|

Welcome to the 3rd edition of our Premium Newsletter.
In this issue NanoNews Now Editor Rocky Rawstern covers "nanomedicine." We will review what is being accomplished today and in the very near future, what we will likely see further down the road, and some of the ways it may change our lives. We will first talk about what nanomedicine is, and then where we are headed. And last, we draw a few conclusions.
For a comprehensive glossary of nanomedicine terms, click here.
In a departure from the format of our first two issues, we are very fortunate to have had the opportunity to collaborate with an acknowledged expert in the field - Robert A. Freitas Jr., author of Nanomedicine, Volume I: Basic Capabilities and Nanomedicine, Volume IIA: Biocompatibility. Having the "father of nanomedicine" involved in this article gives us a unique opportunity to get our minds around some of the myriad ways in which nanotech - when applied to medicine - can and will change our lives.
We left out most "way down the road" ideas, and elected instead to concentrate on things that are generally agreed to be plausible with today's knowledge. We will address "way down the road" in a future issue.
Go directly to the section:
Return to Top
Nanotechnology and Nanomedicine
Robert A. Freitas Jr., September 2003
Nanotechnology is engineering and manufacturing at the molecular scale, thereby taking advantage of the unique properties that exist at that scale. The application of nanotechnology to medicine is called nanomedicine. Nanomedicine subsumes three mutually overlapping and progressively more powerful molecular technologies. First, nanoscale-structured materials and devices that can be fabricated today hold great promise for advanced diagnostics and biosensors, targeted drug delivery and smart drugs, and immunoisolation therapies. Second, biotechnology offers the benefits of molecular medicine via genomics, proteomics, and artificial engineered microbes. Third, and further in the future, molecular machine systems and medical nanorobots will allow instant pathogen diagnosis and extermination, chromosome replacement and individual cell surgery in vivo, and the efficient augmentation and improvement of natural physiological function.
|
Some nanotech patent stats: there are currently 18,347 patents issued relating to nanotech, of these the biggest assignee is Genentech, Inc with 1,427 followed by IBM with 293 and Hitachi with 177. Genentech began its nano patenting activity in 93, IBM in 72 and Hitachi in 71 - all three have been accelerating their nano patenting activities to date. Clearly, going on the sheer number of patents the nanomedicine frontier is being tamed far faster than any other.
Randal Leeb-du Toit, August 2003 General Partner, Seraphima Ventures, Sydney, Australia
|
"There is a growing sense in the scientific and technical community that we are about to enter a golden new era," announced Richard E. Smalley, winner of the 1996 Nobel Prize in Chemistry, in Congressional testimony. On June 22, 1999, Smalley spoke in support of a new National Nanotechnology Initiative before the Subcommittee on Basic Research of the U.S. House Science Committee in Washington, DC. "We are about to be able to build things that work on the smallest possible length scales, atom by atom," Smalley said. "Over the past century we have learned about the workings of biological nanomachines to an incredible level of detail, and the benefits of this knowledge are beginning to be felt in medicine. In coming decades we will learn to modify and adapt this machinery to extend the quality and length of life." He became personally interested in the medical applications of nanotechnology in 1999, after he was diagnosed with a type of non-Hodgkin's lymphoma, for which he was (apparently) successfully treated.
"Twenty years ago," Smalley continued, "without even this crude chemotherapy I would already be dead. But twenty years from now, I am confident we will no longer have to use this blunt tool. By then, nanotechnology will have given us specially engineered drugs which are nanoscale cancer-seeking missiles, a molecular technology that specifically targets just the mutant cancer cells in the human body, and leaves everything else blissfully alone."
|
"Medical advances in nanotechnology are on the threshold of being able to locate and destroy cholesterol molecules on demand. If successful, not only will the need for the number of heart surgeons decrease, but businesses that rely on supplying a cure or treatment for heart disease will be replaced by a technology that prevents the problem from occurring in the first place. Similarly, if depression is found to be molecular in nature and nanotechnology, as believed, can assist in identifying the responsible molecules and help bind new drugs to those molecules, then a number of psychiatrists who treat patients for the disease, as well as Eli Lilly, the make of the antidepressant drug Prozac, might also find that nanotechnology is eroding their business."
"The impact of nanotechnology on drug discovery is broader than just the health-care industry. If the protein amyloid, which has been found to clog the brains of Alzheimer's patients, can be better understood, a treatment for this debilitating illness might be found. In addition to extending the productive life of thousands of people, the new treatment will also relieve untold amounts of pain and anguish suffered by the children and spouses of Alzheimer's victims. It might also significantly reduce the patient load of the 17,000 nursing homes across the country. This is especially true as America's 76 million baby-boomers begin to grow older. The $600 billion long-term-care industry and the burgeoning long-term health-care-insurance industry will need to begin factoring these changes into their strategic-planning processes today. Everything from the numbers of beds required to the monthly premium rate for long-term insurance will need to be revisited."
"Nanosphere reports that within the 2004 to 2005 time frame that it will have a detection system on the market that employs nanosensors to detect the presence of anthrax, smallpox, and a variety of illnesses ten times faster than anything on the market today and 100,000 times more accurately.
Among the companies working on nanosensors for disease detection are Nanosphere, Agilent, Motorola, SorroMed, Molecular Nanosystems, Cantion, and Scienion.
And while it is not known at this time which company or companies will win the battle in the marketplace, nanosensors are a disruptive technology that will impact a number of industries and professions.
If the price of this laboratory diagnostic equipment continues to drop as expected, it is entirely possible that doctors will have such equipment on their desktops within a few years. This suggests that the makers and suppliers of expensive laboratories and laboratory equipment will be adversely affected. If a simple, easy-to-use device can quickly run a test for not just one virus but for hundreds or even thousands, the need to send material to a lab will diminish. If this occurs, those academic institutions training medical lab technicians will need to revisit both how many people they should be training, and what they should be training them to do."
"Another promising area of nanomedicine involves the treatment of diabetes. Tejal Desai, a leading researcher at Boston University, has created a tiny silicon box that contains pancreatic cells taken from animals. The box, which is implanted in diabetics, is surrounded by a material with very specific-size nanopores. The pores are big enough to allow glucose molecules to enter but small enough to keep harmful antibodies out. When the pancreatic cells detect too much glucose, they release insulin - whose molecules are small enough to escape through the pores.
The impact on the treatment of diabetes - an ailment that, according to the NIH, afflicts 16 million Americans and is growing by 800,000 a year, results in 50,000 amputations per year and costs an average of $8,000 per patient a year - will be immense. Hospitals, government workers, insurance administrators, and businesses serving this huge population should all be following the developments of nanotechnology in this field."
"Another company, Angstrom Medica, is developing nanocrystals of various sizes, shapes, and purity that can mix with the body's own cells to help regrow bones. The treatment of osteoporosis, which costs the U.S. healthcare system $15 billion a year and affects 28 million Americans, will be among the first areas to benefit. On the other hand, physical therapists, who serve many of the 280,000 people who fracture their hips every year, might lose business and therefore might have to restructure their models of operation."
"Asthma is also benefiting from nanotech. A new nanoscale device, likened to Whiffle ball, can encapsulate an asthma drug and help it get to the lungs more quickly and more effectively. The result is that the businesses that cater to the more than 17 million Americans who suffer from asthma will need to reexamine the services and products they are offering."
Quantum Dot Corporation might, within the 2004 to 2005 time frame, completely disrupt the $2.8 billion fluorescent tagging market."
From "The Next Big Thing Is Really Small - How nanotechnology will change the future of your business"
The Next Big Thing Is Really Small: How Nanotechnology Will Change the Future of Your Business. J Uldrich & D Newberry. March 2003
Read our      review review
"Combining nanoPhotonics with Biotechnology will enable the proteinomics revolution, including individualized medicine, and the following:
Direct small molecule binding experiments
Structure-Activity optimization
"ligand fishing" - screening for interactions
Phenotyping (protein profiling)
Abundance profiling (ADME/Tox)
Protein activity profiling
Small molecule mediated protein:protein interaction
Orphan receptor characterization
Low affinity protein:protein binding
Many drugs which go to clinical trials and even FDA approval do not make it to market or are withdrawn due to varied responses (including deadly) by human genotypes and phenotypes. With rapid, low cost, desktop profiling capabilities, coupled with high speed, massive database query capability, medications and treatments will be individualized to avoid contra reactions and to optimize treatments."
Bo Varga, Executive Director, www.nanoSIG.org
"Like primitive engineers faced with advanced technology, medicine must "catch up" with the technology level of the human body before it can become really effective. What is the technology level? Since the human body is basically an extremely complex system of interacting molecules (i.e., a molecular machine), the technology required to truly understand and repair the body is molecular machine technology -- nanotechnology. A natural consequence of [our achieving] this level of technology will be the ability to analyze and repair the human body as completely and effectively as we can repair any conventional machine today."
Brian Wowk, "Cell Repair Technology" link
"Nanomedicine: Nanomedicine is here now. Drug delivery from inhalers to tablet formulation and polymer dissolution and phase change studies have been part of Nanomedicine for years. At Veeco we have seen strong growth in both atomic force microscope and interferometer sales for drug discovery, development and production QC over the last two years. Nanomedicine will continue to grow in these and many less traditional applications over the next 10 years. Veeco is planning on becoming a leading supplier of instruments to drug discovery and delivery companies.
More generally, we see the investment in nanotechnology and biotechnology by federal governments world-wide as a trend likely to yield significant business opportunities. However, with the restructuring of the venture community post dot-com I feel there are unfunnded business opportunities are more prevalent than ever. Agile companies that can take advantage of orphan nano-bio technologies will take on a leading role as instrument suppliers and technology developers."
Clark Taylor, VP Strategic Marketing Veeco Instruments
|
Return to Top
Where we are now
Medical Nanomaterials
Initial medical applications of nanotechnology, using nanostructured materials, are already being tested in a wide variety of potential diagnostic and therapeutic areas. A short list follows:
Tagged Nanoparticles
Fluorescent tags are commonplace in medicine and biology, found in everything from HIV tests to experiments that image the inner functions of cells. But different dye molecules must be used for each color, color-matched lasers are needed to get each dye to fluoresce, and dye colors tend to bleed together and fade quickly after one use. "Quantum dot" nanocrystals have none of these shortcomings. These dots are tiny particles measuring only a few nanometers across, about the same size as a protein molecule or a short sequence of DNA. They come in a nearly unlimited palette of sharply-defined colors, can be excited to fluorescence with white light, and can be linked to biomolecules to form long-lived sensitive probes to identify specific compounds. They can track biological events by simultaneously tagging each biological component (e.g., different proteins or DNA sequences) with nanodots of a specific color.
