Home > Press > Endomagnetics Appoints Strategic Distributor
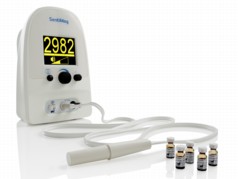 |
| Complete SentiMag system |
Abstract:
Endomagnetics, the company developing a portfolio of products to improve the standard of breast cancer management, has announced it has signed a distribution agreement with Sysmex Europe GmbH, a subsidiary of global healthcare company Sysmex Corporation. Sysmex has the exclusive right to provide sales and support for Endomagnetics' SentiMag® system across the EMEA region. This is Endomagnetics' first multi-region agreement, and it promises to extend widely the clinical use of the company's technology.
Endomagnetics Appoints Strategic Distributor
Cambridge, UK | Posted on March 1st, 2013The SentiMag® system is used as part of the current standard sentinel lymph node (SLNB) technique, used by surgeons to monitor the spread of cancer and to plan interventions. Unlike the present system, Endomagnetics' technology can be used by surgeons as part of an efficient workflow, and does not require specialist technicians to store and manage radioisotopes. Patients are spared the anxiety of radioactivity and the possible need to attend multiple clinics. The system consists of the highly sensitive SentiMag® instrument and its associated tracer, Sienna+®. The system is currently in use in eight countries across the EU: UK, Germany, Poland, France, Italy, Belgium, Holland, and Switzerland.
Dr Marc Thill, Head of the Department of Gynaecology and Obstetrics and the Breast Cancer Centre at Agaplesion Markus Hospital, Frankfurt reported: "After working with the SentiMag® technique in my daily routine for the past few months, I have concluded that this technique is an easy-to-use approach for performing SLNB without radioisotopes or any time-consuming pre-operative preparation of the patient. For that reason it may ultimately replace the standard of care."
Endomagnetics cited Sysmex's strong history of customer support, compatible product pipeline and focus on healthcare innovation in developing this strategic relationship. Sysmex is the global market leader in haematology, and has built on its successes in diagnostics to innovate in the life science field. In particular, Sysmex has developed a molecular diagnostics assay called OSNA (One Step Nucleic Acid Amplification). This assay enables complete lymph nodes to be analysed quickly, and is used routinely for analysing sentinel lymph nodes for breast cancer in about 200 hospitals across Europe.
"SentiMag® offers hospitals and clinics a powerful, efficient new way of locating lymph nodes to verify the spread of breast cancer," said Dr Michael Schaefer, Managing Director of Sysmex Europe. "It is an excellent strategic fit with our product portfolio and an ideal complement to our OSNA system. Our sales teams across the EU have received very positive feedback from customers."
CEO of Endomagnetics Dr Eric Mayes commented: "We are extremely pleased with initial customer feedback, and partnering with a group as strong as Sysmex gives us the geographic scope and customer support we need to really make an impact. We have been extremely impressed with the enthusiasm of the Sysmex team in supporting our joint product launch."
####
About Endomagnetics
Endomagnetics was founded to solve cancer staging and healthcare challenges through the application of advanced magnetic sensing technology and nanotechnology. Endomagnetics is developing a portfolio of medical device products based on a patented ability to detect magnetic materials in the human body with exceptional sensitivity. The technology was originally developed at University College London and the University of Houston.
The company’s first product, the SentiMag®, is an ultrasensitive hand-held probe for tracking injected Sienna+® nanoscale magnetic materials in the human body. Cambridge, UK-based Endomagnetics is in the process of delivering its technology to global markets. For more information please visit www.endomagnetics.com.
About Sysmex
Sysmex is a global group, with more than 40 overseas subsidiaries conducting business in more than 170 countries. It develops, produces and sells instruments, reagents and software used in in-vitro diagnostics in hospitals and testing centres. In the global market, Sysmex ranks in the top 10 in diagnostics. In haematology, it holds the top share of the global market.
Overseas business development has been a focus for Sysmex since it was founded. It operates R&D, production, sales and customer support facilities around the world to ensure products and services are delivered that meet the needs of the customer in each region. Principal clinical areas of activity are: Haematology, Haemostasis, Immunochemistry, Urinalysis, Clinical Chemistry and Life Science.
www.sysmex.com
For more information, please click here
Contacts:
Editorial contact:
Terry Nicklin
KeynotePR Ltd
Tel: +44 (0)7923 540695
Company contact:
Dr Eric Mayes, CEO
Endomagnetics Ltd
Tel: +44 (0)1223 652540
Copyright © Endomagnetics
If you have a comment, please Contact us.Issuers of news releases, not 7th Wave, Inc. or Nanotechnology Now, are solely responsible for the accuracy of the content.
| Related News Press |
News and information
![]() Simulating magnetization in a Heisenberg quantum spin chain April 5th, 2024
Simulating magnetization in a Heisenberg quantum spin chain April 5th, 2024
![]() NRL charters Navy’s quantum inertial navigation path to reduce drift April 5th, 2024
NRL charters Navy’s quantum inertial navigation path to reduce drift April 5th, 2024
![]() Discovery points path to flash-like memory for storing qubits: Rice find could hasten development of nonvolatile quantum memory April 5th, 2024
Discovery points path to flash-like memory for storing qubits: Rice find could hasten development of nonvolatile quantum memory April 5th, 2024
Nanomedicine
![]() New micromaterial releases nanoparticles that selectively destroy cancer cells April 5th, 2024
New micromaterial releases nanoparticles that selectively destroy cancer cells April 5th, 2024
![]() Good as gold - improving infectious disease testing with gold nanoparticles April 5th, 2024
Good as gold - improving infectious disease testing with gold nanoparticles April 5th, 2024
![]() Researchers develop artificial building blocks of life March 8th, 2024
Researchers develop artificial building blocks of life March 8th, 2024
Announcements
![]() NRL charters Navy’s quantum inertial navigation path to reduce drift April 5th, 2024
NRL charters Navy’s quantum inertial navigation path to reduce drift April 5th, 2024
![]() Discovery points path to flash-like memory for storing qubits: Rice find could hasten development of nonvolatile quantum memory April 5th, 2024
Discovery points path to flash-like memory for storing qubits: Rice find could hasten development of nonvolatile quantum memory April 5th, 2024
Alliances/Trade associations/Partnerships/Distributorships
![]() Chicago Quantum Exchange welcomes six new partners highlighting quantum technology solutions, from Chicago and beyond September 23rd, 2022
Chicago Quantum Exchange welcomes six new partners highlighting quantum technology solutions, from Chicago and beyond September 23rd, 2022
![]() University of Illinois Chicago joins Brookhaven Lab's Quantum Center June 10th, 2022
University of Illinois Chicago joins Brookhaven Lab's Quantum Center June 10th, 2022
|
|
||
|
|
||
| The latest news from around the world, FREE | ||
|
|
||
|
|
||
| Premium Products | ||
|
|
||
|
Only the news you want to read!
Learn More |
||
|
|
||
|
Full-service, expert consulting
Learn More |
||
|
|
||








