Home > Press > Nanotechnology breakthrough is big deal for electronics
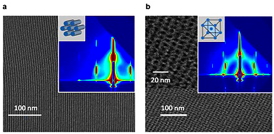 |
| Patterns of two giant surfactant samples in thin-film state. Source: Proceedings of the National Academy of Sciences of the United States of America. |
Abstract:
University of Akron researchers have developed new materials that function on a nanoscale, which could lead to the creation of lighter laptops, slimmer televisions and crisper smartphone visual displays.
Nanotechnology breakthrough is big deal for electronics
Akron, OH | Posted on July 25th, 2013Known as "giant surfactants" - or surface films and liquid solutions - the researchers, led by Stephen Z. D. Cheng, dean of UA's College of Polymer Science and Polymer Engineering, used a technique known as nanopatterning to combine functioning molecular nanoparticles with polymers to build these novel materials.
The giant surfactants developed at UA are large, similar to macromolecules, yet they function like molecular surfactants on a nanoscale, Cheng says. The outcome? Nanostructures that guide the size of electronic products.
Nanopatterning, or self-assembling molecular materials, is the genius behind the small, light and fast world of modern-day gadgetry, and now it has advanced one giant step thanks to the UA researchers who say these new materials, when integrated into electronics, will enable the development of ultra-lightweight, compact and efficient devices because of their unique structures.
During their self-assembly, molecules form an organized lithographic pattern on semiconductor crystals, for use as integrated circuits. Cheng explains that these self-assembling materials differ from common block copolymers (a portion of a macromolecule, comprising manyunits, that has at least one feature which is not present in the adjacent portions) because they organize themselves in a controllable manner at the molecular level.
"The IT industry wants microchips that are as small as possible so that they can manufacture smaller and faster devices," says Cheng, who also serves as the R.C. Musson and Trustees Professor of Polymer Science at UA.
He points out that the current technique can produce the spacing of 22 nanometers only, and cannot go down to the 10 nanometers or less necessary to create tiny, yet mighty, devices. The giant surfactants, however, can dictate smaller-scale electronic components.
"This is exactly what we are pursuing � self-assembling materials that organize at smaller sizes, say, less than 20 or even 10 nanometers," says Cheng, equating 20 nanometers to 1 /4,000th the diameter of a human hair.
An international team of experts, including George Newkome, UA vice president for research, dean of the Graduate School, and professor of Polymer Science at UA; Er-Qiang Chen of Peking University in China; Rong-Ming Ho of National Tsinghua University in Taiwan; An-Chang Shi of McMaster University in Canada; and several doctoral and postdoctoral researchers from Cheng's group, have shown how well-ordered nanostructures in various states, such as in thin films and in solution, offer extensive applications in nanotechnology.
The team's study is highlighted in a pending patent application through the University of Akron Research Foundation and in a recent journal article "Giant surfactants provide a versatile platform for sub-10-nm nanostructure engineering" published in Proceedings of the National Academy of Sciences of the United States of America (110, 10078-10083, 2013).
"These results are not only of pure scientific interest to the narrow group of scientists, but also important to a broad range of industry people," says Cheng, noting that his team is testing real-world applications in nanopatterning technologies and hope to see commercialization in the future.
####
About The University of Akron
The University of Akron offers more than 300 associate, bachelor�s, master�s, doctorate and law degree programs � with accreditations by 35 professional agencies. With nearly 30,000 students and $46.7 million in sponsored research awards, UA is among the nation's strongest public universities focused on innovation, entrepreneurship, and investment in community and economic growth. Programs are targeted to diverse groups of learners, including full-time, part-time and on-line students, veterans, and adults returning to the classroom. The distinctive Akron Experience enhances post-graduate success through internships and co-ops, academic research (both undergraduate and graduate), study abroad, on-campus student employment, and service projects.
For more information, please click here
Contacts:
Denise Henry
Phone: 330-972-6477
Fax: 330-972-6168
Copyright © The University of Akron
If you have a comment, please Contact us.Issuers of news releases, not 7th Wave, Inc. or Nanotechnology Now, are solely responsible for the accuracy of the content.
