Home > Press > The Future in Two Words: Ionic Liquids
 |
Abstract:
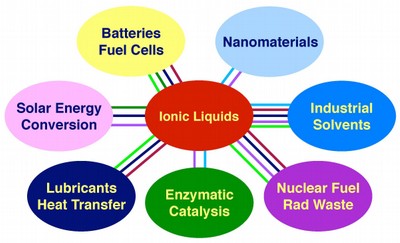
These molecular soups can be many things to many people in many different applications, including next-generation solar cells, hydrogen fuel cells, and lithium batteries. This may be what caught the eye of the U.S. Department of Energy (DOE), awarding Castner and his colleagues a grant for $2.4 million to delve into the nature of charge-transfer properties of ionic liquids. Previously, his Rutgers ionic liquids research was supported by the American Chemical Society's Petroleum Research Fund. In addition to the new DOE funding, the Rutgers ionic liquids fundamental research is also funded by the National Science Foundation.
The Future in Two Words: Ionic Liquids
New Brunswick, NJ | Posted on February 16th, 2010Ionic liquids by definition contain ions - atoms positively or negatively charged because they have too few or too many electrons or some other imbalance in their charge. Even with this imbalance, these ions are stable and exist freely in a solution, not bound to any other atoms as they would be in neutral (uncharged) compounds.
Successful with the DOE proposal, Castner is now the lead principal investigator on a three-year DOE-funded program. With his four co-principal investigators from Penn State, University of Iowa, University of Minnesota, and Brookhaven National Laboratory, Castner and his Rutgers colleagues have assembled a top research team for investigating the properties of ionic liquids.
Batteries are a key energy technology, but they can only charge and discharge their electrical energy relatively slowly - think how long it can take a cell phone or computer battery to recharge. When the sun rises or sets on the Rutgers Solar Farm on the Livingston Campus, or when a hybrid car like a Toyota Prius uses regenerative braking technology, high performance capacitors are required.
New supercapacitors and ultracapacitors based on ionic liquid technology will do an even better job than the current technologies. Castner hopes to merge their basic science projects for understanding ionic liquids to help the Rutgers Energy Storage Research Group develop next-generation ultracapacitors and batteries.
Hydrogen fuel cells, a potential successor to conventional batteries, work best at temperatures well above the boiling point of water; evaporative losses can damage the device performance. Because ionic liquids almost never boil and are stable to high temperatures, fuel cells based on ionic liquids are expected to display enhanced performance.
####
About Rutgers University
Rutgers, The State University of New Jersey, is a leading national public research university and the state�s preeminent, comprehensive public institution of higher education. Rutgers is dedicated to teaching that meets the highest standards of excellence; to conducting research that breaks new ground; and to turning knowledge into solutions for local, national, and global communities.
As it was at our founding in 1766, the heart of our mission is preparing students to become productive members of society and good citizens of the world. Rutgers teaches across the full educational spectrum: preschool to precollege; undergraduate to graduate and postdoctoral; and continuing education for professional and personal advancement. Rutgers is New Jersey�s land-grant institution and one of the nation�s foremost research universities, and as such, we educate, make discoveries, serve as an engine of economic growth, and generate ideas for improving people�s lives.
For more information, please click here
Contacts:
Office of Media Relations
Alexander Johnston Hall
101 Somerset St. New Brunswick, NJ 08901-1281
732-932-7084
Copyright © Rutgers University
If you have a comment, please Contact us.Issuers of news releases, not 7th Wave, Inc. or Nanotechnology Now, are solely responsible for the accuracy of the content.
