Home > Press > BIOX: Amorphous iron oxide nanostructures of bacterial origin for applications including anodes for Li ion batteries
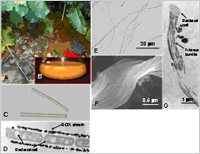 |
| Fig. 1. (A) Ocherous deposits ubiquitously seen in water pools. (B) BIOX deposits collected from a water pool. (C) BIOX sheaths in the deposit. (D) A longitudinal section of a BIOX sheath enveloping bacterial cells. (E) Chain-like BIOX (F) A twisting BIOX bundle comprised of fibrous materials. (G) Fibrous bundles connecting to a bacterial cell. |
Abstract:
Professor Jun Takada, Graduate School of Natural Science and Technology
"Iron-oxidizing bacteria" produce extracellular, uniquely-shaped microsheaths or fibrous bundle nanostructures comprising mainly of iron oxides—known as Biogenous iron oxides (BIOX)—ubiquitously in natural hydrosphere at ambient temperature (Fig. 1).
BIOX: Amorphous iron oxide nanostructures of bacterial origin for applications including anodes for Li ion batteries
Okayama, Japan | Posted on September 19th, 2012Although BIOX has been generally recognized as waste, we have studied its properties for as yet unknown potential industrial applications. Our careful and focused studies revealed BIOX matrix to have the following physical properties: (i) an amorphous state; (ii) consist of organic/inorganic hybrid of nanoparticles of approximately 3 nm diameter; (iii) the nanoparticles are composed of many elements, C, O, Fe, Si and P; (iv) inorganic elements are linked via oxygen.
Importantly, BIOX has a far superior potential (for example a large capacity) as an anode material of Li-ion batteries compared to conventional carbon anodes. In addition, BIOX exhibits an amazing, wide range of functions compared with other materials currently: (i) higher catalytic potential; (ii) higher affinity to human cells; and (iii) brighter color property. All these characters are superior to those of artificially synthesized iron oxides. We are confident that the eco-friendly, nontoxic, and low-cost BIOX will be a next-generation functional material.
Detailed studies of an isolated strain of one type of the bacteria led us to elucidate the incipient mechanism of BIOX formation. Our experiments showed that extracellular secretion of bacterial polymers triggers deposition and binding of aquatic inorganics such as Fe, Si, and P, which results in the unique organic/inorganic hybrid. Further analysis is in progress for a greater insight into how the mechanism and mode of chemical linkages in the BIOX matrix contribute to the aforementioned functions.
Technical publications
H. Hashimoto et al, "Characteristics of hollow microtubes consisting of amorphous iron oxide nanoparticles produced by iron oxidizing bacteria Leptothrix ochracea," Journal of Magnetism and Magnetic Materials, 310, 2405, (2007).
T. Sakai et al, "Chemical modification of biogenous iron oxide to create an excellent enzyme scaffold," Organic Biomolecular Chemistry, 8, 336 (2010).
T. Ema et al, "Highly active lipase immobilized on biogenous iron oxide via an organic bridging group: the dramatic effect of the immobilization support on enzymatic function," Green Chemistry, 13, 3187 (2011).
T. Suzuki et al, "Environmental microbiology: silicon and phosphorus linkage with iron via oxygen in the amorphous matrix of Gallionella ferruginea stalks", Applied and Environmental Microbiology, 78, 236 (2012).
M. Furutani et al, "Initial assemblage of bacterial saccharic fibrils and element deposition to form an immature sheath in cultured Leptothrix sp. strain OUMS1", Minerals, 1, 157, (2011).
K. Mandai et al, "Iron oxide-immobilized palladium catalyst for the solvent-free Suzuki-Miyaura coupling reaction", Tetrahedron Letters, 53, 329, (2012).
H. Hashimoto et al, "Preparation, microstructure, and colour tone of microtubule material composed of hematite/amorphous-silicate nanocomposite from iron oxide of bacterial origin", Dyes and Pigments. Available online 6 July 2012. (doi. 10.1016/j.dyepig.2012.06.024).