Quantum Dot Corporation (QDC) believes this kind of flexibility could offer a cheap and easy way to screen a blood sample for the presence of a number of different viruses at the same time. It could also give physicians a fast diagnostic tool to detect, say, the presence of a particular set of proteins that strongly indicates a person is having a heart attack. On the research front, the ability to simultaneously tag multiple biomolecules both on and inside cells could allow scientists to watch the complex cellular changes and events associated with disease, providing valuable clues for the development of future pharmaceuticals and therapeutics. In mid-2000, Genentech began evaluating the dots for commercial utility in a variety of cellular and molecular assays. A related technology called PEBBLES (Probes Encapsulated by Biologically Localized Embedding), pioneered by Raoul Kopelman at the University of Michigan, allows dye-tagged nanoparticles to be inserted into living cells to monitor metabolism or disease conditions.
In August 2003, QDC, Matsushita, and SC BioSciences agreed to develop, manufacture and market QDC's life science detection products. "Qdot nanotechnology is revolutionizing biological detection," said Takao Kanamura, Senior Managing Director of MKE. "We project products developed under this agreement to generate revenue for QDC in excess of $100 million per year by 2007." Said Carol Lou, president of QDC.
"QDC's Qdot nanocrystals are nanometer scale particles that are used in a broad range of biological detection applications in drug discovery, life science research, and medical diagnostics. The nanocrystals' extraordinary optical properties can be customized by changing their size or composition. Because of the special properties of the nanocrystals, detection can be up to a thousand times brighter than conventional dyes used in many biological tests. Furthermore, the ability to easily use many colors of Qdot nanocrystals dramatically increases the informativeness of these tests."
Quantum dots can also be used to take extremely detailed pictures of the inner walls of capillaries. Watt Webb, at Cornell University did just that, and says the real application is in imaging the brain and cancerous cells in living animals. By adding antibodies to the quantum dots, specific cells can be targeted. "The using of quantum dots, rather than conventional dyes, resulted in a thousand-fold increase in resolution, says Webb." So far the tests have been performed only on mice, who have shown no signs of toxicity.
Artificial Molecular Receptors
Another early goal of nanomedicine is to study how biological molecular receptors work, and then to build artificial binding sites on a made-to-order basis to achieve specific medical results. Buddy D. Ratner at the University of Washington in Seattle has researched the engineering of polymer surfaces containing arrays of artificial receptors. In a recent series of experiments, Ratner and his colleagues used a new radiofrequency-plasma glow-discharge process to imprint a polysaccharide-like film with nanometer-sized pits in the shape of such biologically useful protein molecules as albumin (the most common blood protein), fibrinogen (a clotting protein), lysozyme and ribonuclease (two important enzymes), and immunoglobulin (antibodies). Each protein type sticks only to a pit with the shape of that protein. Ratner's engineered surfaces may be used for quick biochemical separations and assays, as well in biosensors and chemosensors, because such surfaces will selectively absorb from solution only the specific protein whose complementary shape has been imprinted, and only at the specific place on the surface where the shape is imprinted. The RESIST Group at the Welsh School of Pharmacy at Cardiff University and others have looked at how molecularly imprinted polymers could be medically useful in clinical applications such as controlled drug release, drug monitoring devices, and biological and antibody receptor mimics.
Dendrimers
Dendrimers represent yet another nanostructured material that may soon find its way into medical therapeutics. Starburst dendrimers are tree-shaped synthetic molecules with a regular branching structure emanating outward from a core. Dendrimers form nanometer by nanometer, so the number of synthetic steps or "generations" dictates the exact size of the particles in a batch. Each molecule is typically a few nanometers wide though some have been constructed up to 30 nanometers wide, incorporating more than 100,000 atoms. The peripheral layer of the dendrimer particle can be made to form a dense field of molecular groups that serve as hooks for attaching other useful molecules, such as DNA, which hunker down amongst the outermost branches.
In 1998, James R. Baker Jr. co-founded the Center for Biologic Nanotechnology at the University of Michigan to bring together doctors, medical researchers, chemists and engineers to pursue the use of dendrimers as a safer and more effective genetic therapy agent. For Baker, these nanostructures are attractive because they can sneak DNA into cells while avoiding triggering an immune response, unlike viral vectors commonly employed today for transfection. The dendrimer molecule is decorated with specific snippets of DNA, then injected into biological tissue. Upon encountering a living cell, dendrimers of a certain size trigger a process called endocytosis in which the cell's outermost membrane deforms into a tiny bubble, or vesicle. The vesicle encloses the dendrimer which is then admitted into the cell's interior. Once inside, the DNA is released and migrates to the nucleus where it becomes part of the cell's genome. The technique has been tested on a variety of mammalian cell types, with clinical human trials of dendrimer gene therapy originally scheduled to begin in 2001. Donald Tomalia, another co-founder of the Center for Biologic Nanotechnology, reported using glycodendrimer "nanodecoys" to trap and deactivate influenza virus particles. The glycodendrimers present a surface that mimics the sialic acid groups normally found in the mammalian cell membrane, causing virus particles to adhere to the outer branches of the decoys instead of the natural cells.
In July 2003, Starpharma was cleared by the U.S. FDA for human trials of their dendrimer-based anti-HIV product. Their product has been successful in preventing simian-HIV.
Smart Drugs
Medical nanomaterials also may include "smart drugs" that become medically active only in specific circumstances. A good example is provided by Yoshihisa Suzuki at Kyoto University, who has designed a novel drug molecule that releases antibiotic only in the presence of an infection. Suzuki started with the common antibiotic molecule gentamicin and bound it to a hydrogel using a newly developed peptide linker. The linker can be cleaved by a proteinase enzyme manufactured by Pseudomonas aeruginosa, a Gram-negative bacillus that causes inflammation and urinary tract infection, folliculitis, and otitis externa in humans. Tests on rats show that when the hydrogel is applied to a wound site, the antibiotic is not released if no P. aeruginosa bacteria are present. But if any bacteria of this type are present, then the proteolytic enzyme that the microbes naturally produce cleaves the linker and the gentamicin is released, killing the bacteria. "If the proteinase specific to each bacterium [species] can be used for the signal," writes Suzuki, "different spectra of antibiotics could be released from the same dressing material, depending on the strain of bacterium." This specificity of action is highly desirable because the indiscriminate prophylactic use of antibiotics is associated with the emergence of strains of drug-resistant bacteria, and most antibiotics apparently have at least some toxicity for human fibroblasts.
Immunotoxins are another class of smart drugs, in this case activating only in the presence of cancer cells. An immunotoxin molecule is an engineered hybrid of functional protein modules fabricated from two different types of proteins: a toxin and an antibody. Toxin proteins are normally produced and released by infectious bacteria. The protein binds to the surface of a host cell, penetrates it, and kills it. Toxin molecules are so potent that just a few of them can kill a cell. Antibodies are proteins produced by the immune system to recognize and bind to specific foreign materials. An immunotoxin molecule is made by fusing a part of the gene encoding a toxin with a part of the gene encoding an antibody that recognizes surface features on cancer cells. This creates a novel gene that can be used to express a new synthetic protein molecule. This new molecule will bind only to a cancer cell (via a module from the antibody protein), then penetrate it and kill it (via modules from the toxin protein). The first experiments with mice showed that these engineered proteins successfully eliminated certain tumors. Then early in 2000, National Cancer Institute researchers confirmed that an immunotoxin made from a truncated form of Pseudomonas exotoxin was cytotoxic to malignant B-cells taken from patients with hairy cell leukemia. A second clinic trial at the Universitaet zu Koeln in Germany also found that a ricin-based immunotoxin had moderate efficacy against Hodgkin's lymphoma in some patients.
Nanospheres
Nanospheres that encapsulate and deliver drugs are another area in which we may soon see progress. In July 2003, two University of Texas at Austin biomedical engineers (Dr. Nicholas Peppas and Jay Blanchette) announced their latest findings at the Controlled Release Society's 30th Annual Meeting and Exhibition in Glasgow, Scotland. Their project: chemotherapy treatments delivered orally, rather than by injections or infusion. This method should allow for less-frequent dosages, as well as added convenience for the patient who will no longer have to suffer the multiple daily injections. They use hydrogels to overcome the problem of rapid breakdown in digestive system, which allows more of the chemotherapy drug to be delivered to the patient's system. "If you can replace the need to go to the hospital for the injections or infusion on a regular basis, with medication that can be taken in pill or capsule form at home, that's a vast improvement in quality of life for the patient," said Blanchette.
Nanoparticles and Nanoshells
In June 2003, American Pharmaceutical Partners announced success with their protein engineered nanoparticle paclitaxel. Their technology "integrates biocompatible proteins with drugs to create the nanoparticle form of the drug having a size of about 100-200 nanometers (approximately 1/100th the size of a single red blood cell)." Their novel approach avoids the sometimes toxic side-effects from solvents and emulsifiers used to administer insoluble drugs.
Along the same lines, researchers at The University of Nottingham are developing nanoparticle to treat brain tumors more effectively (by reducing the toxic side-effects). Due to the size of the nanoparticles, the drug is so small that it will only be delivered to the tumor and the liver, and not to other tissues within the body.
Magnetic nanoparticles are being developed that can be injected into the nuclei of living cells, where they can probe small areas of the genome.