| Related News Press |
News and information
![]() Simulating magnetization in a Heisenberg quantum spin chain April 5th, 2024
Simulating magnetization in a Heisenberg quantum spin chain April 5th, 2024
![]() NRL charters Navy�s quantum inertial navigation path to reduce drift April 5th, 2024
NRL charters Navy�s quantum inertial navigation path to reduce drift April 5th, 2024
![]() Discovery points path to flash-like memory for storing qubits: Rice find could hasten development of nonvolatile quantum memory April 5th, 2024
Discovery points path to flash-like memory for storing qubits: Rice find could hasten development of nonvolatile quantum memory April 5th, 2024
Possible Futures
![]() Discovery points path to flash-like memory for storing qubits: Rice find could hasten development of nonvolatile quantum memory April 5th, 2024
Discovery points path to flash-like memory for storing qubits: Rice find could hasten development of nonvolatile quantum memory April 5th, 2024
![]() With VECSELs towards the quantum internet Fraunhofer: IAF achieves record output power with VECSEL for quantum frequency converters April 5th, 2024
With VECSELs towards the quantum internet Fraunhofer: IAF achieves record output power with VECSEL for quantum frequency converters April 5th, 2024
Announcements
![]() NRL charters Navy�s quantum inertial navigation path to reduce drift April 5th, 2024
NRL charters Navy�s quantum inertial navigation path to reduce drift April 5th, 2024
![]() Discovery points path to flash-like memory for storing qubits: Rice find could hasten development of nonvolatile quantum memory April 5th, 2024
Discovery points path to flash-like memory for storing qubits: Rice find could hasten development of nonvolatile quantum memory April 5th, 2024
Battery Technology/Capacitors/Generators/Piezoelectrics/Thermoelectrics/Energy storage
![]() What heat can tell us about battery chemistry: using the Peltier effect to study lithium-ion cells March 8th, 2024
What heat can tell us about battery chemistry: using the Peltier effect to study lithium-ion cells March 8th, 2024
![]() A battery�s hopping ions remember where they�ve been: Seen in atomic detail, the seemingly smooth flow of ions through a battery�s electrolyte is surprisingly complicated February 16th, 2024
A battery�s hopping ions remember where they�ve been: Seen in atomic detail, the seemingly smooth flow of ions through a battery�s electrolyte is surprisingly complicated February 16th, 2024
Fuel Cells
![]() Current and Future Developments in Nanomaterials and Carbon Nanotubes: Applications of Nanomaterials in Energy Storage and Electronics October 28th, 2022
Current and Future Developments in Nanomaterials and Carbon Nanotubes: Applications of Nanomaterials in Energy Storage and Electronics October 28th, 2022
Grants/Sponsored Research/Awards/Scholarships/Gifts/Contests/Honors/Records
![]() Discovery points path to flash-like memory for storing qubits: Rice find could hasten development of nonvolatile quantum memory April 5th, 2024
Discovery points path to flash-like memory for storing qubits: Rice find could hasten development of nonvolatile quantum memory April 5th, 2024
![]() Chemical reactions can scramble quantum information as well as black holes April 5th, 2024
Chemical reactions can scramble quantum information as well as black holes April 5th, 2024
Alliances/Trade associations/Partnerships/Distributorships
![]() Chicago Quantum Exchange welcomes six new partners highlighting quantum technology solutions, from Chicago and beyond September 23rd, 2022
Chicago Quantum Exchange welcomes six new partners highlighting quantum technology solutions, from Chicago and beyond September 23rd, 2022
![]() University of Illinois Chicago joins Brookhaven Lab's Quantum Center June 10th, 2022
University of Illinois Chicago joins Brookhaven Lab's Quantum Center June 10th, 2022
Solar/Photovoltaic
![]() Development of zinc oxide nanopagoda array photoelectrode: photoelectrochemical water-splitting hydrogen production January 12th, 2024
Development of zinc oxide nanopagoda array photoelectrode: photoelectrochemical water-splitting hydrogen production January 12th, 2024
![]() Shedding light on unique conduction mechanisms in a new type of perovskite oxide November 17th, 2023
Shedding light on unique conduction mechanisms in a new type of perovskite oxide November 17th, 2023
![]() Inverted perovskite solar cell breaks 25% efficiency record: Researchers improve cell efficiency using a combination of molecules to address different November 17th, 2023
Inverted perovskite solar cell breaks 25% efficiency record: Researchers improve cell efficiency using a combination of molecules to address different November 17th, 2023
![]() Charged �molecular beasts� the basis for new compounds: Researchers at Leipzig University use �aggressive� fragments of molecular ions for chemical synthesis November 3rd, 2023
Charged �molecular beasts� the basis for new compounds: Researchers at Leipzig University use �aggressive� fragments of molecular ions for chemical synthesis November 3rd, 2023
|
|
||
|
|
||
| The latest news from around the world, FREE | ||
|
|
||
|
|
||
| Premium Products | ||
|
|
||
|
Only the news you want to read!
Learn More |
||
|
|
||
|
Full-service, expert consulting
Learn More |
||
|
|
||








