Home > Press > Silicon-coated Nanonets Could Build a Better Lithium-ion Battery
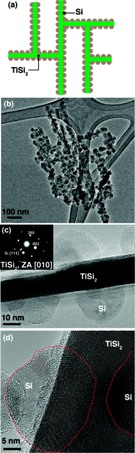 |
| Frame (a) shows a schematic of the Nanonet, a lattice structure of Titanium disilicide (TiSi2), coated with silicon (Si) particles to form the active component for Lithium-ion storage. (b) A microscopic view of the silicon coating on the Nanonets. (c) Shows the crystallinity of the Nanonet core and the Si coating. (d) The crystallinity of TiSi2 and Si (highlighted by the dotted red line) is shown in this lattice-resolved image from transmission electron microscopy. (Source: Nano Letters) |
Abstract:
Unique nanotech structure demonstrates higher speed, capacity and longevity
Silicon-coated Nanonets Could Build a Better Lithium-ion Battery
Chestnut Hill, MA | Posted on February 15th, 2010A tiny scaffold-like titanium structure of Nanonets coated with silicon particles could pave the way for faster, lighter and longer-lasting Lithium-ion batteries, according to a team of Boston College chemists who developed the new anode material using nanotechnology.
The web-like Nanonets developed in the lab of Boston College Assistant Professor of Chemistry Dunwei Wang offer a unique structural strength, more surface area and greater conductivity, which produced a charge/re-charge rate five to 10 times greater than typical Lithium-ion anode material, a common component in batteries for a range of consumer electronics, according to findings published in the current online edition of the American Chemical Society journal Nano Letters.
In addition, the Nanonets proved exceptionally durable, showing a negligible drop-off in capacity during charge and re-charge cycles. The researchers observed an average of 0.1% capacity fade per cycle between the 20th and the 100th cycles.
"As researchers pursue the next generation of re-chargeable Lithium-ion battery technology, a premium has been placed on increased power and a greater battery life span," said Wang. "In that context, the Nanonet device makes a giant leap toward those two goals and gives us a superior anode material."
Lithium-ion batteries are commonly used in consumer electronics devices. This type of rechargeable battery allows Lithium ions to move from the anode electrode to the cathode when in use. When charged, the ions move from cathode back to the anode.
The structure and conductivity of the Nanonets improved the ability to insert and extract Lithium ions from the particulate Silicon coating, the team reported. Running at a charge/discharge rate of 8,400 milliamps per gram (mA/g) - which is approximately five to 10 times greater than similar devices - the specific capacity of the material was greater than 1,000 milliamps-hour per gram (mA-h/g). Typically, laptop Lithium-ion batteries are rated anywhere between 4,000 and 12,000 mA/h, meaning it would only take between four and 12 grams of the Nanonet anode material to achieve similar capacity.
Wang said the capability to preserve the crystalline Titanium Silicon core during the charge/discharge process was the key to achieving the high performance of the Nanonet anode material. Additional research in his lab will examine the performance of the Nanonet as a cathode material.
View the Nano Letters paper at pubs.acs.org/doi/abs/10.1021/nl903345f
####
About Boston College
Boston College is one of the oldest Jesuit, Catholic universities in the United States. U.S. News and World Report ranks Boston College 34th among national universities.
Boston College confers more than 4,000 degrees annually in more than 50 fields of study through nine schools and colleges. Faculty members are committed to both teaching and research and have set new marks for research grant awards over the last ten years, more than $45 million in the last year alone.
For more information, please click here
Contacts:
Ed Hayward
Boston College Office of News & Public Affairs
Copyright © Boston College
If you have a comment, please Contact us.Issuers of news releases, not 7th Wave, Inc. or Nanotechnology Now, are solely responsible for the accuracy of the content.
| Related News Press |
News and information
![]() Simulating magnetization in a Heisenberg quantum spin chain April 5th, 2024
Simulating magnetization in a Heisenberg quantum spin chain April 5th, 2024
![]() NRL charters Navy�s quantum inertial navigation path to reduce drift April 5th, 2024
NRL charters Navy�s quantum inertial navigation path to reduce drift April 5th, 2024
![]() Discovery points path to flash-like memory for storing qubits: Rice find could hasten development of nonvolatile quantum memory April 5th, 2024
Discovery points path to flash-like memory for storing qubits: Rice find could hasten development of nonvolatile quantum memory April 5th, 2024
Possible Futures
![]() Discovery points path to flash-like memory for storing qubits: Rice find could hasten development of nonvolatile quantum memory April 5th, 2024
Discovery points path to flash-like memory for storing qubits: Rice find could hasten development of nonvolatile quantum memory April 5th, 2024
![]() With VECSELs towards the quantum internet Fraunhofer: IAF achieves record output power with VECSEL for quantum frequency converters April 5th, 2024
With VECSELs towards the quantum internet Fraunhofer: IAF achieves record output power with VECSEL for quantum frequency converters April 5th, 2024
Announcements
![]() NRL charters Navy�s quantum inertial navigation path to reduce drift April 5th, 2024
NRL charters Navy�s quantum inertial navigation path to reduce drift April 5th, 2024
![]() Discovery points path to flash-like memory for storing qubits: Rice find could hasten development of nonvolatile quantum memory April 5th, 2024
Discovery points path to flash-like memory for storing qubits: Rice find could hasten development of nonvolatile quantum memory April 5th, 2024
Battery Technology/Capacitors/Generators/Piezoelectrics/Thermoelectrics/Energy storage
![]() What heat can tell us about battery chemistry: using the Peltier effect to study lithium-ion cells March 8th, 2024
What heat can tell us about battery chemistry: using the Peltier effect to study lithium-ion cells March 8th, 2024
![]() A battery�s hopping ions remember where they�ve been: Seen in atomic detail, the seemingly smooth flow of ions through a battery�s electrolyte is surprisingly complicated February 16th, 2024
A battery�s hopping ions remember where they�ve been: Seen in atomic detail, the seemingly smooth flow of ions through a battery�s electrolyte is surprisingly complicated February 16th, 2024
|
|
||
|
|
||
| The latest news from around the world, FREE | ||
|
|
||
|
|
||
| Premium Products | ||
|
|
||
|
Only the news you want to read!
Learn More |
||
|
|
||
|
Full-service, expert consulting
Learn More |
||
|
|
||








