Home > Press > Fitter Frames: Nanotubes Boost Structural Integrity of Composites
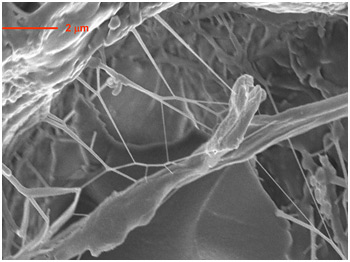 |
| Researchers at Rensselaer have discovered a new technique for provoking unusual crazing behavior in epoxy composites. The crazing, which causes the composite to deform into a network of nanoscale pillar-like fibers that bridge together both sides of a crack and slow its growth, could lead to tougher, more durable components for aircraft and automobiles. |
Abstract:
New research finding could lead to more durable aircraft, automotive components
Fitter Frames: Nanotubes Boost Structural Integrity of Composites
Troy, NY | Posted on March 27th, 2009A new research discovery at Rensselaer Polytechnic Institute could lead to tougher, more durable composite frames for aircraft, watercraft, and automobiles.
Epoxy composites are increasingly being incorporated into the design of new jets, planes, and other vehicles. Composite material frames are extremely lightweight, which lowers the overall weight of the vehicle and boosts fuel efficiency. The downside is that epoxy composites can be brittle, which is detrimental to its structural integrity.
Professor Nikhil Koratkar, of Rensselaer's Department of Mechanical, Aerospace, and Nuclear Engineering, has demonstrated that incorporating chemically treated carbon nanotubes into an epoxy composite can significantly improve the overall toughness, fatigue resistance, and durability of a composite frame.
When subjected to repetitive stress, a composite frame infused with treated nanotubes exhibited a five-fold reduction in crack growth rate as compared to a frame infused with untreated nanotubes, and a 20-fold reduction when compared to a composite frame made without nanotubes.
This newfound toughness and crack resistance is due to the treated nanotubes, which enhance the molecular mobility of the epoxy at the interface where the two materials touch. When stressed, this enhanced mobility enables the epoxy to craze - or result in the formation of a network of pillar-like fibers that bridge together both sides of the crack and slow its growth.
"This crazing behavior, and the bridging fibers it produces, dramatically slows the growth rate of a crack," Koratkar said. "In order for the crack to grow, those fibers have to first stretch, deform plastically, and then break. It takes a lot of energy to stretch and break those fibers, energy that would have otherwise gone toward enlarging the crack."
Results of the study were published this week in the journal Small.
Epoxy composites infused with carbon nanotubes are known to be more resistant to cracks than pure epoxy composites, as the nanotubes stitch, or bridge, the two sides of the crack together. Infusing an epoxy with carbon nanotubes that have been functionalized, or treated, with the chemical group amidoamine, however, results in a completely different bridging phenomenon.
At the interface of the functionalized nanotubes and the epoxy, the epoxy starts to craze, which is a highly unusual behavior for this particular type of composite, Koratkar said. The epoxy deforms, becomes more fluid, and creates connective fibers up to 10 microns in length and with a diameter between 100 nanometers and 1,000 nanometers.
"We didn't expect this at all. Crazing is common in certain types of thermoplastic polymers, but very unusual in the type of epoxy composite we used," Koratkar said. "In addition to improved fatigue resistance and toughness, the treated nanotubes also enhanced the stiffness, hardness, and strength of the epoxy composite, which is very important for structural applications."
Koratkar said the aircraft, boat, and automobile industries are increasingly looking to composites as a building material to make vehicle frames and components lighter. His research group plans to further investigate crazing behavior in epoxy composites, in order to better understand why the chemical treatment of nanotubes initiates crazing.
Co-authors of the paper include Rensselaer Associate Professor Catalin Picu, of the Department of Mechanical, Aerospace, and Nuclear Engineering; Rensselaer doctoral students Wei Zhang and Iti Srivastava; and Yue-Feng Zhu, professor in the Department of Mechanical Engineering at Tsinghua University in China.
Visit Koratkar's Web site http://www.eng.rpi.edu/mane/faculty_details.cfm?facultyID=koratn&type=research for more information on his nanomaterials research.
####
About Rensselaer Polytechnic Institute
RPI is widely recognized for the effectiveness of its hands-on approach to undergraduate education. Rensselaer�s classrooms are a model for intellectual engagement and the interaction of students and faculty.
In both curriculum reform and creative use of new teaching techniques and technologies, Rensselaer�s leadership in interactive learning has helped to further transform the way we teach and learn.