####
About Okayama University
Okayama University is one of the largest comprehensive universities in Japan with roots going back to the Medical Training Place sponsored by the Lord of Okayama and established in 1870. Now with 1,300 faculty and 14,000 students, the University offers courses in specialties ranging from medicine and pharmacy to humanities and physical sciences. Okayama University is located in the heart of Japan approximately 3 hours west of Tokyo by Shinkansen.
For more information, please click here
Contacts:
Okayama University
1-1-1 Tsushima-naka , Kita-ku ,
Okayama 700-8530, Japan
Planning and Public Information Division
Copyright © Okayama University
If you have a comment, please Contact us.Issuers of news releases, not 7th Wave, Inc. or Nanotechnology Now, are solely responsible for the accuracy of the content.
| Related News Press |
News and information
![]() Simulating magnetization in a Heisenberg quantum spin chain April 5th, 2024
Simulating magnetization in a Heisenberg quantum spin chain April 5th, 2024
![]() NRL charters Navy’s quantum inertial navigation path to reduce drift April 5th, 2024
NRL charters Navy’s quantum inertial navigation path to reduce drift April 5th, 2024
![]() Discovery points path to flash-like memory for storing qubits: Rice find could hasten development of nonvolatile quantum memory April 5th, 2024
Discovery points path to flash-like memory for storing qubits: Rice find could hasten development of nonvolatile quantum memory April 5th, 2024
Discoveries
![]() Chemical reactions can scramble quantum information as well as black holes April 5th, 2024
Chemical reactions can scramble quantum information as well as black holes April 5th, 2024
![]() New micromaterial releases nanoparticles that selectively destroy cancer cells April 5th, 2024
New micromaterial releases nanoparticles that selectively destroy cancer cells April 5th, 2024
![]() Utilizing palladium for addressing contact issues of buried oxide thin film transistors April 5th, 2024
Utilizing palladium for addressing contact issues of buried oxide thin film transistors April 5th, 2024
Announcements
![]() NRL charters Navy’s quantum inertial navigation path to reduce drift April 5th, 2024
NRL charters Navy’s quantum inertial navigation path to reduce drift April 5th, 2024
![]() Discovery points path to flash-like memory for storing qubits: Rice find could hasten development of nonvolatile quantum memory April 5th, 2024
Discovery points path to flash-like memory for storing qubits: Rice find could hasten development of nonvolatile quantum memory April 5th, 2024
Battery Technology/Capacitors/Generators/Piezoelectrics/Thermoelectrics/Energy storage
![]() What heat can tell us about battery chemistry: using the Peltier effect to study lithium-ion cells March 8th, 2024
What heat can tell us about battery chemistry: using the Peltier effect to study lithium-ion cells March 8th, 2024
![]() A battery’s hopping ions remember where they’ve been: Seen in atomic detail, the seemingly smooth flow of ions through a battery’s electrolyte is surprisingly complicated February 16th, 2024
A battery’s hopping ions remember where they’ve been: Seen in atomic detail, the seemingly smooth flow of ions through a battery’s electrolyte is surprisingly complicated February 16th, 2024
Nanobiotechnology
![]() New micromaterial releases nanoparticles that selectively destroy cancer cells April 5th, 2024
New micromaterial releases nanoparticles that selectively destroy cancer cells April 5th, 2024
![]() Good as gold - improving infectious disease testing with gold nanoparticles April 5th, 2024
Good as gold - improving infectious disease testing with gold nanoparticles April 5th, 2024
![]() Researchers develop artificial building blocks of life March 8th, 2024
Researchers develop artificial building blocks of life March 8th, 2024
|
|
||
|
|
||
| The latest news from around the world, FREE | ||
|
|
||
|
|
||
| Premium Products | ||
|
|
||
|
Only the news you want to read!
Learn More |
||
|
|
||
|
Full-service, expert consulting
Learn More |
||
|
|
||








