Home > Press > Seeing how a lithium-ion battery works: An exotic state of matter -- a 'random solid solution' -- affects how ions move through battery material
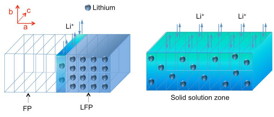 |
| Diagram illustrates the process of charging or discharging the lithium iron phosphate (LFP) electrode. As lithium ions are removed during the charging process, it forms a lithium-depleted iron phosphate (FP) zone, but in between there is a solid solution zone (SSZ, shown in dark blue-green) containing some randomly distributed lithium atoms, unlike the orderly array of lithium atoms in the original crystalline material (light blue). This work provides the first direct observations of this SSZ phenomenon.
Illustration courtesy of the authors |
Abstract:
New observations by researchers at MIT have revealed the inner workings of a type of electrode widely used in lithium-ion batteries. The new findings explain the unexpectedly high power and long cycle life of such batteries, the researchers say.
Seeing how a lithium-ion battery works: An exotic state of matter -- a 'random solid solution' -- affects how ions move through battery material
Cambridge, MA | Posted on June 9th, 2014The findings appear in a paper in the journal Nano Letters co-authored by MIT postdoc Jun Jie Niu, research scientist Akihiro Kushima, professors Yet-Ming Chiang and Ju Li, and three others.
The electrode material studied, lithium iron phosphate (LiFePO4), is considered an especially promising material for lithium-based rechargeable batteries; it has already been demonstrated in applications ranging from power tools to electric vehicles to large-scale grid storage. The MIT researchers found that inside this electrode, during charging, a solid-solution zone (SSZ) forms at the boundary between lithium-rich and lithium-depleted areas � the region where charging activity is concentrated, as lithium ions are pulled out of the electrode.
Li says that this SSZ "has been theoretically predicted to exist, but we see it directly for the first time," in transmission electron microscope (TEM) videos taken during charging.
The observations help to resolve a longstanding puzzle about LiFePO4: In bulk crystal form, both lithium iron phosphate and iron phosphate (FePO4, which is left behind as lithium ions migrate out of the material during charging) have very poor ionic and electrical conductivities. Yet when treated � with doping and carbon coating � and used as nanoparticles in a battery, the material exhibits an impressively high charging rate. "It was quite surprising when this [rapid charging and discharging rate] was first demonstrated," Li says.
"We directly observed a metastable random solid solution that may resolve this fundamental problem that has intrigued [materials scientists] for many years," says Li, the Battelle Energy Alliance Professor of Nuclear Science and Engineering and a professor of materials science and engineering.
The SSZ is a "metastable" state, persisting for at least several minutes at room temperature. Replacing a sharp interface between LiFePO4 and FePO4 that has been shown to contain many additional line defects called "dislocations," the SSZ serves as a buffer, reducing the number of dislocations that would otherwise move with the electrochemical reaction front. "We don't see any dislocations," Li says. This could be important because the generation and storage of dislocations can cause fatigue and limit the cycle life of an electrode.
Unlike conventional TEM imaging, the technique used in this work, developed in 2010 by Kushima and Li, makes it possible to observe battery components as they charge and discharge, which can reveal dynamic processes. "In the last four years, there has been a big explosion of using such in situ TEM techniques to study battery operations," Li says.
A better understanding of these dynamic processes could improve the performance of an electrode material by allowing better tuning of its properties, Li says.
Despite an incomplete understanding to date, lithium iron phosphate nanoparticles are already used at an industrial scale for lithium-ion batteries, Li explains. "The science is lagging behind the application," he says. "It's already scaled up and quite successful on the market. It's one of the success stories of nanotechnology."
"Compared to traditional lithium-ion, [lithium iron phosphate] is environmentally friendly, and very stable," Niu says. "But it's important for this material to be well understood."
While the discovery of the SSZ was made in LiFePO4, Li says, "The same principle may apply to other electrode materials. People are looking for high-power electrode materials, and such metastable states could exist in other electrode materials that are inert in bulk form. � The phenomenon discovered could be very general, and not specific to this material."
###
The research was supported by the National Science Foundation.
