Home > Press > Graphene’s behavior depends on where it sits: New findings show that the material beneath the thin carbon sheets determines how they react chemically and electrically
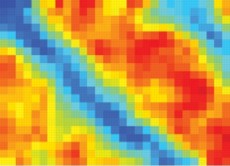 |
| An image (made with Raman spectroscopy) of a graphene layer on top of a patterned substrate shows the difference in chemical reactivity of the side opposite the substrate. The wide red stripe is an area over a silicon dioxide substrate, making the top surface of the graphene highly reactive. The narrow blue stripe is graphene over a layer of hydrocarbon (called OTS), and there is almost no reactivity on the side not in contact with the substrate. Image courtesy Wang et al, from Nature Chemistry |
Abstract:
When you look at a gift-wrapped present, the basic properties of the wrapping paper — say, its colors and texture — are not generally changed by the nature of the gift inside.
Graphene’s behavior depends on where it sits: New findings show that the material beneath the thin carbon sheets determines how they react chemically and electrically
Cambridge, MA | Posted on August 14th, 2012But surprising new experiments conducted at MIT show that a one-atom-thick material called graphene, a form of pure carbon whose atoms are joined in a chicken-wire-like lattice, behaves quite differently depending on the nature of material it's wrapped around. When sheets of graphene are placed on substrates made of different materials, fundamental properties — such as how the graphene conducts electricity and how it interacts chemically with other materials — can be drastically different, depending on the nature of the underlying material.
"We were quite surprised" to discover this altered behavior, says Michael Strano, the Charles and Hilda Roddey Professor of Chemical Engineering at MIT, who is the senior author of a paper published this week in the journal Nature Chemistry. "We expected it to behave like graphite" — a well-known form of carbon, used to make the lead in pencils, whose structure is essentially multiple layers of graphene piled on top of each other.
But its behavior turned out to be quite different. "Graphene is very strange," Strano says. Because of its extreme thinness, in practice graphene is almost always placed on top of some other material for support. When that material underneath is silicon dioxide, a standard material used in electronics, the graphene can readily become "functionalized" when exposed to certain chemicals. But when graphene sits on boron nitride, it hardly reacts at all to the same chemicals.
"It's very counterintuitive," Strano says. "You can turn off and turn on graphene's ability to form chemical bonds, based on what's underneath."
The reason, it turns out, is that the material is so thin that the way it reacts is strongly affected by the electrical fields of atoms in the material beneath it. This means that it is possible to create devices with a micropatterned substrate — made up of some silicon dioxide regions and some coated with boron nitride — covered with a layer of graphene whose chemical behavior will then vary according to the hidden patterning. This could enable, for example, the production of microarrays of sensors to detect trace biological or chemical materials.
Qing Hua Wang, an MIT postdoc who is the lead author of the paper, says, "You could get different molecules of a delicate biological marker to interact [with these regions on the graphene surface] without disrupting the biomolecules themselves." Most current fabrication techniques for such patterned surfaces involve heat and reactive solvents that can destroy these sensitive biological molecules.
Ultimately, graphene could even become a protective coating for many materials, Strano says. For example, the one-atom-thick material, when bonded to copper, completely eliminates that metal's tendency to oxidize (which produces the characteristic blue-green surface of copper roofs). "It can completely turn off the corrosion," he says, "almost like magic … with just the whisper of a coating."
To explain why graphene behaves the way it does, "we came up with a new electron-transfer theory" that accounts for the way it is affected by the underlying material, Strano says. "A lot of chemists had missed this," and as a result had been confused by seemingly unpredictable changes in how graphene reacts in different situations. This new understanding can also be used to predict the material's behavior on other substrates, he says.
James Tour, a professor of chemistry and of computer science at Rice University who was not involved in this research, says, "This is the first systematic study of the substrate's effect on graphene's chemical reactivity. This is a very carefully conducted study with convincing results. I predict that it will become a frequently cited publication."
