Home > Press > Scientists make structure analysis of protein molecules several times faster
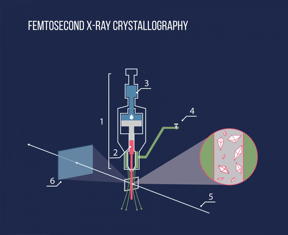 |
| Using the injector (1), the protein crystals dissolved in the lipid medium (2) are exposed to an X-ray beam (5), after which the scattered beam hits a detector (6). Pressure is created in the injector by a hydraulic piston (3), a special gas flow (4) is applied to maintain the straight shape of the lipid stream. CREDIT: MIPT |
Abstract:
Scientists at Moscow Institute of Physics and Technology (MIPT) and several universities in the US came up with a technology for faster structure analysis of receptor proteins, which are important for human health.
Scientists make structure analysis of protein molecules several times faster
Moscow, Russia | Posted on October 12th, 2016An international team of scientists has learnt how to determine the spatial structure of a protein obtained with an X-ray laser using the sulfur atoms it contains. This development is the next stage in the project of a group led by Vadim Cherezov to create an effective method of studying receptor proteins. A detailed description of the study has been published in the journal Science Advances.
Receptor proteins (GPCRs) allow signals to be transmitted within cells, which, in turn, enables the cells to obtain information about their environment and interact with one another. As a result, we are able to see, feel, maintain blood pressure etc., i.e. everything that is needed for the functioning of our bodies. Any disorders in the way these proteins work can result in serious consequences, such as blindness. Developing medicines to restore the normal function of receptors is not possible without a precise understanding of the way in which GPCRs operate, which, as with other proteins, is determined by their spatial structure, i.e. the way in which the protein folds.
The best method of doing this is to use X-ray crystallography. For X-rays, a crystal is a three-dimensional diffraction lattice in which the radiation is scattered on the atoms.
A particular problem with this method is obtaining protein crystals. In order to do this, receptor proteins have to be extracted from a cell membrane and placed in a special lipid environment. Then, by selecting the temperature and using substances to speed up the nucleation process, the protein crystallizes.
One challenge with GPCRs is that they are highly mobile and dynamic molecules that frequently change their spatial structure. This means that it is difficult for them to grow large crystals that are needed for the classical diffraction procedure. This procedure involves exposing the crystal to radiation at different angles for a relatively long period of time. X-rays ionize the atoms, which destroys the protein molecules. Large crystals of a few dozen microns are what is needed to compensate for this effect.
Thanks to the new experimental method of Serial Femtosecond Crystallography, it is now possible to solve this problem. The method has been developed over the past few years by an international team of scientists from Arizona State University and the University of Zurich, SLAC National Accelerator Laboratory in Stanford, the iHuman Institute at ShanghaiTech University, the Institute of Biophysics of the Chinese Academy of Sciences, the CFEL center in Hamburg, the University of Southern California and MIPT. One of the leaders of the team is Vadim Cherezov, a professor at the University of Southern California and MIPT. The method is based on the use of new generation X-ray sources - free-electron lasers. The radiation they emit is so powerful that it fully ionizes atoms in a crystal as it passes through, essentially destroying it. However, as the laser pulse has a very short duration (a few femtoseconds, 10-15 s), a diffraction pattern can be recorded before the atoms move from their position. This has meant that the scientists are able to avoid the difficulties associated with radiation damage.
As the crystal is destroyed immediately, it is not possible to measure it at different orientations. To solve this problem, scientists collect and process data from several crystals. Using a special injector, the lipid environment in which the crystals are situated is exposed to an X-ray pulse. The whole process is similar to squeezing toothpaste out of a tube.
The result is millions of diffraction images that need to be processed: by selecting images with crystals, finding their orientation, and then putting them together in a three-dimensional diffraction pattern. Two parameters must be known in order to decipher the structure: the amplitude and the phase of the reflected radiation. The amplitude value is measured on a detector during the experiment, however determining the phase is a complex task and there are a number of ways of solving the problem.
For example, if we know of a certain protein that has a similar structure, we can use it as a first approximation. Of course, this is not possible in all cases.
Another popular method is to use an effect known as anomalous scattering. This occurs when the X-ray wavelength is close to the electron transition energy in the atoms, which results in the wave being absorbed and re-emitted. This causes a change in the amplitudes and phases. If the amplitudes are measured very precisely, the differences between them can be used to reconstruct the phases. However, most of the atoms that make up proteins (carbon, oxygen and nitrogen) are not suitable for this. A relatively heavy element found in almost all proteins is sulfur, and this is the element the researchers used in their most recent study to reconstruct the phases.
Special software had to be developed specifically for the task. Out of 7 million images obtained, the researchers had to pick out those with diffracted reflections. They then had to determine the orientation of the crystal and the intensity of all reflections and subsequently collate all the data obtained. 600,000 diffraction patterns were found and then used to successfully reconstruct the structure of a protein with a resolution of 2.5�. By combining the data with the results obtained at a different X-ray wavelength, the researchers were able to increase the resolution to 1.9�. This level of precision not only enables the structure of receptor proteins to be determined with high accuracy, but also allows scientists to see molecules of water, ions and lipids that surround them, which is extremely important for understanding how proteins function and modeling their interaction with other substances.
"When I participated in a study to determine the structure of the first receptor protein, it took me about a year to obtain crystals that were large enough to conduct classical X-ray diffraction. We hope that the method we have developed will help to greatly increase the speed of this process," said Prof. Cherezov commenting on the significance of the research.
Of the 800 receptor proteins that exist, we currently know the structure of only 34. The experimental method developed by the scientists will significantly speed up the studies of the remaining proteins. This, in turn, will help in developing new and effective drugs to treat a vast number of diseases.