| Related Links |
| Related News Press |
News and information
![]() Quantum computer improves AI predictions April 17th, 2026
Quantum computer improves AI predictions April 17th, 2026
![]() Flexible sensor gains sensitivity under pressure April 17th, 2026
Flexible sensor gains sensitivity under pressure April 17th, 2026
![]() A reusable chip for particulate matter sensing April 17th, 2026
A reusable chip for particulate matter sensing April 17th, 2026
![]() Detecting vibrational quantum beating in the predissociation dynamics of SF6 using time-resolved photoelectron spectroscopy April 17th, 2026
Detecting vibrational quantum beating in the predissociation dynamics of SF6 using time-resolved photoelectron spectroscopy April 17th, 2026
Chip Technology
![]() A reusable chip for particulate matter sensing April 17th, 2026
A reusable chip for particulate matter sensing April 17th, 2026
![]() Metasurfaces smooth light to boost magnetic sensing precision January 30th, 2026
Metasurfaces smooth light to boost magnetic sensing precision January 30th, 2026
Self Assembly
![]() Diamond glitter: A play of colors with artificial DNA crystals May 17th, 2024
Diamond glitter: A play of colors with artificial DNA crystals May 17th, 2024
![]() Liquid crystal templated chiral nanomaterials October 14th, 2022
Liquid crystal templated chiral nanomaterials October 14th, 2022
![]() Nanoclusters self-organize into centimeter-scale hierarchical assemblies April 22nd, 2022
Nanoclusters self-organize into centimeter-scale hierarchical assemblies April 22nd, 2022
![]() Atom by atom: building precise smaller nanoparticles with templates March 4th, 2022
Atom by atom: building precise smaller nanoparticles with templates March 4th, 2022
Discoveries
![]() Quantum computer improves AI predictions April 17th, 2026
Quantum computer improves AI predictions April 17th, 2026
![]() Flexible sensor gains sensitivity under pressure April 17th, 2026
Flexible sensor gains sensitivity under pressure April 17th, 2026
![]() A reusable chip for particulate matter sensing April 17th, 2026
A reusable chip for particulate matter sensing April 17th, 2026
![]() Detecting vibrational quantum beating in the predissociation dynamics of SF6 using time-resolved photoelectron spectroscopy April 17th, 2026
Detecting vibrational quantum beating in the predissociation dynamics of SF6 using time-resolved photoelectron spectroscopy April 17th, 2026
Materials/Metamaterials/Magnetoresistance
![]() First real-time observation of two-dimensional melting process: Researchers at Mainz University unveil new insights into magnetic vortex structures August 8th, 2025
First real-time observation of two-dimensional melting process: Researchers at Mainz University unveil new insights into magnetic vortex structures August 8th, 2025
![]() Researchers unveil a groundbreaking clay-based solution to capture carbon dioxide and combat climate change June 6th, 2025
Researchers unveil a groundbreaking clay-based solution to capture carbon dioxide and combat climate change June 6th, 2025
![]() A 1960s idea inspires NBI researchers to study hitherto inaccessible quantum states June 6th, 2025
A 1960s idea inspires NBI researchers to study hitherto inaccessible quantum states June 6th, 2025
![]() Institute for Nanoscience hosts annual proposal planning meeting May 16th, 2025
Institute for Nanoscience hosts annual proposal planning meeting May 16th, 2025
Announcements
![]() A fundamentally new therapeutic approach to cystic fibrosis: Nanobody repairs cellular defect April 17th, 2026
A fundamentally new therapeutic approach to cystic fibrosis: Nanobody repairs cellular defect April 17th, 2026
![]() UC Irvine physicists discover method to reverse �quantum scrambling� : The work addresses the problem of information loss in quantum computing system April 17th, 2026
UC Irvine physicists discover method to reverse �quantum scrambling� : The work addresses the problem of information loss in quantum computing system April 17th, 2026
Interviews/Book Reviews/Essays/Reports/Podcasts/Journals/White papers/Posters
![]() A fundamentally new therapeutic approach to cystic fibrosis: Nanobody repairs cellular defect April 17th, 2026
A fundamentally new therapeutic approach to cystic fibrosis: Nanobody repairs cellular defect April 17th, 2026
![]() UC Irvine physicists discover method to reverse �quantum scrambling� : The work addresses the problem of information loss in quantum computing system April 17th, 2026
UC Irvine physicists discover method to reverse �quantum scrambling� : The work addresses the problem of information loss in quantum computing system April 17th, 2026
Patents/IP/Tech Transfer/Licensing
![]() Getting drugs across the blood-brain barrier using nanoparticles March 3rd, 2023
Getting drugs across the blood-brain barrier using nanoparticles March 3rd, 2023
![]() Metasurfaces control polarized light at will: New research unlocks the hidden potential of metasurfaces August 13th, 2021
Metasurfaces control polarized light at will: New research unlocks the hidden potential of metasurfaces August 13th, 2021
![]() Arrowhead Pharmaceuticals Announces Closing of Agreement with Takeda November 27th, 2020
Arrowhead Pharmaceuticals Announces Closing of Agreement with Takeda November 27th, 2020
Industrial
![]() Tiny nanosheets, big leap: A new sensor detects ethanol at ultra-low levels January 30th, 2026
Tiny nanosheets, big leap: A new sensor detects ethanol at ultra-low levels January 30th, 2026
![]() Quantum interference in molecule-surface collisions February 28th, 2025
Quantum interference in molecule-surface collisions February 28th, 2025
![]() Boron nitride nanotube fibers get real: Rice lab creates first heat-tolerant, stable fibers from wet-spinning process June 24th, 2022
Boron nitride nanotube fibers get real: Rice lab creates first heat-tolerant, stable fibers from wet-spinning process June 24th, 2022
|
|
||
|
|
||
| The latest news from around the world, FREE | ||
|
|
||
|
|
||
| Premium Products | ||
|
|
||
|
Only the news you want to read!
Learn More |
||
|
|
||
|
Full-service, expert consulting
Learn More |
||
|
|
||








