Home > Press > Proteomics Study Yields Clues As To How Tuberculosis Might Be Thwarting The Immune System
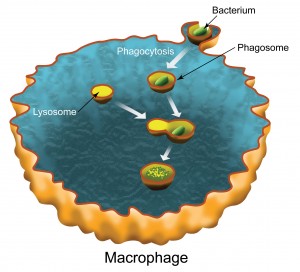 |
| Phagocytosis is the process by which a macrophage type white blood cell engulfs a bacterium in a membrane-bound shell called a phagosome. The phagosome fuses with a lysosome which carries digestive enzymes that destroy the bacterium. (Image by Flavio Robles, Berkeley Lab Public Affairs-CSO) |
Abstract:
A link between the immune system and the self-cleaning system by which biological cells rid themselves of obsolete or toxic parts may one day yield new weapons in the fight against tuberculosis and other deadly infectious diseases. Scientists with the U.S. Department of Energy's (DOE) Lawrence Berkeley National Laboratory (Berkeley Lab) have discovered proteins residing in both systems that point to "cross-talk" between them.
Proteomics Study Yields Clues As To How Tuberculosis Might Be Thwarting The Immune System
Berkeley, CA | Posted on November 5th, 2008In a collaboration between the research groups of Carolyn Bertozzi, director of Berkeley Lab's Molecular Foundry nanoscience center, and Jay Keasling, director of Berkeley Lab's Physical Biosciences Division, profiles were obtained for 546 different types of proteins in the membrane of a phagosome, an organelle of macrophages (a type of white blood cell) that essentially "eats" and destroys invading organisms (a process called phagocytosis). This represents the most comprehensive proteomic analysis of a phagosomal membrane to date.
"We were able to identify many new proteins that were not previously known to reside in the phagosome," said Wenqing Shui, a member of both the Bertozzi and Keasling research groups, and a proteomics specialist who was the lead author on a paper reporting these results in the Proceedings of the National Academy of Sciences (PNAS).
"One of the new proteins exclusively found in our study, LC3-II, is considered a marker of autophagy, the process that enables cells to clean up their own cytoplasm," Shui said. "Not only was LC3-II present in the phagosome, its level was increased upon the induction of autophagy in macrophages, and reduced when autophagy was suppressed. This indicates cross-talking between autophagy and phagocytosis that may play an important role in the response of the immune system."
The PNAS paper is entitled: "Membrane proteomics of phagosomes suggests a connection to autophagy." Co-authoring this paper in addition to Shui, Bertozzi and Keasling were Leslie Sheu, Jun Liuc, Brian Smart, Christopher Petzold, Tsung-yen Hsieh and Austin Pitcher. Bertozzi and Keasling are also professors at the University of California at Berkeley. In addition, Bertozzi is an investigator with the Howard Hughes Medical Institute, and Keasling is director of the Joint BioEnergy Institute.
When bacteria or other foreign particles invade the body, the first line of defense are the macrophages, which engulf and contain the invaders within the membrane-bound shells of their phagosomes. Once safely contained, the invaders can be killed with digestive enzymes from another cell organelle, called a lysosome, which fuses with the phagosome. Macrophages, like other kinds of cells, also use lysosomal enzymes for internal housekeeping. However, until now there has been no direct biochemical evidence of a link between phagocytosis and autophagy.
Working with latex bead-containing phagosomes isolated in cell lines from mice, Shui and her colleagues performed a detailed analysis of the protein contents of the phagosomal membrane. Unlike earlier proteomic studies, which profiled the entire organelle and focused on abundant water-soluble protein species, the study by the Bertozzi-Keasling groups was membrane-specific and included hydrophobic protein species that are present in the membrane in relatively low amounts.
We were able to demonstrate the endogenous level of LC3-II in macrophage phagosomes through the combination of sensitive proteomic techniques and biochemical assays," said Shui. "This is an excellent show-case of how a non-biased high-throughput proteomic study can shed new light on the diverse functions and pathways an organelle may engage in."