Also in July of 2003, researchers at Rice University announced the development of a point-of-care whole blood immunoassay, using gold nanoshells, which yields results (in laboratory tests) in under 30 minutes. Currently, testing blood samples can take hours, delaying treatment. Using this new technology will allow - for instance - emergency room doctors to rapidly diagnose a variety of ailments, potentially saving lives. These same nanoshells (each slightly larger than a polio virus) are being used to detect and treat cancer cells, also at Rice University. Nanoshells offer advantages over traditional cancer treatments: earlier detection; more detailed imaging; fast, noninvasive imaging; and integrated detection and treatment. Among the companies that are currently developing nanomaterials as intravenous and topical drugs include Nanospectra Biosciences (a Rice spinoff), C Sixty, Advectus Life and American Pharmaceutical Partners. Additional cancer treatment research is being done at University of Buffalo, where they are developing a ceramic-based nanoparticle for photodynamic cancer therapy (PDT) that does not make the patient sensitive to light, as does traditional therapy.
Fullerene-based Pharmaceuticals
Soluble derivatives of fullerenes such as C60 - a soccer-ball-shaped arrangement of 60 carbon atoms per molecule - have shown great utility as pharmaceutical agents. These derivatives, many already in clinical trials, have good biocompatibility and low toxicity even at relatively high dosages. Fullerene compounds may serve as antiviral agents (most notably against HIV), antibacterial agents (E. coli, Streptococcus, Mycobacterium tuberculosis, etc.), antitumor and anticancer therapies, antioxidants and anti-apoptosis agents which may include treatments for amyotrophic lateral sclerosis (ALS or Lou Gehrig's disease) and Parkinson's disease, and other applications. The leading company in this area is C Sixty.
Building new organs and bones
Researchers at MIT and Harvard Medical School have built a functioning vascular system. By etching networks of paths (from three millimetres to 10 microns) onto silicon wafers, and using it as a mould for a layer of biodegradable polymer, they were able to produce mini artificial vascular system. "Eventually, we want to be able to replace whole organs with several layers of these constructs. So in the next 10-15 years, we will hopefully have reached a point where we can do this procedure clinically in human patients," said lead researcher Mohammad Kaazempur-Mofrad to NewScientist.com.
By decreasing the size of calcium phosphorus (two of the primary constituents of bone) to the nanoscale (30 nanometers in thickness and 60 nanometers in width), Cui Fuzhai (a materials science professor at Tsinghua University) has created "nano bone" - a replacement for damaged bone. "At this size, the properties of calcium phosphorus change. 'On a large scale (the calcium phosphorus) won't degrade, but on a nanoscale it will,' Cui said." What changes? Nanoscale calcium phosphorus degrades after a time (6 months), at which time the space has been filled with natural bone. A scales above the nano, calcium phosphorus does not degrade, so scale does matter. To-date Cui and his team has successfully implanted nano bones in dozens of patients, and received approval from China's FDA in November 2002. Hospitals are the first likely recipient of this new technology, and may start using it within months.
"Imagine having bones woven with a fabric such that one could fall out of a building and walk away. Imagine that in the event of a fire, microscopic vessels just ten billionths of a meter wide, pressurized with 1,000 atmospheres of pure oxygen could sense oxygen levels in the blood and provide hours of respiratory requirements for the body. Imagine medical nanites being injected into the bloodstream, consuming atherosclerotic plaques in the walls of the blood vessels; repairing cell damage caused by cancer. Or imagine nanomouthwashes that could eliminate gum disease and tooth decay - nanomachines acting as security guards and attacking any foreign entity in the body. Sounds like something from a science fiction movie? Absolutely not. Welcome to the world of nanomedicine." Albert Tsai link
The Nanopill
Credit researchers at McGill for creating the "nanopill"- a new way to deliver drugs inside cells.
Researchers at McGill University in Montreal recently created a "nanopill" from two polymer molecules - one water-repellant, the other hydrophobic - that self-assemble into a sphere called a micelle. In tests, the 20-45 nanometer structures were small enough to pass through the wall of an animal cell and deliver their cargo of drugs to specific structures within the cell.
Two of the companies currently working on a drug-delivery system based on this type of molecule are Insert Therapeutics Inc. in Pasadena, Calif., and Berlin-based Capsulution NanoScience AG.
Nanopore Immunoisolation Devices
Mauro Ferrari, director of the Biomedical Engineering Center at Ohio State University and chairman of the BioMEMS Consortium on Medical Therapeutics, and Tejal Desai have created what could be considered one of the earliest therapeutically useful nanomedical devices. Along with collaborators at the Biomedical Microdevices Center at the University of California at Berkeley, Ferrari and Desai employed bulk micromachining to fabricate tiny cell-containing chambers within single crystalline silicon wafers. The chambers interface with the surrounding biological environment through polycrystalline silicon filter membranes which are micromachined to present a high density of uniform nanopores as small as 20 nanometers in diameter. These pores are large enough to allow small molecules such as oxygen, glucose, and insulin to pass, but are small enough to impede the passage of much larger immune system molecules such as immunoglobulins and graft-borne virus particles. Safely ensconced behind this artificial barrier, immunoisolated encapsulated rat pancreatic cells may receive nutrients and remain healthy for weeks, happily secreting insulin back out through the pores, while the immune system remains blissfully unaware of the foreign cells which it would normally attack and reject.
Ferrari and Desai believe that microcapsules containing replacement islets of Langerhans cells - most likely easily-harvested piglet islet cells - could be implanted beneath the skin of some diabetes patients. This could temporarily restore the body's delicate glucose control feedback loop without the need for powerful immunosuppressants that can leave the patient at serious risk for infection. Supplying encapsulated new cells to the body could also be a valuable way to treat other enzyme or hormone deficiency diseases, including encapsulated neurons which could be implanted in the brain and then be electrically stimulated to release neurotransmitters, possibly as part of a future treatment for Alzheimer's or Parkinson's diseases. Tejal Desai has been active in continuing this work, and U.S. patents are in process.
Nanopore Sensors and DNA Sequencing
The flow of materials through nanopores can also be externally regulated. The first artificial voltage-gated molecular nanosieve was fabricated by Charles R. Martin and colleagues at Colorado State University in 1995. Martin's membrane contains an array of cylindrical gold nanotubules with inside diameters as small as 1.6 nanometers. When the tubules are positively charged, positive ions are excluded and only negative ions are transported through the membrane. When the membrane receives a negative voltage, only positive ions can pass. Future similar nanodevices may combine voltage gating with pore size, shape, and charge constraints to achieve precise control of ion transport with significant molecular specificity. In 1997, an exquisitely sensitive ion channel switch biosensor was built by an Australian research group. The scientists estimated that their sensor could detect a minute change in chemical concentration equivalent to a single sugar cube tossed into Sidney harbor, or roughly one part in a billion billion.
Daniel Branton at Harvard University has conducted an ongoing series of experiments using an electric field to drive a variety of RNA and DNA polymers through the central nanopore of an alpha-hemolysin protein channel mounted in a lipid bilayer similar to the outer membrane of a living cell. As early as 1996, the researchers had determined that the individual nucleotides comprising the polynucleotide strands must be passing single-file through the 2.6 nanometer-wide nanopore, and that changes in ionic current could be used to measure polymer length. By 1998, Branton had shown that the nanopore could be used to rapidly discriminate between pyrimidine and purine segments (the two types of nucleotide bases) along a single RNA molecule, and Branton continues to perfect this approach.. In 2000, the scientists demonstrated the ability to distinguish between DNA chains of similar length and composition that differ only in base pair sequence. A similar research effort at the University of California at Santa Cruz has produced nanopore devices with read rates potentially up to 1000 bases per second. Because nanopores can rapidly discriminate and characterize DNA polymers at low copy number, future refinements of this experimental approach may eventually provide a low-cost high-throughput method for very rapid genome sequencing.
Biological Robots
Biotechnology originally contemplated the application of biological systems and organisms to technical and industrial processes, but in recent times the field has expanded to include genetic engineering and the emerging fields of genomics, proteomics, transcriptomics, gene chips, artificial chromosomes, and even biobotics. Biotechnology now takes as its ultimate goal no less than the engineering of all biological systems, even completely designed organic living systems, using biological instrumentalities or "wet" nanotechnology. There are many good summaries of biotechnology elsewhere, so here we focus on efforts to engineer natural nanomachines to create new cellular devices.
During the 1990s, bioengineered viruses of various types and certain other vectors routinely were being used in experimental genetic therapies as "devices" to target and penetrate certain cell populations, with the objective of inserting therapeutic DNA sequences into the nuclei of human target cells in vivo. Retrovirally-altered lymphocytes (T cells) began to be injected into humans for therapeutic purposes. Another example was the use, by Neurotech (Paris), of genetically modified cerebral endothelial cell vectors to attack glioblastoma. This was the first therapeutic use of genetically engineered endothelial cells in humans; Phase I/II clinical studies were underway in 2000.
Engineered bacteria were also being pursued by Vion Pharmaceuticals in collaboration with Yale University. In their "Tumor Amplified Protein Expression Therapy" program, antibiotic-sensitive Salmonella typhimurium (food poisoning) bacteria were attenuated by removing the genes that produce purines vital to bacterial growth. The tamed strain could not survive very long in healthy tissue, but quickly multiplied 1000-fold inside tumors which are rich in purines. The engineered bacteria were available in multiple serotypes to avoid potential immune response in the host, and Phase I human clinical trials were underway in 2000 using clinical dosages. The next step would be to add genes to the bacterium to produce anticancer proteins that can shrink tumors, or to modify the bacteria to deliver various enzymes, genes, or prodrugs for tumor cell growth regulation.
In 1998, Glen Evans, then at the University of Texas Southwestern Medical Center, described the possible construction of synthetic genomes and artificial organisms. His proposed strategy involved determining or designing the DNA sequence for the genome, synthesizing and assembling the genome, then introducing the synthetic DNA into an enucleated pluripotent host cell to create an artificial organism. Genome engineers could modify an existing microbe by adding a biochemical pathway borrowed from other organisms, though this remains a difficult task because tailoring an existing system to match unique requirements demands detailed knowledge about the pathway. But ultimately, says Adam P. Arkin at Lawrence Berkeley National Laboratory, "we want to learn to program cells the same way we program computers." Some genome engineers have started by building the biological equivalent of the most basic switch in a computer - a digital flip-flop. "Cells switch genes on and off all the time," observes MIT's Thomas F. Knight, Jr., who has pioneered some of this research. A cellular toggle switch, made of DNA and some well-characterized regulatory proteins, might be devised to turn on a specific gene when exposed to a particular chemical. These could be used in gene therapies - implanted genes might be controlled with single doses of specially selected drugs, one to switch the gene on, another to switch it off.