####
For more information, please click here
Contacts:
Asya Shepunova
7-916-813-0267
Copyright © Moscow Institute of Physics and Technology
If you have a comment, please Contact us.Issuers of news releases, not 7th Wave, Inc. or Nanotechnology Now, are solely responsible for the accuracy of the content.
| Related Links |
| Related News Press |
News and information
![]() Simulating magnetization in a Heisenberg quantum spin chain April 5th, 2024
Simulating magnetization in a Heisenberg quantum spin chain April 5th, 2024
![]() NRL charters Navy�s quantum inertial navigation path to reduce drift April 5th, 2024
NRL charters Navy�s quantum inertial navigation path to reduce drift April 5th, 2024
![]() Discovery points path to flash-like memory for storing qubits: Rice find could hasten development of nonvolatile quantum memory April 5th, 2024
Discovery points path to flash-like memory for storing qubits: Rice find could hasten development of nonvolatile quantum memory April 5th, 2024
Imaging
![]() Nanoscale CL thermometry with lanthanide-doped heavy-metal oxide in TEM March 8th, 2024
Nanoscale CL thermometry with lanthanide-doped heavy-metal oxide in TEM March 8th, 2024
![]() The USTC realizes In situ electron paramagnetic resonance spectroscopy using single nanodiamond sensors November 3rd, 2023
The USTC realizes In situ electron paramagnetic resonance spectroscopy using single nanodiamond sensors November 3rd, 2023
![]() Observation of left and right at nanoscale with optical force October 6th, 2023
Observation of left and right at nanoscale with optical force October 6th, 2023
Crystallography
![]() First measurement of electron energy distributions, could enable sustainable energy technologies June 5th, 2020
First measurement of electron energy distributions, could enable sustainable energy technologies June 5th, 2020
![]() How to trick electrons to see the hidden face of crystals: Researchers try a trick for complete 3D analysis of submicron crystals August 3rd, 2019
How to trick electrons to see the hidden face of crystals: Researchers try a trick for complete 3D analysis of submicron crystals August 3rd, 2019
![]() 3-D-printed jars in ball-milling experiments June 29th, 2017
3-D-printed jars in ball-milling experiments June 29th, 2017
![]() Novel nozzle saves crystals: Double flow concept widens spectrum for protein crystallography March 17th, 2017
Novel nozzle saves crystals: Double flow concept widens spectrum for protein crystallography March 17th, 2017
Possible Futures
![]() Discovery points path to flash-like memory for storing qubits: Rice find could hasten development of nonvolatile quantum memory April 5th, 2024
Discovery points path to flash-like memory for storing qubits: Rice find could hasten development of nonvolatile quantum memory April 5th, 2024
![]() With VECSELs towards the quantum internet Fraunhofer: IAF achieves record output power with VECSEL for quantum frequency converters April 5th, 2024
With VECSELs towards the quantum internet Fraunhofer: IAF achieves record output power with VECSEL for quantum frequency converters April 5th, 2024
Nanomedicine
![]() New micromaterial releases nanoparticles that selectively destroy cancer cells April 5th, 2024
New micromaterial releases nanoparticles that selectively destroy cancer cells April 5th, 2024
![]() Good as gold - improving infectious disease testing with gold nanoparticles April 5th, 2024
Good as gold - improving infectious disease testing with gold nanoparticles April 5th, 2024
![]() Researchers develop artificial building blocks of life March 8th, 2024
Researchers develop artificial building blocks of life March 8th, 2024
Announcements
![]() NRL charters Navy�s quantum inertial navigation path to reduce drift April 5th, 2024
NRL charters Navy�s quantum inertial navigation path to reduce drift April 5th, 2024
![]() Discovery points path to flash-like memory for storing qubits: Rice find could hasten development of nonvolatile quantum memory April 5th, 2024
Discovery points path to flash-like memory for storing qubits: Rice find could hasten development of nonvolatile quantum memory April 5th, 2024
Interviews/Book Reviews/Essays/Reports/Podcasts/Journals/White papers/Posters
![]() Simulating magnetization in a Heisenberg quantum spin chain April 5th, 2024
Simulating magnetization in a Heisenberg quantum spin chain April 5th, 2024
![]() Discovery points path to flash-like memory for storing qubits: Rice find could hasten development of nonvolatile quantum memory April 5th, 2024
Discovery points path to flash-like memory for storing qubits: Rice find could hasten development of nonvolatile quantum memory April 5th, 2024
Tools
![]() Ferroelectrically modulate the Fermi level of graphene oxide to enhance SERS response November 3rd, 2023
Ferroelectrically modulate the Fermi level of graphene oxide to enhance SERS response November 3rd, 2023
![]() The USTC realizes In situ electron paramagnetic resonance spectroscopy using single nanodiamond sensors November 3rd, 2023
The USTC realizes In situ electron paramagnetic resonance spectroscopy using single nanodiamond sensors November 3rd, 2023
Nanobiotechnology
![]() New micromaterial releases nanoparticles that selectively destroy cancer cells April 5th, 2024
New micromaterial releases nanoparticles that selectively destroy cancer cells April 5th, 2024
![]() Good as gold - improving infectious disease testing with gold nanoparticles April 5th, 2024
Good as gold - improving infectious disease testing with gold nanoparticles April 5th, 2024
![]() Researchers develop artificial building blocks of life March 8th, 2024
Researchers develop artificial building blocks of life March 8th, 2024
|
|
||
|
|
||
| The latest news from around the world, FREE | ||
|
|
||
|
|
||
| Premium Products | ||
|
|
||
|
Only the news you want to read!
Learn More |
||
|
|
||
|
Full-service, expert consulting
Learn More |
||
|
|
||








