Home > Press > Cartilage damaged from exercise may aid in early osteoarthritis detection
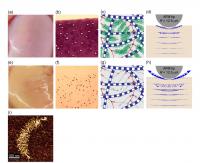 |
| These are images of (a) normal human joint cartilage, and (e) cartilage with very early matrix GAG loss. (b) Histologic image of a normal cartilage stained with Toluidine Blue to visualize content of GAGs. (f) Histologic image of a GAG-depleted cartilage (c) Schematic of extracellular matrix of normal cartilage composed mainly of collagen fibers and aggrecan (g) Schematic of matrix of GAG-depleted cartilage (d,h) Schematic of AFM-based dynamic compression of normal (d) and GAG-depleted (h) cartilage, which results in intra-tissue fluid flow velocity depicted by the blue arrows (from Finite element model computer simulations): loss of GAG (h) enables more fluid to flow out of the cartilage at high loading rates. (i) Human aggrecan imaged by Atomic Force Microscopy (AFM)
Credit: Biophysical Journal, Nia et al. |
Abstract:
Osteoarthritis is the most common joint disorder, affecting about one-third of older adults, and currently there is no cure. A study published by Cell Press April 2nd in the Biophysical Journal reveals how the nanoscale biomechanical properties of cartilage at joints change at the earliest stages of osteoarthritis, making the tissue more prone to damage during fast physical activities. The findings could improve early detection of the disease as well as tissue engineering strategies to repair damaged cartilage in patients.
Cartilage damaged from exercise may aid in early osteoarthritis detection
Cambridge, MA | Posted on April 2nd, 2013"Our techniques enable detection of the earliest loss of mechanical function associated with daily activities involving high loading rates, such as running and jumping," says senior study author Alan Grodzinsky of the Massachusetts Institute of Technology. "The findings can also be used to evaluate replacement tissue to ensure that it can survive these daily activities."
Osteoarthritis is a painful condition marked by the deterioration of cartilage�firm, rubbery tissue that cushions bones and prevents them from rubbing together. At the earliest stages of the disease, cartilage loses molecules called glycosaminoglycans (GAGs), which reduces the ability of the tissue to resist impact caused by physical activity. But until now, little was known about how GAG loss affects the functioning of cartilage across a wide spectrum of activities, from walking to running and jumping.
To address this question, Grodzinsky and his team developed a new system to measure the biomechanical properties of cartilage in response to cyclic compression forces that simulated a range of physical activities, each occurring at a different timescale. GAG-depleted cartilage was less capable of increasing its stiffness to deal with forces associated with high-rate activities such as running, when compared with normal tissue. Moreover, GAG loss resulted in a dramatic increase in the ability of fluids to flow out of cartilage, which is expected to diminish protection against impact caused by fast activities.
Together, the findings show how GAG depletion at the earliest disease stages could affect the nanoscale properties of cartilage, reducing the ability of this tissue to withstand high-rate activities. "We discovered that GAG-depleted tissue is most vulnerable to high rates of loading and not just the magnitude of the load," Grodzinsky says. "This finding suggests that people with early degradation of cartilage, even before such changes would be felt as pain, should be careful of dynamic activities such as running or jumping."
Biophysical Journal, Nia et al.: "High-Bandwidth AFM-Based Rheology Reveals that Cartilage is Most Sensitive to High Loading Rates at Early Stages of Impairment."
####
For more information, please click here
Contacts:
Mary Beth O'Leary
617-397-2802
Copyright © Cell Press
If you have a comment, please Contact us.Issuers of news releases, not 7th Wave, Inc. or Nanotechnology Now, are solely responsible for the accuracy of the content.
| Related News Press |
News and information
![]() Simulating magnetization in a Heisenberg quantum spin chain April 5th, 2024
Simulating magnetization in a Heisenberg quantum spin chain April 5th, 2024
![]() NRL charters Navy�s quantum inertial navigation path to reduce drift April 5th, 2024
NRL charters Navy�s quantum inertial navigation path to reduce drift April 5th, 2024
![]() Discovery points path to flash-like memory for storing qubits: Rice find could hasten development of nonvolatile quantum memory April 5th, 2024
Discovery points path to flash-like memory for storing qubits: Rice find could hasten development of nonvolatile quantum memory April 5th, 2024
Nanomedicine
![]() New micromaterial releases nanoparticles that selectively destroy cancer cells April 5th, 2024
New micromaterial releases nanoparticles that selectively destroy cancer cells April 5th, 2024
![]() Good as gold - improving infectious disease testing with gold nanoparticles April 5th, 2024
Good as gold - improving infectious disease testing with gold nanoparticles April 5th, 2024
![]() Researchers develop artificial building blocks of life March 8th, 2024
Researchers develop artificial building blocks of life March 8th, 2024
Discoveries
![]() Chemical reactions can scramble quantum information as well as black holes April 5th, 2024
Chemical reactions can scramble quantum information as well as black holes April 5th, 2024
![]() New micromaterial releases nanoparticles that selectively destroy cancer cells April 5th, 2024
New micromaterial releases nanoparticles that selectively destroy cancer cells April 5th, 2024
![]() Utilizing palladium for addressing contact issues of buried oxide thin film transistors April 5th, 2024
Utilizing palladium for addressing contact issues of buried oxide thin film transistors April 5th, 2024
Announcements
![]() NRL charters Navy�s quantum inertial navigation path to reduce drift April 5th, 2024
NRL charters Navy�s quantum inertial navigation path to reduce drift April 5th, 2024
![]() Discovery points path to flash-like memory for storing qubits: Rice find could hasten development of nonvolatile quantum memory April 5th, 2024
Discovery points path to flash-like memory for storing qubits: Rice find could hasten development of nonvolatile quantum memory April 5th, 2024
Interviews/Book Reviews/Essays/Reports/Podcasts/Journals/White papers/Posters
![]() Simulating magnetization in a Heisenberg quantum spin chain April 5th, 2024
Simulating magnetization in a Heisenberg quantum spin chain April 5th, 2024
![]() Discovery points path to flash-like memory for storing qubits: Rice find could hasten development of nonvolatile quantum memory April 5th, 2024
Discovery points path to flash-like memory for storing qubits: Rice find could hasten development of nonvolatile quantum memory April 5th, 2024
|
|
||
|
|
||
| The latest news from around the world, FREE | ||
|
|
||
|
|
||
| Premium Products | ||
|
|
||
|
Only the news you want to read!
Learn More |
||
|
|
||
|
Full-service, expert consulting
Learn More |
||
|
|
||