Arcady Mushegian of Akkadix Corp. has looked at the genes present in the genomes of fully sequenced microbes to see which ones are always conserved in nature. He concludes that as few as 300 genes are all that may be required for life, constituting the minimum possible genome for a functional microbe. An organism containing this minimal gene set would be able to perform the dozen or so functions required for life - manufacturing cellular biomolecules, generating energy, repairing damage, transporting salts and other molecules, responding to environmental chemical cues, and replicating. The minimal microbe - a basic cellular chassis - could be specified by a genome only 150,000 nucleotides bases in length. By 2001Glen Evans could already produce made-to-order DNA strands that were 10,000 nucleotide bases in length and was striving to increase this length by at least a factor of ten. An engineered full-genome DNA, once synthesized, could be placed inside an empty cell membrane - most likely a living cell from which the nuclear material had been removed. Used in medicine, these artificial biorobots could be designed to produce useful vitamins, hormones, enzymes or cytokines in which a patient's body was deficient, or to selectively absorb and metabolize into harmless end products harmful substances such as poisons, toxins, or indigestible intracellular detritus, or even to perform useful mechanical tasks. One private company, engeneOS, was formed in 2000 to pursue the construction of these artificial biological devices, and in 2001 another company, Robiobotics LLC, put forward a business plan to pursue "whole genome engineering" and to begin seeking funding. But several other groups may be even further along in wetware engineering.
In November 2002, J. Craig Venter, of human genome-sequencing fame, and Hamilton O. Smith, a Nobel laureate, announced their new company, Institute for Biological Energy Alternatives (IBEA), had received a $3 million, three-year grant from the Energy Department to create a minimalist organism, starting with the M. genitalium microorganism. Working with a research staff of 25 people, the scientists will remove all genetic material from the organism, then synthesize an artificial string of genetic material resembling a naturally occurring chromosome that they hope will contain the minimum number of M. genitalium genes needed to sustain life. The artificial chromosome will be inserted into the hollowed-out cell, which will then be tested for its ability to survive and reproduce. To ensure safety, Smith and Venter said the cell will be deliberately hobbled to render it incapable of infecting people; it also will be strictly confined, and designed to die if it does manage to escape into the environment.
In early 2003, Glen Evans' new company Egea Biosciences may have vaulted into the lead, having been granted "the first [patent] to include broad claims for the chemical synthesis of entire genes and networks of genes comprising a genome, the 'operating system' of living organisms." According to the company, Egea's proprietary GeneWriterô and Protein Programmingô technology has been proven in extensive proof of concept studies and has: (1) produced libraries of more than 1,000,000 programmed proteins, (2) produced over 200 synthetic genes and proteins, (3) produced the largest gene ever chemically synthesized of over 16,000 bases, (4) engineered proteins for novel functions, (5) improved protein expression through codon optimization, and (6) developed custom genes for protein manufacturing in specific host cells. Egea's software allows researchers to author new DNA sequences that the company's hardware can then manufacture to specification with a base-placement error of only ~10-4, which Evans calls "word processing for DNA". According to Egea's patent, one "preferred embodiment of the invention" would include the synthesis of "a gene of 100,000 bp ... from one thousand 100-mers. The overlap between 'pairs' of plus and minus oligonucleotides is 75 bases, leaving a 25 base pair overhang. In this method, a combinatorial approach is used where corresponding pairs of partially complementary oligonucleotides are hybridized in the first step. A second round of hybridization then is undertaken with appropriately complementary pairs of products from the first round. This process is repeated a total of 10 times, each round of hybridization reducing the number of products by half. Ligation of the products then is performed." The result would be a strand of DNA 100,000 base pairs in length, long enough to make a very simple bacterial genome. Evans says his prototype machine can synthesize 10,000 bases in two days, and that 100,000 bp strands might require "a matter of weeks" to synthesize using a future next-generation machine. "Pretty soon, we won't have to store DNA in large refrigerators," says Tom Knight. "We'll just write it when we need it."
Besides their direct medical applications, biobots might be employed in molecular construction. Gerald J. Sussman at MIT notes that when computer parts are reduced to the size of single molecules, engineered microbes could be directed to lay down complex electronic circuits. "Bacteria are like little workhorses for nanotechnology; they're wonderful at manipulating things in the chemical and ultramicroscopic worlds," he says. "You could train them to become electricians and plumbers, hire them with sugar and harness them to build structures for you."
|
An engineered full-genome DNA, once synthesized, could be placed inside an empty cell membrane - most likely a living cell from which the nuclear material had been removed. Used in medicine, these artificial biorobots could be designed to produce useful vitamins, hormones, enzymes or cytokines in which a patient's body was deficient, or to selectively absorb and metabolize into harmless end products harmful substances such as poisons, toxins, or indigestible intracellular detritus, or even to perform useful mechanical tasks. One private company, engeneOS, was formed in 2000 to pursue the construction of these artificial biological devices, and in 2001 another company, Robiobotics LLC, put forward a business plan to pursue "whole genome engineering" and to begin seeking funding. But several other groups may be even further along in wetware engineering.
Robert A. Freitas Jr.
Responding to the question "how is nanotechnology likely to effect medicine?" Paul Holister had this to say:
In the third world, vaccines previously requiring injection will be delivered orally (using nanoencapsulation), increasing enormously the number of people that can benefit.
Pharmaceutical companies should be able to recoup some of their money on drugs that have proven effected but failed approval because of high toxicity. Targeted drug delivery (using nanocapsules and nanoparticles) will be the technology behind this.
Nanostructured electronics and sensors should start to offer effective prosthetics for the blind and deaf. Various nanomaterials will contribute here by improving biocompatibility.
A large amount of diagnosis will move out of the lab and into the doctor's office. This may start to include tailoring of treatment to a person's genetic make-up.
Drug discovery and screening will become much more rapid and cost-effective. Requirements for PCR in this process will diminish considerably.
Rapid complete genome sequencing (within a day) will probably become feasible. This will accelerate understanding of the activity of genes and gene groups through the use of comparitive techniques on a large number of full genomes.
Paul Holister, Chief Architect of the Nanotechnology Opportunity ReportTM, and editor of TNT weekly. CIO of CMP Cientifica and Founder and Research Director of the ENA.
Responding to the question "how is nanotechnology likely to effect medicine?" Wil McCarthy had this to say:
The only medicines that ever seem to "fix" anything are molecular medicines like antibiotics and cancer drugs, and macroscopic, mechanical therapies like bone setting and surgery. But surgery is incredibly crude: cutting big holes through a person to get at something inside -- usually something really small. There's a lot of room to do better, with smaller holes and more versatile instruments, and with nanosurgery as the end result. Also, working up from the molecular level, we may start to see polyfunctional, shape-changing molecules which can serve multiple duties inside the body. Why cut at all, when you can send in the molecular mechanics and fix the problem at its root?
Wil McCarthy http://www.wilmccarthy.com/
Engineer, Columnist, Author of:
Hacking Matter: Levitating Chairs, Quantum Mirages, and the Infinite Weirdness of Programmable Atoms, and The Wellstone, and The Collapsium
Responding to the question "how is nanotechnology likely to effect medicine?" Mark Ratner and Daniel Ratner had this to say:
Medicine is one of the first fields to feel the transforming effect of nanotechnology, and by decade's end almost every part of the field will be affected. For one early example of how nanotechnology is having an impact in the medical industry, consider the problem of bioavailability.
Bioavailability is a drug's ability to reach the site where it will be effective so that it can treat a given condition - drugs that have problems with bioavailability can be washed right through the body without having their full effect. It is estimated that waste due to bioavailability problems costs consumers some $45 billion per year, but research in nanotechnology has shown that just reducing drug particle sizes to the nanoscale and thus increasing surface area and reactivity can reduce this problem hugely. One of the nicest things about this idea is that it doesn't require any changes for patients or practitioners - a massive savings can be made just by changing the way that certain drugs are manufactured.
Nanotechnology means more than cost savings for medicine. Consider one of the very oldest problems - diagnosis. When you go into a doctor's office, you are routinely tested for a variety of conditions, but blood tests take days and typically only screen for a few dozen problems. More extensive examination, blood tests, stool samples, throat cultures, and biopsies may be required to determine the presence of many diseases and even then they may not be conclusive. A combination of nanotechnology-based sensors and lab-on-a-chip technology will change this completely. Possibly by the end of the decade, it will be possible to go to a doctor's office, have a single sample taken, and, while you read a magazine in the waiting room, instantly and accurately screen you for thousands of diseases and even determine your genetic predisposition to conditions such as some forms of cancer and heart disease. Much of the fundamental technology to do this exists and just needs to be refined.
Another place where nanotechnology will shine is in human repair, the field of helping the body to repair itself. Broken bones are perhaps the best example of this. When a bone is broken now, it may take several months to heal, it may not set properly, and compound fractures may require steel plates, bone nails, bolts or other structural components which may remain inside a patient for the rest of his or her life. Human repair work indicates that it may be possible to do this much more efficiently. Nanotechnology-enabled smart materials can be injected into the site of an injury, self-assemble into fibers which bridge the fracture, and allow new bone to form around them. This could take a fraction of the time it takes for bones to heal now. For those with injuries that cannot heal naturally such as the loss of a limb, nanotechnology-based bionics and prosthetics will actually allow much more movement than is possible with current products. Vision systems to restore sight to the blind and vastly improved hearing aids are also on the horizon.
While all of these changes will be huge, perhaps the most exciting developments will be in nanotechnology-based drugs and therapies. Time release drugs and drugs involving a biofeedback mechanism (such as keeping the insulin level in the blood constant for diabetics) will be much more practical and reliable using nanotechnology. Even more radically, sensor technology will be combined with drugs to create smart drugs which home in on their targets (for example tumors) and destroy them without damaging anything else. This targeting will reduce or eliminate the toxic trail and unpleasant side effects of chemotherapy and radiotherapy as well as many other treatments. There is even reason to believe that more general antiviral drugs (analogous to antibiotics for bacteria) may be possible relatively soon through nanotechnology.
