Home > Press > Novel MOF shell-derived surface modification of Li-rich layered oxide cathode
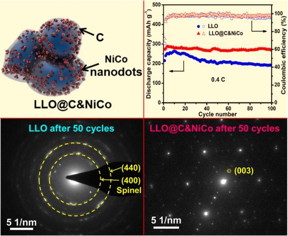 |
| This is a schematic illustration of LLO@C&NiCo; Cycling performance and the corresponding Coulombic efficiencies tested at current densities of 0.4 C; SAED patterns of LLO and LLO@C&NiCo after 50 cycles at 0.4 C. CREDIT �Science China Press |
Abstract:
Rapid development of portable electronics and electric vehicles requires lithium-ion batteries (LIBs) to have high energy/power density, low cost, good safety and long lifespan. In a commercial LIB, the traditional cathode and anode materials are LiCoO2 and graphite, respectively. Compared with the commercial graphite (theoretical?capacity 372 mAh g-1), the LiCoO2 has a low specific capacity of 150 mAh g-1 which becomes a big bottleneck of the battery breakthroughs. Among numerous cathode materials, Li-rich layered oxide (LLO) materials have attracted increasing attention as promising candidates because of their high specific capacity (> 250 mAh g-1) and high operating voltage (> 3.5 V vs. Li+/Li). However, an undesired spinel growth in the layered host structure usually occurred from the surface during the long-term cycling, which led to the fast capacity fading and voltage decaying.
Novel MOF shell-derived surface modification of Li-rich layered oxide cathode
Beijing, China | Posted on December 29th, 2017Prof. L. Q. Mai has led a research team at Wuhan University of Technology aiming to improve the electrochemical performance of electrode materials. They have developed a facile and general carbon coating technology [1]. The unique NiCo nanodots decorated carbon shell was constructed on the as-prepared Li1.2Mn0.54Ni0.13Co0.13O2 nanoparticles masterly on this basis [2]. The obtained cathode exhibits enhanced cycling and rate capability with a capacity retention of 95% after 100 cycles at 0.4 C, 90% after 300 cycles at 2 C and a high capacity of 159 mAh g-1 at 5 C, respectively.
The in-situ X-ray diffraction, electrochemical impedance spectroscopy and selected area electron diffraction analyses after cycling demonstrate that the as a cathode material for LIBs exhibiting superior electrochemical performances, which is due to its unique protective C&NiCo shell. It promotes the electron conductivity (5 times), reduces the diffusion impedance, provides a robust structure for LLO which suppresses the undesired formation of the spinel phase initiated from the particle surface during cycling, and also protects the surface structure from side reactions at the electrode/electrolyte interface.
###
This research was supported by the National Key Research and Development Program of China (2016YFA0202603) and the National Basic Research Program of China (2013CB934103).
####
For more information, please click here
Contacts:
L. Q. Mai
Copyright © Science China Press
If you have a comment, please Contact us.Issuers of news releases, not 7th Wave, Inc. or Nanotechnology Now, are solely responsible for the accuracy of the content.
| Related Links |
| Related News Press |
News and information
![]() Simulating magnetization in a Heisenberg quantum spin chain April 5th, 2024
Simulating magnetization in a Heisenberg quantum spin chain April 5th, 2024
![]() NRL charters Navy�s quantum inertial navigation path to reduce drift April 5th, 2024
NRL charters Navy�s quantum inertial navigation path to reduce drift April 5th, 2024
![]() Discovery points path to flash-like memory for storing qubits: Rice find could hasten development of nonvolatile quantum memory April 5th, 2024
Discovery points path to flash-like memory for storing qubits: Rice find could hasten development of nonvolatile quantum memory April 5th, 2024
Possible Futures
![]() Discovery points path to flash-like memory for storing qubits: Rice find could hasten development of nonvolatile quantum memory April 5th, 2024
Discovery points path to flash-like memory for storing qubits: Rice find could hasten development of nonvolatile quantum memory April 5th, 2024
![]() With VECSELs towards the quantum internet Fraunhofer: IAF achieves record output power with VECSEL for quantum frequency converters April 5th, 2024
With VECSELs towards the quantum internet Fraunhofer: IAF achieves record output power with VECSEL for quantum frequency converters April 5th, 2024
Announcements
![]() NRL charters Navy�s quantum inertial navigation path to reduce drift April 5th, 2024
NRL charters Navy�s quantum inertial navigation path to reduce drift April 5th, 2024
![]() Discovery points path to flash-like memory for storing qubits: Rice find could hasten development of nonvolatile quantum memory April 5th, 2024
Discovery points path to flash-like memory for storing qubits: Rice find could hasten development of nonvolatile quantum memory April 5th, 2024
Interviews/Book Reviews/Essays/Reports/Podcasts/Journals/White papers/Posters
![]() Simulating magnetization in a Heisenberg quantum spin chain April 5th, 2024
Simulating magnetization in a Heisenberg quantum spin chain April 5th, 2024
![]() Discovery points path to flash-like memory for storing qubits: Rice find could hasten development of nonvolatile quantum memory April 5th, 2024
Discovery points path to flash-like memory for storing qubits: Rice find could hasten development of nonvolatile quantum memory April 5th, 2024
Automotive/Transportation
![]() Researchers� approach may protect quantum computers from attacks March 8th, 2024
Researchers� approach may protect quantum computers from attacks March 8th, 2024
![]() Tests find no free-standing nanotubes released from tire tread wear September 8th, 2023
Tests find no free-standing nanotubes released from tire tread wear September 8th, 2023
Battery Technology/Capacitors/Generators/Piezoelectrics/Thermoelectrics/Energy storage
![]() What heat can tell us about battery chemistry: using the Peltier effect to study lithium-ion cells March 8th, 2024
What heat can tell us about battery chemistry: using the Peltier effect to study lithium-ion cells March 8th, 2024
![]() A battery�s hopping ions remember where they�ve been: Seen in atomic detail, the seemingly smooth flow of ions through a battery�s electrolyte is surprisingly complicated February 16th, 2024
A battery�s hopping ions remember where they�ve been: Seen in atomic detail, the seemingly smooth flow of ions through a battery�s electrolyte is surprisingly complicated February 16th, 2024
|
|
||
|
|
||
| The latest news from around the world, FREE | ||
|
|
||
|
|
||
| Premium Products | ||
|
|
||
|
Only the news you want to read!
Learn More |
||
|
|
||
|
Full-service, expert consulting
Learn More |
||
|
|
||