Written by David Chandler, MIT News Office
####
For more information, please click here
Contacts:
Andrew Carleen
617-253-1682
Copyright © Massachusetts Institute of Technology
If you have a comment, please Contact us.Issuers of news releases, not 7th Wave, Inc. or Nanotechnology Now, are solely responsible for the accuracy of the content.
| Related News Press |
News and information
![]() Simulating magnetization in a Heisenberg quantum spin chain April 5th, 2024
Simulating magnetization in a Heisenberg quantum spin chain April 5th, 2024
![]() NRL charters Navy�s quantum inertial navigation path to reduce drift April 5th, 2024
NRL charters Navy�s quantum inertial navigation path to reduce drift April 5th, 2024
![]() Discovery points path to flash-like memory for storing qubits: Rice find could hasten development of nonvolatile quantum memory April 5th, 2024
Discovery points path to flash-like memory for storing qubits: Rice find could hasten development of nonvolatile quantum memory April 5th, 2024
Imaging
![]() Nanoscale CL thermometry with lanthanide-doped heavy-metal oxide in TEM March 8th, 2024
Nanoscale CL thermometry with lanthanide-doped heavy-metal oxide in TEM March 8th, 2024
![]() The USTC realizes In situ electron paramagnetic resonance spectroscopy using single nanodiamond sensors November 3rd, 2023
The USTC realizes In situ electron paramagnetic resonance spectroscopy using single nanodiamond sensors November 3rd, 2023
![]() Observation of left and right at nanoscale with optical force October 6th, 2023
Observation of left and right at nanoscale with optical force October 6th, 2023
Govt.-Legislation/Regulation/Funding/Policy
![]() NRL charters Navy�s quantum inertial navigation path to reduce drift April 5th, 2024
NRL charters Navy�s quantum inertial navigation path to reduce drift April 5th, 2024
![]() Discovery points path to flash-like memory for storing qubits: Rice find could hasten development of nonvolatile quantum memory April 5th, 2024
Discovery points path to flash-like memory for storing qubits: Rice find could hasten development of nonvolatile quantum memory April 5th, 2024
![]() Chemical reactions can scramble quantum information as well as black holes April 5th, 2024
Chemical reactions can scramble quantum information as well as black holes April 5th, 2024
Discoveries
![]() Chemical reactions can scramble quantum information as well as black holes April 5th, 2024
Chemical reactions can scramble quantum information as well as black holes April 5th, 2024
![]() New micromaterial releases nanoparticles that selectively destroy cancer cells April 5th, 2024
New micromaterial releases nanoparticles that selectively destroy cancer cells April 5th, 2024
![]() Utilizing palladium for addressing contact issues of buried oxide thin film transistors April 5th, 2024
Utilizing palladium for addressing contact issues of buried oxide thin film transistors April 5th, 2024
Announcements
![]() NRL charters Navy�s quantum inertial navigation path to reduce drift April 5th, 2024
NRL charters Navy�s quantum inertial navigation path to reduce drift April 5th, 2024
![]() Discovery points path to flash-like memory for storing qubits: Rice find could hasten development of nonvolatile quantum memory April 5th, 2024
Discovery points path to flash-like memory for storing qubits: Rice find could hasten development of nonvolatile quantum memory April 5th, 2024
Interviews/Book Reviews/Essays/Reports/Podcasts/Journals/White papers/Posters
![]() Simulating magnetization in a Heisenberg quantum spin chain April 5th, 2024
Simulating magnetization in a Heisenberg quantum spin chain April 5th, 2024
![]() Discovery points path to flash-like memory for storing qubits: Rice find could hasten development of nonvolatile quantum memory April 5th, 2024
Discovery points path to flash-like memory for storing qubits: Rice find could hasten development of nonvolatile quantum memory April 5th, 2024
Tools
![]() Ferroelectrically modulate the Fermi level of graphene oxide to enhance SERS response November 3rd, 2023
Ferroelectrically modulate the Fermi level of graphene oxide to enhance SERS response November 3rd, 2023
![]() The USTC realizes In situ electron paramagnetic resonance spectroscopy using single nanodiamond sensors November 3rd, 2023
The USTC realizes In situ electron paramagnetic resonance spectroscopy using single nanodiamond sensors November 3rd, 2023
Battery Technology/Capacitors/Generators/Piezoelectrics/Thermoelectrics/Energy storage
![]() What heat can tell us about battery chemistry: using the Peltier effect to study lithium-ion cells March 8th, 2024
What heat can tell us about battery chemistry: using the Peltier effect to study lithium-ion cells March 8th, 2024
![]() A battery�s hopping ions remember where they�ve been: Seen in atomic detail, the seemingly smooth flow of ions through a battery�s electrolyte is surprisingly complicated February 16th, 2024
A battery�s hopping ions remember where they�ve been: Seen in atomic detail, the seemingly smooth flow of ions through a battery�s electrolyte is surprisingly complicated February 16th, 2024
|
|
||
|
|
||
| The latest news from around the world, FREE | ||
|
|
||
|
|
||
| Premium Products | ||
|
|
||
|
Only the news you want to read!
Learn More |
||
|
|
||
|
Full-service, expert consulting
Learn More |
||
|
|
||