Wang adds that "it's a pretty general result" that can be used to predict the chemical behavior of many different configurations. "We think other groups can take this idea and really develop different things with it," she says. Tour agrees, saying, "The graphene-sensing community will be inspired by this work to explore many more substrates in an effort to optimize graphene reactivity."
As for the MIT team, she says, "the next step is, we're digging into the details of how bilayer graphene reacts. It seems to behave differently" than the single-layer material.
The work was primarily supported by the U.S. Office of Naval Research.
####
For more information, please click here
Copyright © MIT
If you have a comment, please Contact us.Issuers of news releases, not 7th Wave, Inc. or Nanotechnology Now, are solely responsible for the accuracy of the content.
| Related News Press |
News and information
![]() Simulating magnetization in a Heisenberg quantum spin chain April 5th, 2024
Simulating magnetization in a Heisenberg quantum spin chain April 5th, 2024
![]() NRL charters Navy’s quantum inertial navigation path to reduce drift April 5th, 2024
NRL charters Navy’s quantum inertial navigation path to reduce drift April 5th, 2024
![]() Discovery points path to flash-like memory for storing qubits: Rice find could hasten development of nonvolatile quantum memory April 5th, 2024
Discovery points path to flash-like memory for storing qubits: Rice find could hasten development of nonvolatile quantum memory April 5th, 2024
Graphene/ Graphite
![]() NRL discovers two-dimensional waveguides February 16th, 2024
NRL discovers two-dimensional waveguides February 16th, 2024
Govt.-Legislation/Regulation/Funding/Policy
![]() NRL charters Navy’s quantum inertial navigation path to reduce drift April 5th, 2024
NRL charters Navy’s quantum inertial navigation path to reduce drift April 5th, 2024
![]() Discovery points path to flash-like memory for storing qubits: Rice find could hasten development of nonvolatile quantum memory April 5th, 2024
Discovery points path to flash-like memory for storing qubits: Rice find could hasten development of nonvolatile quantum memory April 5th, 2024
![]() Chemical reactions can scramble quantum information as well as black holes April 5th, 2024
Chemical reactions can scramble quantum information as well as black holes April 5th, 2024
Discoveries
![]() Chemical reactions can scramble quantum information as well as black holes April 5th, 2024
Chemical reactions can scramble quantum information as well as black holes April 5th, 2024
![]() New micromaterial releases nanoparticles that selectively destroy cancer cells April 5th, 2024
New micromaterial releases nanoparticles that selectively destroy cancer cells April 5th, 2024
![]() Utilizing palladium for addressing contact issues of buried oxide thin film transistors April 5th, 2024
Utilizing palladium for addressing contact issues of buried oxide thin film transistors April 5th, 2024
Announcements
![]() NRL charters Navy’s quantum inertial navigation path to reduce drift April 5th, 2024
NRL charters Navy’s quantum inertial navigation path to reduce drift April 5th, 2024
![]() Discovery points path to flash-like memory for storing qubits: Rice find could hasten development of nonvolatile quantum memory April 5th, 2024
Discovery points path to flash-like memory for storing qubits: Rice find could hasten development of nonvolatile quantum memory April 5th, 2024
Military
![]() NRL charters Navy’s quantum inertial navigation path to reduce drift April 5th, 2024
NRL charters Navy’s quantum inertial navigation path to reduce drift April 5th, 2024
![]() What heat can tell us about battery chemistry: using the Peltier effect to study lithium-ion cells March 8th, 2024
What heat can tell us about battery chemistry: using the Peltier effect to study lithium-ion cells March 8th, 2024
![]() New chip opens door to AI computing at light speed February 16th, 2024
New chip opens door to AI computing at light speed February 16th, 2024
|
|
||
|
|
||
| The latest news from around the world, FREE | ||
|
|
||
|
|
||
| Premium Products | ||
|
|
||
|
Only the news you want to read!
Learn More |
||
|
|
||
|
Full-service, expert consulting
Learn More |
||
|
|
||








