Home > Press > Improved Ion Mobility Is Key to New Hydrogen Storage Compound
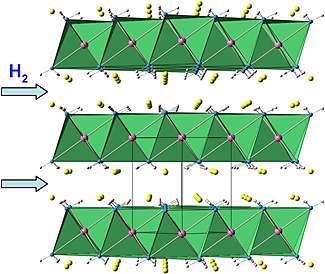 |
| The atomic structure of the mix of lithium amide with lightweight metal hydrides shows layers of calcium that the lithium ions can sprint through. This facilitates hydrogen storage and release.
Credit: NIST |
Abstract:
A materials scientist at the National Institute of Standards and Technology (NIST) has deciphered the structure of a new class of materials that can store relatively large quantities of hydrogen within its crystal structure for later release. The new analysis* may point to a practical hydrogen storage material for automobile fuel cells and similar applications.
Improved Ion Mobility Is Key to New Hydrogen Storage Compound
GAITHERSBURG, MD | Posted on May 13th, 2008The abundant element hydrogen could play a role in replacing carbon-based fuels for transportation in the future, but researchers first must develop a method to store and release large amounts of the highly flammable, odorless invisible gas economically and safely. There are materials that are known to trap relatively large quantities of hydrogen, at normal pressures, but to date they all require heating to fairly high temperatures to release the hydrogen.
Hui Wu, a research associate from the University of Maryland working in a cooperative research program at the NIST Center for Neutron Research, has been investigating a new hydrogen storage compound that mixes lithium amide with lightweight metal hydrides. Lithium amide can hold more than 10 percent of hydrogen by weight, well above the 6 percent target set by the U.S. Department of Energy as a 2010 goal for a hydrogen storage material for transportation. The material absorbs and releases hydrogen reversibly, but both absorbing and releasing the hydrogen requires high temperatures and also produces a toxic byproduct, ammonia.
Metal hydrides also store hydrogen, though not as well, but recently it's been shown that a combination of the two not only can store significant quantifies of hydrogen but also can release it at lower temperatures than the lithium amide alone (about 100 degrees Celsius) while generating much less ammonia.
To understand how the compound achieves this, Wu used neutron analysis to work out the atomic structure of the material, which she found consists of layers of calcium between which lithium ions travel rapidly. The easy travel allows the material to transfer the hydrogen at lower temperatures. Also the hydrogen ions in the amide and hydride mixture combine easily and release hydrogen at lower temperature without creating much ammonia.
"I found that the mobility of small ions in the mixed amide-hydride system greatly improves hydrogen storage properties," Wu explains. "This finding helps us understand how hydrogen travels in and out of these systems and that may lead to a rational development of better materials for hydrogen storage."
* H. Wu. Structure of ternary imide Li2Ca(NH)2 and hydrogen storage mechanisms in amide-hydride system. Journal of the American Chemical Society ASAP Article, Web release date: April 30, 2008
####
About NIST
From automated teller machines and atomic clocks to mammograms and semiconductors, innumerable products and services rely in some way on technology, measurement, and standards provided by the National Institute of Standards and Technology.
Founded in 1901, NIST is a non-regulatory federal agency within the U.S. Department of Commerce. NIST's mission is to promote U.S. innovation and industrial competitiveness by advancing measurement science, standards, and technology in ways that enhance economic security and improve our quality of life.
For more information, please click here
Contacts:
Evelyn Brown
(301) 975-5661
Copyright © NIST
If you have a comment, please Contact us.Issuers of news releases, not 7th Wave, Inc. or Nanotechnology Now, are solely responsible for the accuracy of the content.
| Related News Press |
News and information
![]() Quantum computer improves AI predictions April 17th, 2026
Quantum computer improves AI predictions April 17th, 2026
![]() Flexible sensor gains sensitivity under pressure April 17th, 2026
Flexible sensor gains sensitivity under pressure April 17th, 2026
![]() A reusable chip for particulate matter sensing April 17th, 2026
A reusable chip for particulate matter sensing April 17th, 2026
![]() Detecting vibrational quantum beating in the predissociation dynamics of SF6 using time-resolved photoelectron spectroscopy April 17th, 2026
Detecting vibrational quantum beating in the predissociation dynamics of SF6 using time-resolved photoelectron spectroscopy April 17th, 2026
Discoveries
![]() Quantum computer improves AI predictions April 17th, 2026
Quantum computer improves AI predictions April 17th, 2026
![]() Flexible sensor gains sensitivity under pressure April 17th, 2026
Flexible sensor gains sensitivity under pressure April 17th, 2026
![]() A reusable chip for particulate matter sensing April 17th, 2026
A reusable chip for particulate matter sensing April 17th, 2026
![]() Detecting vibrational quantum beating in the predissociation dynamics of SF6 using time-resolved photoelectron spectroscopy April 17th, 2026
Detecting vibrational quantum beating in the predissociation dynamics of SF6 using time-resolved photoelectron spectroscopy April 17th, 2026
Announcements
![]() A fundamentally new therapeutic approach to cystic fibrosis: Nanobody repairs cellular defect April 17th, 2026
A fundamentally new therapeutic approach to cystic fibrosis: Nanobody repairs cellular defect April 17th, 2026
![]() UC Irvine physicists discover method to reverse �quantum scrambling� : The work addresses the problem of information loss in quantum computing system April 17th, 2026
UC Irvine physicists discover method to reverse �quantum scrambling� : The work addresses the problem of information loss in quantum computing system April 17th, 2026
Energy
![]() Sensors innovations for smart lithium-based batteries: advancements, opportunities, and potential challenges August 8th, 2025
Sensors innovations for smart lithium-based batteries: advancements, opportunities, and potential challenges August 8th, 2025
![]() Simple algorithm paired with standard imaging tool could predict failure in lithium metal batteries August 8th, 2025
Simple algorithm paired with standard imaging tool could predict failure in lithium metal batteries August 8th, 2025
Fuel Cells
![]() Deciphering local microstrain-induced optimization of asymmetric Fe single atomic sites for efficient oxygen reduction August 8th, 2025
Deciphering local microstrain-induced optimization of asymmetric Fe single atomic sites for efficient oxygen reduction August 8th, 2025
![]() Current and Future Developments in Nanomaterials and Carbon Nanotubes: Applications of Nanomaterials in Energy Storage and Electronics October 28th, 2022
Current and Future Developments in Nanomaterials and Carbon Nanotubes: Applications of Nanomaterials in Energy Storage and Electronics October 28th, 2022
|
|
||
|
|
||
| The latest news from around the world, FREE | ||
|
|
||
|
|
||
| Premium Products | ||
|
|
||
|
Only the news you want to read!
Learn More |
||
|
|
||
|
Full-service, expert consulting
Learn More |
||
|
|
||