The LC3-II protein is a critical component of the autophagy machinery and the discovery that the level of its presence in phagosomes is modulated by autophagic activity (along with several other newly identified phagosome proteins not previously associated with autophagy), points to autophagy playing a heretofore unknown role in immune response, particularly against intracellular pathogens such as mycobacterium tuberculosis.
As Shui explained, "After mycobacterium tuberculosis are phagocytosed into the macrophage cell, they are able to subvert various host defense mechanisms, including the killing of bacilli in the phagosome, and survive well inside the cell. Given that induced autophagy activity appears to enhance mycobacterial killing in the phagosome (from a study by the University of New Mexico's Vojo Deretic and his research group), we speculate that mycobacteria may produce specific factors to counteract the bactericidal effect of autophagy activation."
The Bertozzi and Keasling research groups are now examining whether certain mycobacterial products can modulate macrophage autophagy activity. They are also looking for proteins that could specifically mediate the autophagy as well as the phagosome maturation process.
Said Shui, "We might be able to open new avenues for pharmacologic intervention of tuberculosis as well as other infectious diseases."
This research was supported by grants from the National Institutes of Health and the U.S. Department of Energy's Genomics:GTL program.
####
About Berkeley Lab
Berkeley Lab is a U.S. Department of Energy national laboratory located in Berkeley, California. It conducts unclassified scientific research and is managed by the University of California.
For more information, please click here
Contacts:
Lynn Yarris
(510) 486-5375
Copyright © Berkeley Lab
If you have a comment, please Contact us.Issuers of news releases, not 7th Wave, Inc. or Nanotechnology Now, are solely responsible for the accuracy of the content.
| Related News Press |
News and information
![]() Simulating magnetization in a Heisenberg quantum spin chain April 5th, 2024
Simulating magnetization in a Heisenberg quantum spin chain April 5th, 2024
![]() NRL charters Navy�s quantum inertial navigation path to reduce drift April 5th, 2024
NRL charters Navy�s quantum inertial navigation path to reduce drift April 5th, 2024
![]() Discovery points path to flash-like memory for storing qubits: Rice find could hasten development of nonvolatile quantum memory April 5th, 2024
Discovery points path to flash-like memory for storing qubits: Rice find could hasten development of nonvolatile quantum memory April 5th, 2024
Nanomedicine
![]() New micromaterial releases nanoparticles that selectively destroy cancer cells April 5th, 2024
New micromaterial releases nanoparticles that selectively destroy cancer cells April 5th, 2024
![]() Good as gold - improving infectious disease testing with gold nanoparticles April 5th, 2024
Good as gold - improving infectious disease testing with gold nanoparticles April 5th, 2024
![]() Researchers develop artificial building blocks of life March 8th, 2024
Researchers develop artificial building blocks of life March 8th, 2024
Discoveries
![]() Chemical reactions can scramble quantum information as well as black holes April 5th, 2024
Chemical reactions can scramble quantum information as well as black holes April 5th, 2024
![]() New micromaterial releases nanoparticles that selectively destroy cancer cells April 5th, 2024
New micromaterial releases nanoparticles that selectively destroy cancer cells April 5th, 2024
![]() Utilizing palladium for addressing contact issues of buried oxide thin film transistors April 5th, 2024
Utilizing palladium for addressing contact issues of buried oxide thin film transistors April 5th, 2024
Announcements
![]() NRL charters Navy�s quantum inertial navigation path to reduce drift April 5th, 2024
NRL charters Navy�s quantum inertial navigation path to reduce drift April 5th, 2024
![]() Discovery points path to flash-like memory for storing qubits: Rice find could hasten development of nonvolatile quantum memory April 5th, 2024
Discovery points path to flash-like memory for storing qubits: Rice find could hasten development of nonvolatile quantum memory April 5th, 2024
|
|
||
|
|
||
| The latest news from around the world, FREE | ||
|
|
||
|
|
||
| Premium Products | ||
|
|
||
|
Only the news you want to read!
Learn More |
||
|
|
||
|
Full-service, expert consulting
Learn More |
||
|
|
||