All of the developments we've discussed are likely to be seen in some form in the next decade. They will require some retraining for doctors and health care practitioners, but should require relatively little expensive equipment or knowledge from patients. In the future even more will be possible, and it seems likely that nanotechnology will give medicine a renaissance at least as significant as Paracelsus' reformation of medieval medicine or Pasteur's invention of antibiotics.
Mark Ratner and Daniel Ratner, Authors of:
Nanotechnology: A Gentle Introduction to the Next Big Idea, and the forthcoming "Nanotechnology and Homeland Security." (October 2003) Our recommendation "U.S. policy-makers and -shapers: READ THIS BOOK! Then get to work."
A nanotech-built surgical robot with a full sensor suite could be smaller than a hypodermic needle.
Molecular Manufacturing: Start Planning
Chris Phoenix, Director of Research, the Center for Responsible Nanotechnology
NanoNews Now (NN) Editor Rocky Rawstern interviews Neil Gordon (NG) about nanomedicine.
NN: How do you envision nanotech enabling advances in medicine?
NG: There are signs that we are in the midst of an explosion of new health-related technologies. Genomics, proteomics, stem cells, structured-based drug design, photodynamic therapy, combinatory chemistry, and intercellular signaling, provide tremendous insight and new directions for medical research. When nanotechnology and nanoscience are used along with these technologies, the likelihood of significant medical advances and breakthroughs is unprecedented. Some of the ways that nanotech will enable advances in medicine are as follows:
Nanotechnology Tools will allow researchers to visualize, measure and manipulate items at the nanoscale such as cells, bacteria and viruses, and to detect single molecules. Among other things, this will provide new information about biological processes for understanding cellular function including real time measurements of single molecules in living cells and the development of the collateral chemistry and instrumentation.
Nanomaterials will solve unique biological and medical challenges with synthetic and hybrid nanostructures that can be functionalized to carry out highly focused tasks. Examples include sensing and repairing biological lesions and damages just as biological nanostructures (e.g. white-blood cells and wound-healing molecules); detecting and treating diseases after measuring various parameters such as electrical changes from biological molecules; drug delivery systems that can pass through natural defense mechanisms including the blood-brain barrier; and longer lasting and better performing materials for bone replacements, prostheses, and implants. As life contains a collection of molecular materials and biological structures in the nanoscale, nanostructured materials are ideally sized to complement nature. Consider the size of selected materials from nature and nanotechnology typically referenced to have the following dimensions:
Nature
DNA (width) 2 nm
Protein 5 - 50 nm
Virus 75 - 100 nm
Materials internalized by cells < 100 nm
Bacteria 1,000 - 10,000 nm
White Blood Cell 10,000 nm
Nanotechnology
Nanoparticles 1 - 100 nm
Fullerene (C60) 1 nm
Nanotube (Single wall) 1 nm x 50,000 nm
Quantum Dot (CdSe) 8 nm
Dendrimer 10 nm
Nanotube (Multi wall) 20 nm x 50,000 nm
Nanotechnology Devices - along with informatics tools will enable minimally invasive detection, diagnosis, and management of disease and injury. Examples include biosensors and detectors to detect trace quantities of bacteria, airborne pathogens, biological hazards, and disease signatures; "Lab-on-a-chip" applications for DNA testing and implantable fluid injection systems; and miniature moving parts for heart pacemakers and surgical devices. While nano devices are increasingly becoming possible to make in the lab, larger scale micro devices are effective solutions in most cases and avoid many difficulties associated with production and packaging. However, nano devices can bring significant improvements in the way microscale devices work. This includes higher throughput DNA sequencers with a higher density of spots, reducing the area and time needed to be sampled for drug discovery and diagnostics; and smaller fluidic systems that more efficiently conduct chemical experiments since fluids move through nanoscale pipes with laminar flow, which avoids turbulence and mixing.
Intelligent Materials and Machines is a fascinating yet controversial area of nanotechnology which comprises many forms of research, much of it blue-sky. Far reaching concepts such as tiny robots, nanorobots or nanobots, are being conceptualized that may be injected into the body to attack infections or undesirable cells. Other potential applications include intelligent materials that can sense external stimuli and alter their properties to adapt to changes in the environment, molecular machines which can construct materials atom by atom, and molecular assemblers that can mass produce molecular machines. Virtually no projects in this area are near commercialization that I am aware of.
NN: What changes are we likely to see in the coming decade regarding advances in nanomedicine?
NG: In preparation for the Canadian Institutes of Health Research Nanomedicine Workshop last year, I was asked to define the major areas of advances in nanomedicine. Some of the major areas are summarized below and are more thoroughly described in the Nanomedicine Taxonomy (PDF) that I co-authored with Uri Sagman, the co-founder of C Sixty.
Biopharmaceutics
Drug Delivery - a wide range of new technologies for developing customized solutions that optimize the delivery of pharmaceutical products.
Drug Discovery - advanced solutions and new paradigms for decreasing the discovery and development times for new drugs, and potentially reducing the development costs.
Implantable Materials
Tissue Repair and Replacement - a new generation of biocompatible nanomaterials for repairing and replacing human tissues.
Structural Implant Materials - a new generation of biocompatible materials that can be used as implants or temporary biosorbable structures.
Implantable Devices
Assessment and Treatment Devices - sensing technologies that provide more accurate and timely medical information for diagnosing disease, and miniature devices that can administer treatment automatically if required.
Sensory Aids - a new generation of smaller and potentially more powerful devices to restore lost vision and hearing functions.
Surgical Aids
Operating Tools - medical devices that allow surgeons to perform familiar tasks with greater precision and safety, monitor physiological and biomechanical parameters more accurately, and perform new tasks that are not currently done.
Diagnostic Tools
Genetic Testing - new solutions for increasing the speed and accuracy of identifying genes and genetic materials for drug discovery and development, and for treatment-linked disease diagnostics products.
Imaging - new imaging technologies that provide high quality images not possible with current devices, along with new methods of treatment.
NN: What should health-care-industry businesses be doing now to prepare for the potentially disruptive technologies?
NG: Short term, education and intelligence are key. With so many new technologies and research projects taking place, it is becoming increasingly difficult to keep track of finding and leveraging early-stage innovation that is really strategic for leading companies in the health care industry. While trade publications, market reports and trade shows are useful, networking and collaborative initiatives can bring a higher level of quality information.
Longer term, production and distribution paradigms may have to be rethought. As we expect more varieties of medical products and pharmaceutics, it is not clear where the production capacity will come from. For example, as more types of drug delivery systems are possible, there will be many more types of specialty products hitting the market than ever before. With shorter production runs and unique materials such as fullerenes, dendrimers and quantum dots, production may not be feasible with older and/or large-scale production plants and contract manufacturers.
Limited shelf space in today's pharmacies for drugs, diagnostic tests and other medical devices may lead to speciality outlets and a new series of regulations for selling to consumers, doctors and hospitals who need considerable guidance in understanding the consequences of using these new products.
Neil Gordon P.Eng, MBA
President of the Canadian NanoBusiness Alliance & Partner-Nanotechnology with Sygertech
"A revolution has begun in science, engineering and technology, based on the ability to characterize, manipulate and organize matter systematically at the nanometer scale. Nanotechnology is beginning to provide the ability to work at this level -- to control nanoscale structures, using them as building blocks to construct larger material components, systems and architectures to form supramolecular structures with fundamentally new molecular organization. Within these larger scale assemblies, the control and construction of substructures and components remains at the nanometer scale. Nanotechnology offers opportunities for the creation and utilization of materials, devices and systems by building from the level of atoms and molecules. The key resultant opportunity is the exploitation of novel and improved properties that emerge at the nanoscale. Nanotechnology is expected to play an important role in scientific disciplines such as physics, material science, biology, medicine, engineering and computer simulation."
National Institutes of Health (NIH)
"Nanoscience and Nanotechnology in Biology and Medicine" Link
Responding to the question "how is nanotechnology likely to effect medicine?" William Atkinson had this to say:
From The Hulk's fearsome 'nano-meds' to the programmable molecular nanocatalysts in Agent Cody Banks, there's a Hollywood view that nanotechnology will enable near-magical advances in medicine in the next few years. Having surveyed the field for a book-length report, I can confidently say that such forecasts are inaccurate because they're much too timid. Real nanomedicine will vastly exceed such weak-kneed forecasts. Grounded in solid nanoscience, it will invoke the incredible power of the STM, AFM, and other nanoscopic instrumentation in imaging and manipulating individual atoms and molecules to the benefit of us all. Aging, an artifact of telomeric degradation, will be arrested and reversed. Laparoscopy, today's surgery through tiny incisions, will give way to nanoscopy - minimally invasive surgical intervention through sweat pores. I fully expect to read this prediction aloud to a clamouring audience in AD 2023. As medical consumers, we ain't seen nothing yet.
William Illsey Atkinson
Author, Nanocosm (AMACOM 2003)
|
Return to Top
Where we are headed
Medical Nanorobotics
The third major branch of nanomedicine - molecular nanotechnology (MNT) or nanorobotics - takes as its purview the engineering of all complex mechanical medical systems constructed from the molecular level. Just as biotechnology extends the range and efficacy of treatment options available from nanomaterials, the advent of molecular nanotechnology will again expand enormously the effectiveness, comfort and speed of future medical treatments while at the same time significantly reducing their risk, cost, and invasiveness. MNT will allow doctors to perform direct in vivo surgery on individual human cells.
DNA-Based Nanodevices
Early mechanical nanorobots might be made, at least in part, of DNA. The idea of using DNA to build nanoscale objects has been pioneered by Nadrian Seeman at New York University. Two decades ago, Seeman recognized that a strand of DNA has many advantages as a construction material. First, it is a relatively stiff polymer. Its intermolecular interaction with other strands can be readily predicted and programmed due to the base-pair complementarity of nucleotides, the fundamental building blocks of genetic material. DNA also tends to self-assemble. Arbitrary sequences are readily manufactured using conventional biotechnological techniques, and DNA is readily manipulated and modified by a large number of enzymes. During the 1980s, Seeman worked to develop strands of DNA that would zip themselves up into more and more complex shapes - first tiny squares, then three-dimensional stick-figure cubes comprised of 480 nucleotides each, then a truncated octahedron containing 2550 nucleotides. By the mid-1990s, Seeman could fabricate nanoscale DNA stick figures of almost any regular geometric shape, by the billions per batch.