Contacts:
Jason B. Gorss
Manager of Media Relations
(518) 276-6098
Copyright © Rensselaer Polytechnic Institute
If you have a comment, please Contact us.Issuers of news releases, not 7th Wave, Inc. or Nanotechnology Now, are solely responsible for the accuracy of the content.
| Related News Press |
News and information
![]() Simulating magnetization in a Heisenberg quantum spin chain April 5th, 2024
Simulating magnetization in a Heisenberg quantum spin chain April 5th, 2024
![]() NRL charters Navy�s quantum inertial navigation path to reduce drift April 5th, 2024
NRL charters Navy�s quantum inertial navigation path to reduce drift April 5th, 2024
![]() Discovery points path to flash-like memory for storing qubits: Rice find could hasten development of nonvolatile quantum memory April 5th, 2024
Discovery points path to flash-like memory for storing qubits: Rice find could hasten development of nonvolatile quantum memory April 5th, 2024
Marine/Watercraft
![]() Strain-sensing smart skin ready to deploy: Nanotube-embedded coating detects threats from wear and tear in large structures July 15th, 2022
Strain-sensing smart skin ready to deploy: Nanotube-embedded coating detects threats from wear and tear in large structures July 15th, 2022
![]() A sunlight-driven �self-healing� anti-corrosion coating May 27th, 2022
A sunlight-driven �self-healing� anti-corrosion coating May 27th, 2022
![]() Quantum tech in space? Scientists design remote monitoring system for inaccessible quantum devices February 11th, 2022
Quantum tech in space? Scientists design remote monitoring system for inaccessible quantum devices February 11th, 2022
![]() Expanding the freedom of design: powder coating on FRP thanks to conductive gelcoats with graphene nanotubes March 3rd, 2021
Expanding the freedom of design: powder coating on FRP thanks to conductive gelcoats with graphene nanotubes March 3rd, 2021
Possible Futures
![]() Discovery points path to flash-like memory for storing qubits: Rice find could hasten development of nonvolatile quantum memory April 5th, 2024
Discovery points path to flash-like memory for storing qubits: Rice find could hasten development of nonvolatile quantum memory April 5th, 2024
![]() With VECSELs towards the quantum internet Fraunhofer: IAF achieves record output power with VECSEL for quantum frequency converters April 5th, 2024
With VECSELs towards the quantum internet Fraunhofer: IAF achieves record output power with VECSEL for quantum frequency converters April 5th, 2024
Nanotubes/Buckyballs/Fullerenes/Nanorods/Nanostrings
![]() Tests find no free-standing nanotubes released from tire tread wear September 8th, 2023
Tests find no free-standing nanotubes released from tire tread wear September 8th, 2023
![]() Detection of bacteria and viruses with fluorescent nanotubes July 21st, 2023
Detection of bacteria and viruses with fluorescent nanotubes July 21st, 2023
Announcements
![]() NRL charters Navy�s quantum inertial navigation path to reduce drift April 5th, 2024
NRL charters Navy�s quantum inertial navigation path to reduce drift April 5th, 2024
![]() Discovery points path to flash-like memory for storing qubits: Rice find could hasten development of nonvolatile quantum memory April 5th, 2024
Discovery points path to flash-like memory for storing qubits: Rice find could hasten development of nonvolatile quantum memory April 5th, 2024
Automotive/Transportation
![]() Researchers� approach may protect quantum computers from attacks March 8th, 2024
Researchers� approach may protect quantum computers from attacks March 8th, 2024
![]() Tests find no free-standing nanotubes released from tire tread wear September 8th, 2023
Tests find no free-standing nanotubes released from tire tread wear September 8th, 2023
Aerospace/Space
![]() Under pressure - space exploration in our time: Advancing space exploration through diverse collaborations and ethical policies February 16th, 2024
Under pressure - space exploration in our time: Advancing space exploration through diverse collaborations and ethical policies February 16th, 2024
![]() Bridging light and electrons January 12th, 2024
Bridging light and electrons January 12th, 2024
![]() Manufacturing advances bring material back in vogue January 20th, 2023
Manufacturing advances bring material back in vogue January 20th, 2023
|
|
||
|
|
||
| The latest news from around the world, FREE | ||
|
|
||
|
|
||
| Premium Products | ||
|
|
||
|
Only the news you want to read!
Learn More |
||
|
|
||
|
Full-service, expert consulting
Learn More |
||
|
|
||








