Home > Press > Edible Gas Storage
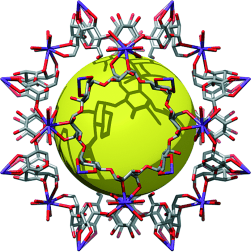 |
| Let them eat MOFs: Take a spoonful of sugar (-cyclodextrin to be precise), a pinch of salt (most alkali metal salts will suffice), and a swig of alcohol (Everclear fits the bill), and you have a robust, renewable, nanoporous (Langmuir surface area 1320 m2 g−1) metal–organic framework for breakfast (CD-MOF-1; see picture, C gray, O red, K purple; yellow sphere: pore). |
Abstract:
A spoonful of sugar, a pinch of salt, and a splash of alcohol - those are the ingredients used by scientists to generate a new class of robust nanoporous metal-organic frameworks. However, the sugar is not ordinary table sugar, but ă-cyclodextrin, produced from biorenewable cornstarch.
Edible Gas Storage
Posted on September 1st, 2010As Fraser Stoddart and a team of scientists from Northwestern University in Evanston (IL, USA), the University of California in Los Angeles (USA), and the University of St. Andrews (UK) report in the journal Angewandte Chemie, this simple recipe could be the basis for a new class of biocompatible porous crystals made of renewable natural products.
Metal-organic frameworks (MOF) are well-ordered, lattice-like crystals. The nodes of the lattices are complexes of transition metals (such as copper, zinc, nickel, or cobalt); organic molecules make up the connections between the nodes. Within their pores, the MOFs can store gases such as hydrogen or carbon dioxide. Furthermore, they can be used for separation of materials, for catalysis, or for the targeted transport of drugs in the body. Most previously prepared MOFs are made of building blocks that stem from petrochemicals. Stoddart and his team set themselves a challenge to synthesize MOFs from natural products. "The problem is that natural building blocks are generally not symmetrical," according to Stoddart, "this lack of symmetry seems to prevent them from crystallizing as highly ordered, porous frameworks."
ă-Cyclodextrin provided the solution to this problem: it comprises eight asymmetrical glucose residues arranged in ring, which is itself symmetrical. In many countries (for example the USA and Japan), cyclodextrins are approved for use as food additives. The second ingredient in the frameworks is an alkali metal salt. Suitable candidates include ordinary table salt (sodium chloride), the common salt substitute potassium chloride, or potassium benzoate, an approved preservative. These ingredients are dissolved in water and then crystallized by vapor diffusion with an alcohol. It is even possible to use commercially available sources such as grain alcohol. "These ingredients are all substances that can be obtained cheaply, in high quality, and of food-grade purity," says Stoddart.
The resulting crystals consist of cubes made from six ă-cyclodextrin molecules that are linked in three dimensions by potassium ions. These cubes are perfectly arranged to form a porous framework with easily accessible pores. "This arrangement is a previously unknown one," says Stoddart. "The pore volume encompasses 54% of the solid body." Particularly atypical of porous materials is the fact that when dissolved in water, the framework simply dissociates back to its components, which can then be crystallized again with alcohol. Says Stoddart: "In this way a degraded framework can easily be recycled or regenerated."
Author: J. Fraser Stoddart, Northwestern University, Evanston (USA), stoddart.northwestern.edu
Title: Metal-Organic Frameworks from Edible Natural Products
Angewandte Chemie International Edition, dx.doi.org/10.1002/anie.201002343
####
For more information, please click here
Contacts:
Editorial office
Copyright © Angewandte Chemie International Edition
If you have a comment, please Contact us.Issuers of news releases, not 7th Wave, Inc. or Nanotechnology Now, are solely responsible for the accuracy of the content.
| Related News Press |
News and information
![]() Quantum computer improves AI predictions April 17th, 2026
Quantum computer improves AI predictions April 17th, 2026
![]() Flexible sensor gains sensitivity under pressure April 17th, 2026
Flexible sensor gains sensitivity under pressure April 17th, 2026
![]() A reusable chip for particulate matter sensing April 17th, 2026
A reusable chip for particulate matter sensing April 17th, 2026
![]() Detecting vibrational quantum beating in the predissociation dynamics of SF6 using time-resolved photoelectron spectroscopy April 17th, 2026
Detecting vibrational quantum beating in the predissociation dynamics of SF6 using time-resolved photoelectron spectroscopy April 17th, 2026
Possible Futures
![]() A fundamentally new therapeutic approach to cystic fibrosis: Nanobody repairs cellular defect April 17th, 2026
A fundamentally new therapeutic approach to cystic fibrosis: Nanobody repairs cellular defect April 17th, 2026
![]() UC Irvine physicists discover method to reverse ‘quantum scrambling’ : The work addresses the problem of information loss in quantum computing system April 17th, 2026
UC Irvine physicists discover method to reverse ‘quantum scrambling’ : The work addresses the problem of information loss in quantum computing system April 17th, 2026
Academic/Education
![]() Rice University launches Rice Synthetic Biology Institute to improve lives January 12th, 2024
Rice University launches Rice Synthetic Biology Institute to improve lives January 12th, 2024
![]() Multi-institution, $4.6 million NSF grant to fund nanotechnology training September 9th, 2022
Multi-institution, $4.6 million NSF grant to fund nanotechnology training September 9th, 2022
Announcements
![]() A fundamentally new therapeutic approach to cystic fibrosis: Nanobody repairs cellular defect April 17th, 2026
A fundamentally new therapeutic approach to cystic fibrosis: Nanobody repairs cellular defect April 17th, 2026
![]() UC Irvine physicists discover method to reverse ‘quantum scrambling’ : The work addresses the problem of information loss in quantum computing system April 17th, 2026
UC Irvine physicists discover method to reverse ‘quantum scrambling’ : The work addresses the problem of information loss in quantum computing system April 17th, 2026
Research partnerships
![]() Lab to industry: InSe wafer-scale breakthrough for future electronics August 8th, 2025
Lab to industry: InSe wafer-scale breakthrough for future electronics August 8th, 2025
![]() HKU physicists uncover hidden order in the quantum world through deconfined quantum critical points April 25th, 2025
HKU physicists uncover hidden order in the quantum world through deconfined quantum critical points April 25th, 2025
|
|
||
|
|
||
| The latest news from around the world, FREE | ||
|
|
||
|
|
||
| Premium Products | ||
|
|
||
|
Only the news you want to read!
Learn More |
||
|
|
||
|
Full-service, expert consulting
Learn More |
||
|
|
||