In 1999, Seeman reported yet another breakthrough - the construction of a mechanical DNA-based device that might serve as the basis for a nanoscale robotic actuator. The mechanism has two rigid double-stranded DNA arms a few nanometers long that can be made to rotate between fixed positions by introducing a positively charged cobalt compound into the solution surrounding the molecules, causing the bridge region to be converted from the normal B-DNA structure to the unusual Z-DNA structure. The free ends of the arms shift position by 2-6 nanometers during this fully reversible structural conversion, like a hinge opening and closing. "It's a very simple nanomachine," admits Seeman, "but in the scheme of molecular devices it's huge because it generates more than four times the amount of movement produced by typical molecular devices." A large version of the device might function as an elbow, while smaller devices could serve as finger joints. By 2002, Seeman had demonstrated a mechanical DNA-based rotary motor.
Bernard Yurke at Bell Laboratories and Andrew Turberfield at the University of Oxford synthesized another DNA actuator using three single strands of artificial DNA which, when placed together, find their complementary partners and self-assemble to form a V-shaped structure. The open mouth of this nanotweezer can be made to close by adding a special "fuel" strand which binds to the single-stranded DNA dangling from the ends of the arms of the tweezers and zips them closed. A special "removal" strand, when added, binds to the fuel strand and pulls it away, opening the nanotweezers again. The cycle may then be repeated.
Nanotweezers
In 1999, Philip Kim and Charles Lieber at Harvard University created the first general-purpose nanotweezer. Its working end is a pair of electrically controlled carbon nanotubes made from a bundle of multiwalled carbon nanotubes. To operate the tweezers, a voltage is applied across the electrodes, causing one nanotube arm to develop a positive electrostatic charge and the other to develop a negative charge. The attractive force can be increased or decreased by varying the applied voltage - 8.5 volts completely closes the arms, while lower voltages give different degrees of grip. Using the tool, Kim and Lieber have successfully grasped 500-nanometer clusters of polystyrene spheres, about the same size scale as cellular substructures. They were also able to remove a semiconductor wire 20 nanometers wide from a mass of entangled wires. At present, each of the tweezer's arms is about 50 nanometers wide and 4 microns long. But by growing single-walled nanotubes directly onto the electrodes, the researchers hope to produce nanotweezers small enough to grab individual macromolecules.
Nanomotors
Other researchers are developing nanomotors for future nanorobots. Most notably, Carlo Montemagno at Cornell University has modified a natural biomotor to incorporate nonbiological parts, creating the first artificial hybrid nanomotor. Montemagno started with natural ATPase, a ubiquitous enzyme found in virtually every living organism and which helps to convert food into usable energy in living cells. The moving part of an ATPase molecule is a central protein shaft (or rotor, in electric-motor terms) that rotates in response to electrochemical reactions with each of the molecule's three proton channels (comparable to the electromagnets in the stator coil of an electric motor). ATP (adenosine triphosphate) is the fuel that powers the molecular motor's motion.
Using the tools of genetic engineering, Montemagno added metal-binding amino acid residues to the ATPase. This allowed each motor molecule to bind tightly to nanoscale nickel pedestals prepared by electron beam lithography. Properly oriented motor molecules 12 nanometers in diameter were then attached to the pedestals with a precision approaching 15 nanometers, and a silicon nitride bar a hundred nanometers long was bound to the rotor subunit of each motor molecule, all by self-assembly. In a microscopic video presentation, dozens of bars could be seen spinning like a field of tiny propellers. The group's first integrated molecular motor ran for 40 minutes at 3-4 revolutions per second. Subsequent motors have been operated for hours continuously by feeding them plenty of ATP. Montemagno has been measuring things like horsepower and motor efficiency, simple tests that would be familiar to any mechanical engineer inspecting a car engine. Montemagno is also trying to build a solar-powered, biomolecular motor-driven autonomous nanodevice, wherein light energy is converted into ATP which then serves as a fuel source for the motor. "We think we'll be able to make autonomous devices that are powered by light on a scale of 1 micron or less, smaller than bacteria," he says.
Montemagno is developing a chemical means of switching his hybrid motors on and off reliably. By engineering a secondary binding site tailored to a cell's signaling cascade, he plans to use the sensory system of the living cell to control nanodevices implanted within the cell. Montemagno envisions tiny chemical factories operating inside living cells. He speculates that these nanofactories could be targeted to specific cells, such as those of tumors, where they would synthesize and deliver chemotherapy agents. Within three years he expects to have a motor assembled within a living cell, with the cell's physiology providing the energy to run it. "My 10-year goal is to make a device that harvests single molecules within a living cell, maybe a cellular pharmacy that produces a drug, stores it within the cell, and then based upon some signal, releases it," Montemagno said in 2000. "For a technology that wasn't expected to produce a useful device before the year 2050, I think we've made a pretty good start. But we have a long way to go before it's safe to turn these little machines loose in the human body."
Nanomotor research is progressing in other laboratories as well. For instance, a 78-atom chemically-powered rotating motor was synthesized in 1999 as a proof of principle by chemist T. Ross Kelly at Boston College. Ben Feringa at the University of Groningen in the Netherlands has built an artificial 58-atom motor molecule that spins when illuminated by solar energy. Another potential nanorobot power source is a modified microbial fuel cell - laboratory demonstrations of such cells contain captive bacteria or immobilized enzymes which, when fed organic material, convert chemical energy into electricity that could be used to power tiny motors.
Nanocomputers
Truly effective medical nanorobots may require onboard computers to allow a physician to properly monitor and control their work. Molecular electronics or "moletronics" is a hot research topic in nanotechnology right now. For example, in 2000, a collaborative effort between UCLA and Hewlett Packard produced the first laboratory demonstration of completely reversible room-temperature molecular switches that could be employed in nanoscale memories, using mechanically interlinked ring molecules called catenanes. Two independent companies - Molecular Electronics Corp. in Texas and California Molecular Electronics Corp. in California - have sprung up with the explicit goal of building the first commercial molecular electronic devices including memories and other computational components of computers, possibly in the next few years, using techniques of self-assembly.
Positional Assembly
As machine structures become more complex, getting all the parts to spontaneously self-assemble in the right sequence will be increasingly difficult. To build such complex structures, it makes more sense to design a mechanism that can assemble a molecular structure by what is called positional assembly - that is, picking and placing molecular parts exactly where you want them. A device capable of positional assembly would work much like the robot arms that manufacture cars on automobile assembly lines in Detroit, or which insert electronic components onto computer circuit boards with blinding speed in Silicon Valley. Using the positional assembly approach, the robot manipulator picks up a part, moves it to the workpiece, installs it, then repeats the procedure over and over with many different parts until the final product is fully assembled.
One of the leading proponents of positional assembly is Zyvex Corp., a privately-held molecular nanotechnology company headquartered in Richardson, Texas. Zyvex was the first firm to espouse an explicit goal of using positional assembly to manufacture atomically precise structures, or more specificially, "a user-controlled fabrication tool capable of creating molecularly precise structures with 3-dimensional capability in an economically viable manner." As a first step toward this goal, in 1998 Zyvex demonstrated the ability to use three independently-controlled inch-long robotic arms to manipulate tiny carbon nanotubes in three dimensions, under the watchful eye of a scanning electron microscope that can monitor objects and motions as small as 6 nanometers at near-video scan rates. Zyvex still has a very long way to go before it can assemble nanoscale parts into useful machines, but its work is a small step in the right direction and the research continues today.
Nanomedical Diagnosis and Treatment
The idea of placing autonomous self-powered nanorobots inside of us might seem a bit odd, but actually the human body already teems with such nanodevices. For instance, more than 40 trillion single-celled microbes swim through our colon, outnumbering our tissue cells almost ten to one. Many bacteria move by whipping around a tiny tail, or flagellum, that is driven by a 30-nanometer biological ionic nanomotor powered by pH differences between the inside and the outside of the bacterial cell. Our bodies also maintain a population of more than a trillion motile biological nanodevices called fibroblasts and white cells such as neutrophils and lymphocytes, each measuring perhaps 10 microns in size. These beneficial natural nanorobots are constantly crawling around inside of us, repairing damaged tissues, attacking invading microbes, and gathering up foreign particles and transporting them to various organs for disposal from the body.
The greatest power of nanomedicine will emerge in a decade or two when we learn to design and construct complete artificial nanorobots using nanometer-scale parts and subsystems including sensors, motors, manipulators, power plants, and molecular computers. If we make the reasonable assumption that we will someday be able to build these complex medical nanorobots, and build them cheaply enough and in sufficiently large numbers to be useful therapeutically, then what are the medical implications? We have space here to describe only a few of the many possibilities.
One thing that would change dramatically is clinical diagnostics and treatment. Consider a patient who goes to his doctor with a mild fever, nasal congestion, discomfort, and cough. In the nanomedical era, taking and analyzing microbial samples will be as quick and convenient as the electronic measurement of body temperature using a tympanic thermometer in a late 20th-century clinical office or hospital. The physician faces the patient and pulls from her pocket a lightweight handheld device resembling a pocket calculator. She unsnaps a self-sterilizing cordless pencil-sized probe from the side of the device and inserts the business end of the probe into the patient's opened mouth in the manner of a tongue depressor. The ramifying probe tip contains billions of nanoscale molecular assay receptors mounted on hundreds of self-guiding retractile stalks. Each assay receptor is sensitive to the chemical signature of one of thousands of specific bacterial coats or viral capsids.
