Home > Press > Nano world: Where towers construct themselves: How physicists get control on the self-assembly process
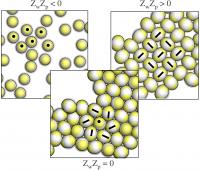 |
| Overall neutral IPCs do not assemble in the vicinity of a charged bottom wall which attract the patches (left), assemble into a close packed structure when the substrate is neutral (center), and form open aggregates when the wall attract the equators (right).
Credit: Copyright: American Chemical Society/ACS 2014 |
Abstract:
Imagine a tower builds itself into the desired structure only by choosing the appropriate bricks. Absurd - and however, in the nano world this is reality: There an unordered crowd of components can initiate the formation of an ordered structure - a process known as self-assembly. The physicists Christos Likos (University of Vienna), Emanuela Bianchi and Gerhard Kahl (both Vienna University of Technology) investigate how they can control the ordering of such self-assembling structures and found out how to switch the assembly process on and off. The results are now published in the high-impact journal Nano Letters.
Nano world: Where towers construct themselves: How physicists get control on the self-assembly process
Vienna, Austria | Posted on June 2nd, 2014Materials with specific properties at the nano- and micro-scale level are heavily sought after due to the broad spectrum of applications in electronics, photovoltaics and biomimetic material synthesis. For many of these applications, mesoscopic crystalline structures are often needed and hence scientists have to deal with the challenge of developing reliable, efficient and cheap methods to produce target structures with specific symmetries and physical properties. Nowadays, rather than relying on externally-controlled tools, most of the fabrication methods are based on the self-assembly of carefully chosen/synthesized base units. The macroscopic counterpart would correspond to building a tower or a bridge just by choosing the appropriate bricks and letting them self-organize into the desired structure.
In the vast realm of functional nano- and micro-scale materials, the realization of mono- and bi-layer assemblies on surfaces is of paramount relevance. Low-dimensional systems with well-defined features have indeed applications as e.g. antireflection coatings, biosensors, data-storage, optical and photovoltaic devices, or catalysts. The properties of these materials strongly depend on a delicate balance between the properties of the assembling units and those of the underlying surface.
In our contribution we focused on nano-units with a complicated surface pattern, consisting of regions with different surface charge. The investigated units are mostly negatively charged with the exception of the positively charged polar regions on the top and at the bottom of the particles. Similar non-homogeneously charged units appear either in biosystems, e.g. viral capsids and proteins, or in experimentally synthesized systems, e.g. virus-like nano-particles, spotted vesicles and nano-cubes covered with specific metals.
In the up-coming paper we focused on the self-assembly of the described heterogeneously charged particles in the vicinity of a homogeneously charged substrate. Our computer simulations showed how complex structures at the nano-scale level can spontaneously emerge and how it is possible to reliably control the ordering of the particles into specific, quasi two-dimensional aggregates. Depending on different parameters, such as the particle/wall charge and the extension of the charged regions on the particle surface, our units can form surface layers with different densities (and possibly different responses to external stimuli): sometimes particles assemble into close-packed, hexagonally ordered crystalline aggregates, sometimes they form open, square-like layers, sometimes they do not assemble at all. Our work was able to investigate the variety of the self-assembled structures offered by the chosen bricks and to characterise the specific collective behaviors occurring on tuning the relevant parameters of these systems. Moreover and most importantly, we showed that upon subtle changes of either the pH of the solution or of the electrical charge of the substrate, it is possible to reversibly switch the assembly process on and off as well as to induce a transformation from a one specific spatial/orientational arrangement to another.
####
About University of Vienna
The University of Vienna, founded in 1365, is one of the oldest and largest universities in Europe. About 9,500 employees, 6,700 of who are academic employees, work at 15 faculties and four centres. This makes the University of Vienna Austria's largest research and education institution. About 92,000 national and international students are currently enrolled at the University of Vienna. With more than 180 degree programmes, the University offers the most diverse range of studies in Austria. The University of Vienna is also a major provider of continuing education. In 2015, the Alma Mater Rudolphina Vindobonensis celebrates its 650th Anniversary.
