2024
Discoveries
![]() Chemical reactions can scramble quantum information as well as black holes April 5th, 2024
Chemical reactions can scramble quantum information as well as black holes April 5th, 2024
![]() New micromaterial releases nanoparticles that selectively destroy cancer cells April 5th, 2024
New micromaterial releases nanoparticles that selectively destroy cancer cells April 5th, 2024
![]() Utilizing palladium for addressing contact issues of buried oxide thin film transistors April 5th, 2024
Utilizing palladium for addressing contact issues of buried oxide thin film transistors April 5th, 2024
Announcements
![]() NRL charters Navy�s quantum inertial navigation path to reduce drift April 5th, 2024
NRL charters Navy�s quantum inertial navigation path to reduce drift April 5th, 2024
![]() Discovery points path to flash-like memory for storing qubits: Rice find could hasten development of nonvolatile quantum memory April 5th, 2024
Discovery points path to flash-like memory for storing qubits: Rice find could hasten development of nonvolatile quantum memory April 5th, 2024
Interviews/Book Reviews/Essays/Reports/Podcasts/Journals/White papers/Posters
![]() Simulating magnetization in a Heisenberg quantum spin chain April 5th, 2024
Simulating magnetization in a Heisenberg quantum spin chain April 5th, 2024
![]() Discovery points path to flash-like memory for storing qubits: Rice find could hasten development of nonvolatile quantum memory April 5th, 2024
Discovery points path to flash-like memory for storing qubits: Rice find could hasten development of nonvolatile quantum memory April 5th, 2024
|
|
||
|
|
||
| The latest news from around the world, FREE | ||
|
|
||
|
|
||
| Premium Products | ||
|
|
||
|
Only the news you want to read!
Learn More |
||
|
|
||
|
Full-service, expert consulting
Learn More |
||
|
|
||