The patient says "Ahh," and a few seconds later a three-dimensional color-coded map of the throat area appears on a display panel held in the doctor's hand. A bright spot on the screen marks the exact location where the first samples are being taken. Underneath the color map scrolls a continuously updated microflora count, listing in the leftmost column the names of the ten most numerous microbial and viral species that have been detected, key biochemical marker codes in the middle column, and measured population counts in the right column. The number counts flip up and down a bit as the physician directs probe stalks to various locations in the pharynx to obtain a representative sampling, with special attention to sores or exudate. After a few more seconds, the data for two of the bacterial species suddenly highlight in red, indicating the distinctive molecular signatures of specific toxins or pathological variants. One of these two species is a known, and unwelcome, bacterial pathogen. The diagnosis is completed and the infectious microbe is promptly exterminated using a patient-inhaled aerosol of mobile nanorobots which the physician has programmed to seek out and destroy that one microbial strain. After a few minutes the nanorobots have finished their work and are retrieved by the doctor. A resurvey with the diagnostic probe reveals no evidence of the pathogen.
Improved Human Abilities
Another major change that nanomedicine will bring is the ability to dramatically extend natural human capabilities. As a simple example, a few years ago I designed an artificial mechanical red cell called a "respirocyte." Still entirely theoretical, the respirocyte measures 1 micron in diameter and just floats along in the bloodstream. It is a spherical nanorobot made of 18 billion atoms precisely arranged in a diamondoid structure to make a tiny pressure tank that can be pumped full of up to 3 billion oxygen (O2) and carbon dioxide (CO2) molecules. Later on, these gases can be released from the tank in a controlled manner using tiny molecular pumps. Gases are stored onboard at pressures up to about 1000 atmospheres.
Respirocytes mimic the action of the natural hemoglobin-filled red blood cells. Gas concentration sensors on the outside of each device let the nanorobot know when it is time to load O2 and unload CO2 (at the lungs), or vice versa (at the tissues). Each respirocyte can store and transport 236 times as much gas per unit volume as a natural red cell. So the injection of a 5 cc therapeutic dose of 50% respirocyte saline suspension, a total of 5 trillion individual nanorobots, into the human bloodstream can exactly replace the gas carrying capacity of the patient's entire 5.4 liters of blood. But up to 1 liter of respirocyte suspension could safely be added to the bloodstream, which could keep a patient's tissues safely oxygenated for up to 4 hours in the event a heart attack caused the heart to stop beating. Or it would enable a healthy person to sit quietly at the bottom of a swimming pool for four hours, holding his breath, or to sprint at top speed for at least 15 minutes without breathing.
Similarly, an artificial mechanical platelet or "clottocyte" could make possible complete hemostasis in just 1 second, even for moderately large wounds, a response time 100-1000 times faster than the natural system. The basic clottocyte is conceived as a serum oxygen/glucose-powered spherical nanorobot, 2 microns in diameter, that contains a compactly-folded fiber mesh. Upon command from its control computer, the device unfurls its mesh packet in the vicinity of an injured blood vessel - following, say, a cut through the skin. Soluble thin films coating certain parts of the mesh dissolve upon contact with plasma water, revealing sticky sections (e.g., complementary to blood group antigens unique to red cell surfaces) in desired patterns. Blood cells are immediately trapped in the overlapping artificial nettings released by multiple neighboring activated clottocytes, and bleeding halts at once. While up to 300 natural platelets might be broken and still be insufficient to initiate a self-perpetuating clotting cascade, even a single clottocyte, upon reliably detecting a blood vessel break, can rapidly communicate this fact to its neighboring devices, immediately triggering a progressive carefully-controlled mesh-release cascade. Clottocytes may perform a clotting function that is equivalent in its essentials to that performed by biological platelets, but at only 0.01% of the bloodstream concentration of those cells or about 20 nanorobots per cubic millimeter of serum. Hence clottocytes appear to be about 10,000 times more effective as clotting agents than an equal volume of natural platelets.
Chromosome Replacement Therapy
Medical nanorobots will also be able to intervene at the cellular level, performing in vivo cytosurgery. The most likely site of pathological function in the cell is the nucleus - more specifically, the chromosomes. In one simple cytosurgical procedure, a nanorobot controlled by a physician would extract existing chromosomes from a diseased cell and insert new ones in their place. This is called chromosome replacement therapy. The replacement chromosomes will be manufactured to order, outside of the patient's body in a laboratory benchtop production device that includes a molecular assembly line, using the patient's individual genome as the blueprint. The replacement chromosomes are appropriately demethylated, thus expressing only the appropriate exons that are active in the cell type to which the nanorobot has been targeted. If the patient chooses, inherited defective genes could be replaced with nondefective base-pair sequences, permanently curing a genetic disease. Given the speed with which nanorobots can be administered and their potential rapidity of action, it is possible that an entire whole-body procedure could be completed in one hour or less. Robert Austin at Princeton University has also begun early thinking along these lines, hoping someday to design a nanoprobe capable of identifying biological markers that are specific for targeted diseases. "Then you just pop open the cells, remove the bad DNA from that cell, and repair it on a single-cell level," he says. "That's a long way down the road, but it will happen."
In the first half of the 21st century, nanomedicine should eliminate virtually all common diseases of the 20th century, and virtually all medical pain and suffering as well. Only conditions that involve a permanent loss of personality and memory information in the brain - such as an advanced case of Alzheimer's disease or a massive head trauma - may remain incurable in the nanomedical era. Because aging is believed to be the result of a number of interrelated molecular processes and malfunctions in cells, and because cellular malfunctions will be largely reversible, middle-aged and older people who gain access to an advanced nanomedicine can expect to have most of their youthful health and beauty restored. And they may find few remaining limits to human longevity in this wonderfully vigorous state. It is a bright future that lies ahead for medicine, but we shall all have to work very long and very hard to bring it to fruition.
Nanorobots
Respirocytes: Artificial Mechanical Red Cell
The artificial red blood cell or "respirocyte" is a bloodborne spherical 1-micron diamondoid 1000-atmosphere pressure vessel with active pumping powered by endogenous serum glucose, able to deliver 236 times more oxygen to the tissues per unit volume than natural red cells and to manage carbonic acidity. An onboard nanocomputer and numerous chemical and pressure sensors enable complex device behaviors remotely reprogrammable by the physician via externally applied acoustic signals. Primary applications will include transfusable blood substitution; partial treatment for anemia, perinatal/neonatal and lung disorders; enhancement of cardiovascular/neurovascular procedures, tumor therapies and diagnostics; prevention of asphyxia; artificial breathing; and a variety of sports, veterinary, battlefield and other uses. See "A Mechanical Artificial Red Cell: Exploratory Design in Medical Nanotechnology" for details.
Clottocytes: Artificial Mechanical Platelets (artificial platelets)
The artificial mechanical platelet or clottocyte may allow complete hemostasis in as little as ~1 second, even in moderately large wounds. This response time is on the order of 100-1000 times faster than the natural system. (The) baseline clottocyte is conceived as a serum oxyglucose-powered spherical nanorobot ~2 microns in diameter (~4 micron3 volume) containing a fiber mesh that is compactly folded onboard. Upon command from its control computer, the device promptly unfurls its mesh packet in the immediate vicinity of an injured blood vessel -- following, say, a cut through the skin. Soluble thin films coating certain parts of the mesh dissolve upon contact with plasma water, revealing sticky sections (e.g., complementary to blood group antigens unique to red cell surfaces) in desired patterns. Blood cells are immediately trapped in the overlapping artificial nettings released by multiple neighboring activated clottocytes, and bleeding halts at once. See "Clottocytes: Artificial Mechanical Platelets" for details.
Microbivores: Artificial Mechanical Phagocytes (artificial white cells)
An artificial mechanical phagocytes of microscopic size, called a "microbivore," whose primary function is to destroy microbiological pathogens found in the human bloodstream using a digest and discharge protocol. The microbivore is an oblate spheroidal nanomedical device measuring 3.4 microns in diameter along its major axis and 2.0 microns in diameter along its minor axis, consisting of 610 billion precisely arranged structural atoms in a gross geometric volume of 12.1 micron3. The device may consume up to 200 pW of continuous power while completely digesting trapped microbes at a maximum throughput of 2 micron3 of organic material per 30-second cycle. Microbivores are up to ~1000 times faster-acting than either natural or antibiotic-assisted biological phagocytic defenses, and are ~80 times more efficient as phagocytic agents than macrophages, in terms of volume/sec digested per unit volume of phagocytic agent. See "Microbivores: Artificial Mechanical Phagocytes using Digest and Discharge Protocol" for details, and the Summary article.
Return to Top
Why is it important to pay attention to nanotechnology and nanomedicine?
As advances in our understanding of how things operate at the nanoscale are applied to medicine, we will begin to see (already are seeing) current technologies being disrupted. For example, quantum dots will soon make obsolete the $2.8 billion per year fluorescent tagging market. Manufacturers in this field will need to accommodate new technologies, or they will be overtaken by companies that do.
Nanoknowledge is enabling new technologies that themselves will enable phenomenal changes in the way we diagnose and treat illness and disease; changes that will be as strange and wonderful to us as modern diagnostic tools and treatments would be to someone living 100 years ago. Imagine some of the things we've all seen on Star Trek, such as the medical tricorder, as an everyday part of our lives. An Italian company is already testing a hand-held diagnostic . Given the rate of advance in sensor technology, we could see a 21st century version of the tricoder before the end of this decade.
As the depth and breadth of our understanding of nanoscale science grows (coincident with our realization of its cross-disciplinary nature), scientists and business-people alike are finding myriad new ways to apply advanced technology to our everyday lives. How many of you are thinking about investing in a Segway, or a Hummer, a 6.5 Mega Pixel digital camera, or the multi-Gbyte memory-stick for it, or any of thousands of advanced technologies in the guise of high-end consumer products. Just open any of dozens of high-tech catalogs to get an idea of just how far we've come. Or take a look through the past year's issues of Popular Science, SmallTimes, The Harrow Technology Report, or Science Magazine (among many) to get a to-the-point look at today and tomorrow, as high tech further integrates into our lives.
Nanomedicine will bring changes; some predictable and some not. We can more accurately forecast those changes by understanding the technology and its potential.
Return to Top
What does this mean to me?
A) What does all this mean to me?
- Fewer sick days.
- Lower cost for health care insurance.
- You live longer, in greater comfort, with additional vitality.
- You have more time for leisure activities, hobbies, education, work, etc.
B) What if I am a business-person:
- All the above, plus:
- Improved employee retention.
- Lower product costs.
- Higher profit margins.