For more information, please click here
Contacts:
Christos Likos
43-142-777-3230
Copyright © University of Vienna
If you have a comment, please Contact us.Issuers of news releases, not 7th Wave, Inc. or Nanotechnology Now, are solely responsible for the accuracy of the content.
| Related Links |
| Related News Press |
News and information
![]() Quantum computer improves AI predictions April 17th, 2026
Quantum computer improves AI predictions April 17th, 2026
![]() Flexible sensor gains sensitivity under pressure April 17th, 2026
Flexible sensor gains sensitivity under pressure April 17th, 2026
![]() A reusable chip for particulate matter sensing April 17th, 2026
A reusable chip for particulate matter sensing April 17th, 2026
![]() Detecting vibrational quantum beating in the predissociation dynamics of SF6 using time-resolved photoelectron spectroscopy April 17th, 2026
Detecting vibrational quantum beating in the predissociation dynamics of SF6 using time-resolved photoelectron spectroscopy April 17th, 2026
Physics
![]() UC Irvine physicists discover method to reverse �quantum scrambling� : The work addresses the problem of information loss in quantum computing system April 17th, 2026
UC Irvine physicists discover method to reverse �quantum scrambling� : The work addresses the problem of information loss in quantum computing system April 17th, 2026
![]() Quantum computers simulate fundamental physics: shedding light on the building blocks of nature June 6th, 2025
Quantum computers simulate fundamental physics: shedding light on the building blocks of nature June 6th, 2025
Self Assembly
![]() Diamond glitter: A play of colors with artificial DNA crystals May 17th, 2024
Diamond glitter: A play of colors with artificial DNA crystals May 17th, 2024
![]() Liquid crystal templated chiral nanomaterials October 14th, 2022
Liquid crystal templated chiral nanomaterials October 14th, 2022
![]() Nanoclusters self-organize into centimeter-scale hierarchical assemblies April 22nd, 2022
Nanoclusters self-organize into centimeter-scale hierarchical assemblies April 22nd, 2022
![]() Atom by atom: building precise smaller nanoparticles with templates March 4th, 2022
Atom by atom: building precise smaller nanoparticles with templates March 4th, 2022
Discoveries
![]() Quantum computer improves AI predictions April 17th, 2026
Quantum computer improves AI predictions April 17th, 2026
![]() Flexible sensor gains sensitivity under pressure April 17th, 2026
Flexible sensor gains sensitivity under pressure April 17th, 2026
![]() A reusable chip for particulate matter sensing April 17th, 2026
A reusable chip for particulate matter sensing April 17th, 2026
![]() Detecting vibrational quantum beating in the predissociation dynamics of SF6 using time-resolved photoelectron spectroscopy April 17th, 2026
Detecting vibrational quantum beating in the predissociation dynamics of SF6 using time-resolved photoelectron spectroscopy April 17th, 2026
Announcements
![]() A fundamentally new therapeutic approach to cystic fibrosis: Nanobody repairs cellular defect April 17th, 2026
A fundamentally new therapeutic approach to cystic fibrosis: Nanobody repairs cellular defect April 17th, 2026
![]() UC Irvine physicists discover method to reverse �quantum scrambling� : The work addresses the problem of information loss in quantum computing system April 17th, 2026
UC Irvine physicists discover method to reverse �quantum scrambling� : The work addresses the problem of information loss in quantum computing system April 17th, 2026
Interviews/Book Reviews/Essays/Reports/Podcasts/Journals/White papers/Posters
![]() A fundamentally new therapeutic approach to cystic fibrosis: Nanobody repairs cellular defect April 17th, 2026
A fundamentally new therapeutic approach to cystic fibrosis: Nanobody repairs cellular defect April 17th, 2026
![]() UC Irvine physicists discover method to reverse �quantum scrambling� : The work addresses the problem of information loss in quantum computing system April 17th, 2026
UC Irvine physicists discover method to reverse �quantum scrambling� : The work addresses the problem of information loss in quantum computing system April 17th, 2026
|
|
||
|
|
||
| The latest news from around the world, FREE | ||
|
|
||
|
|
||
| Premium Products | ||
|
|
||
|
Only the news you want to read!
Learn More |
||
|
|
||
|
Full-service, expert consulting
Learn More |
||
|
|
||