- Plus what follows:
C) What if I own or operate a business that is involved in the health care industry:
- Keeping abreast of the latest developments in nanotech is a must, so that my business is not disrupted by new technology.
- Finding ways to adjust my products and services to take advantage of nanotech advances is also a must.
- Being prepared for radical change is, again, a must.
D) How about if I am a doctor, or make my living in modern medicine?
- Everything in A.
- Decreased turnaround in detection, diagnosis, and treatment.
- New tools with which to diagnose, that are smaller, faster, more intelligent, less invasive, and less expensive.
- Lives saved that otherwise may not have been.
In the near future, chances are that you, your children and siblings, co-workers, employees and friends, will get sick less often, which will likely make your life better in many ways. You will incur fewer hospital stays and expenses - which will lower your health insurance, giving you more time and funds to spend on leisure activities.
Return to Top
In closing, these quotes:
"The net result of all these advances in drug discovery, disease detection, and drug delivery will be a paradigm shift of gigantic proportions."
"The question is not if nanotechnology is going to happen but when. Nanotechnology is fueling advances all across the modern industrial spectrum, and things are happening today. And advances that might be thought of as being ten years away today could, as a result of new developments, be only five years away tomorrow."
"It is therefore not too soon to begin thinking about how you and your business will be required to change in the years ahead. Because in many ways nanotechnology is not just going to be the next big thing, it is going to be everything."
From The Next Big Thing Is Really Small: How Nanotechnology Will Change the Future of Your Business. Jack Uldrich & Deb Newberry. March 2003
"New discoveries show that a nanoscale pathogen triggers heart disease, and that a nanobiotic drug gradually gets rid of it. This may transform the business of treating heart disease from damage control to infection prevention. Through that process, the costliest disease in America could be cut down to size -- nano-size."
Douglas Mulhall and Katja Hansen, authors, "Has Heart Disease Been Cured?"
www.calcify.com
Nanoscience will impact medical diagnostic capability,
with orders if magnitude improvement over the next
five years. The growth of in-home test capability
will greatly reduce parental worry and trips to the
clinic.
With the recent breakthrough research we may now be
able to glimpse the "peak" of medical understanding.
How long it takes to climb the mountain is dependent
upon many factors with many boulders and canyons yet
to be discovered and conquered.
Deb Newberry
Return to Top
Nanotechnology and Nanomedicine Artwork can be viewed at these locations:
The Nanomedicine Gallery
Nanotechnology Now Art Gallery
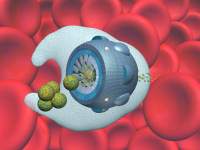
Gobblebot: Artificial White Blood Cell - Tim Fonseca (click to enlarge)
|

Generic Nanobots - Tim Fonseca
(click to enlarge)
|
Regarding the artwork: artwork in these galleries comes from the fertile imaginations of both artists and scientists. Each piece represents the "idea" of a particular device. Should it eventually be build, the actual device may bear little resemblance to the artist's concept - it will however, likely perform as expected.
Return to Top
For further research, here are pages we found valuable:
Nanomedicine Page including a FAQ and links to other valuable resources. By Robert A. Freitas Jr.
Technical Works on Medical Nanorobotics and Nanotechnology By Robert A. Freitas Jr.
Nanomedicine Book Site including free online access to the Nanomedicine technical book series. By Robert A. Freitas Jr.
NanoBiotechnology Glossary By JPK Instruments AG.
Nanomedicine - The Medical Revolution By Albert Tsai.
Molecular Manufacturing: Start Planning By Chris Phoenix.
Nanotechnology and Life Extension By Chris Phoenix.
Return to Top
Interview with Quantum Dot Corporation
NanoNews Now (NN) Editor Rocky Rawstern interviews Quantum Dot Corporation about - what else? - quantum dots!
NN: Please explain to our readers about "nanocrystals" in terms of their technical aspects.
Nanocrystals are nanometer-sized semi-conductor crystals. Just like the larger light emitting semiconductors, for example the ones seen in new traffic lights, these semiconductor nanocrystals emit light. Instead of electrically pumping them, however, we use a different color of light to energize the nanocrystals. The nanocrystals then emit light. The color and brightness of the emitted light is a function of the nanocrystal size, geometry, and synthesis quality. Our QdotTM nanocrystals have several separate layers, each of which contributes to the overall performance, in particular in our focus market of biological detection.
NN: What role has nanoscience played in the development of nanocrystals?
We have had to invent processing and analysis methods as we have developed our nanocrystal synthesis methods. Nanocrystals are the product of nanoscience, and so the two are very interlinked.
NN: How does the term "quantum dot" relate to nanocrystals?
A 'quantum dot' is the technical term for a semi-conductor that confines an electron-hole pair in 3 dimensions - there are also quantum wires and quantum wells that restrict the confinement to 2 or 1 dimension, respectively. Researchers have achieved the creation of quantum dots through the use of electric fields in bulk semi-conductors, and a range of other methods. A nanocrystal is one form of a quantum dot. As they are synthesized and used in a solution, not on a surface, they are the easiest to make and use in bulk.
NN: How does your company produce them?
We produce nanocrystal cores in bulk solutions using methods we have invented here, and also based upon some methods licensed in from MIT and U. Arkansas. The nanocrystal cores form spontaneously in these solutions and, by careful control of the manufacturing conditions, cores of the appropriate size and shape are made. These cores then form the seed for the next shell layer that we grow on the top of these cores, forming a core-shell structure. These structures are the subject of several patents licensed exclusively to Quantum Dot for biological applications. In addition, we have found that high quality biological materials require a high quality shell - a subject of additional patents and know-how. In order to make our Qdot conjugates (for biology), we then grow a polymer layer around the core/shell structure and attach the biological molecules - again these compositions are the subject of many issued and pending patents. This polymer layer process is critical for retaining the bright optical properties of quantum dots when they are in aqueous bio-friendly buffers. So, our products comprise a lot more than just a simple nanocrystal core. Poor methods or technology at any of these steps results in non-functional products. We have built a tremendous franchise of know-how and IP around the methods and compositions of matter required to make useful biological products.
NN: How are nanocrystals used today? What advantages do they bring to the user?
Qdot conjugates, the biologically active quantum dots, are revolutionizing biomolecule detection. These conjugates are extremely bright - up to 1,000 times brighter than conventional methods (as reported by Prof. Watt Webb and colleagues from Cornell and Quantum Dot Corporation in Science on May 30, 2003). They are completely photostable compared to existing biological detection methods, and they allow the detection of many biological molecules at the same time, through the use of different colors of Qdot bioconjugates.
NN: Who developed your technology, and what is their background?
Our scientific founders (primarily Prof. Bawendi from MIT, and Prof. Alivisatos from UC Berkeley) made the initial steps in the use of quantum dots in biological applications. Our company has taken those initial constructs and made real commercial products. Inorganic chemistry, organic chemistry, biochemistry, immunohistochemistry, optics, manufacturing, were all required skills.
NN: How far have you taken the technology? What applications may we see in the future?
We have been supplying our bio-conjugates to life science researchers since November 2002. In the future you'll see a broader array of products on the market, addressing larger segments of the life science research marketplace. You'll also see the launch of our Qdot analyzer, a reader developed by Panasonic for use with a range of Qdot assays in drug discovery and life science research. In the future, you'll also see our technology being used by corporate partners in different in-vitro and in-vivo diagnostic methods.
NN: What is the connection between your company and academia?
We have strong links with UC Berkeley and MIT (see #7). We have a federal grant jointly with Carnegie Mellon. We also have a CRADA agreement with NIH. We have a strong collaboration with Cornell. All of these cover applications and new materials development. Many of our customers are academic biological researchers who we support and interact with.
Company profile:
Quantum Dot Corporation, Hayward, CA
Qdot conjugates - nanometer-sized semi-conductors attached to biological molecules - provide dramatic improvements in sensitivity, information content, and ease-of-use in a broad range of biological detection methods. Quantum Dot Corporation was founded in 1998 from technology licensed out of UC Berkeley (Prof. Alivisatos) and MIT (Prof. Bawendi). We've raised $37.5MM in equity funding, and about $7 million in government grants since then. We have been selling our products to drug discovery and university researchers since November 2002. Having traveled the road from academic curiosity to commercial product, we now have quality manufacturing and a portfolio of products that, our customers tell us, far out-perform the best alternatives. We have a dominant intellectual property position - our portfolio numbers 29 issued patents and >90 under prosecution, covering all aspects of nanocrystal quantum dots including synthesis, compositions, and their applications in biology.
We recently commenced a major strategic partnership with Matsushita Panasonic for Qdot instrumentation - the first jointly developed products will enter customer beta testing in October, 2003 and be launched in April 2004, for high throughput biological research and subsequent diagnostic testing.
Existing biological detection methods, such as fluorescent dyes and gold particles, suffer from many problems such as poor sensitivity, poor stability, and low information content. In many application areas, our products are rapidly displacing these existing techniques. With collaborators and customers, we are finding powerful new applications. For example, our first demonstration of sensitive, high resolution, detection of blood flow was published in May, with scientists from Cornell, in the most highly regarded of all scientific journals, Science. Our markets are varied (several different application areas with markets >$1B each), and are growing at 10-40% per year, an ideal combination for a new company marketing a disruptive technology. With great products currently being marketed, many short-term products close to launch, and many long-term prospects, Quantum Dot Corporation is the world's leading commercial nano-biotechnology corporation.
Return to Top
IN THE NEXT ISSUE
Issue #4 will cover the VC world. It will land in your mailbox October 6th, 2003.
Infamous Quotes:
"There is no reason anyone would want a computer in their home." (Ken Olsen, Digital Equipment Corp, 1977)
"Computers in the future may weigh no more than 1.5 tons." (Popular Mechanics, 1949)
"I think there is a world market for maybe five computers." (IBM's Thomas Watson, 1943)
"This 'telephone' has too many shortcomings to be seriously considered as a means of communication. The device is inherently of no value to us." (Western Union internal memo, 1876)
And the lesson is? It's a tough game to call.
Need advice? Check out NanoStrategies
© Copyright 1999-2003 7thWave, Inc. All rights reserved. More information on
copyright, and disclaimers regarding the content of this newsletter can be
found here
|