Home > Press > Study Improves Understanding of Surface Molecules in Controlling Size of Gold Nanoparticles
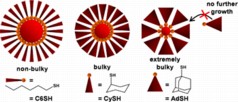 |
| The bulkier the ligand, the fewer ligands can sit side-by-side -- leading to a smaller nanoparticle. |
Abstract:
Use of bulky ligands (BLs) in the synthesis of metal nanoparticles (NPs) gives smaller core sizes, sharpens the size distribution, and alters the discrete sizes. For BLs, the highly curved surface of small NPs may facilitate growth, but as the size increases and the surface flattens, NP growth may terminate when the ligand monolayer blocks BLs from transporting metal atoms to the NP core. Batches of thiolate-stabilized Au NPs were synthesized using equimolar amounts of 1-adamantanethiol (AdSH), cyclohexanethiol (CySH), or n-hexanethiol (C6SH). The bulky CyS- and AdS-stabilized NPs have smaller, more monodisperse sizes than the C6S-stabilized NPs. As the bulkiness increases, the near-infrared luminescence intensity increases, which is characteristic of small Au NPs. Four new discrete sizes were measured by MALDI-TOF mass spectrometry, Au30(SAd)18, Au39(SAd)23, Au65(SCy)30, and Au67(SCy)30. No Au25(SAd)18 was observed, which suggests that this structure would be too sterically crowded. Use of BLs may also lead to the discovery of new discrete sizes in other systems.
Study Improves Understanding of Surface Molecules in Controlling Size of Gold Nanoparticles
Raleigh, NC | Posted on June 18th, 2012North Carolina State University researchers have shown that the "bulkiness" of molecules commonly used in the creation of gold nanoparticles actually dictates the size of the nanoparticles - with larger so-called ligands resulting in smaller nanoparticles. The research team also found that each type of ligand produces nanoparticles in a particular array of discrete sizes.
"This work advances our understanding of nanoparticle formation, and gives us a new tool for controlling the size and characteristics of gold nanoparticles," says Dr. Joseph Tracy, an assistant professor of materials science and engineering at NC State and co-author of a paper describing the research. Gold nanoparticles are used in industrial chemical processes, as well as medical and electronics applications.
When creating gold nanoparticles, scientists often use organic molecules called ligands to facilitate the process. The ligands effectively bring gold atoms together in a solution to create the nanoparticles. In the process, ligands essentially line up side by side and surround the nanoparticles in all three dimensions.
The researchers wanted to see whether the bulkiness of the ligands affected nanoparticle size, and opted to assess three types of thiol ligands - a family of ligands commonly used to synthesize gold nanoparticles. Specifically, the molecules bound to the gold nanoparticles are linear hexanethiolate (-SC6), cyclohexanethiolate (-SCy) and 1-adamantanethiolate (-SAd). Each of these ligands has a bulkier configuration than the last.
For example, picture each ligand as a slice of pie, with a gold atom attached to the pointed end. -SC6 looks like a very narrow slice of pie. -SCy is slightly larger, and -SAd is the largest of the three - with the "crust" end of the pie wedge far wider than the pointed end.
The researchers found that the bulkiness of the ligands determined the size of the nanoparticles. Because fewer -SAd and -SCy ligands can line up next to each other in three dimensions, fewer gold atoms are brought together in the core. Therefore, the nanoparticles are smaller. -SC6, the least bulky of the thiolates, can create the largest nanoparticles.
"While we've shown that this is an effective means of controlling size in gold nanoparticles, we think it may have implications for other materials as well," says Peter Krommenhoek, a Ph.D. student at NC State and lead author of the paper. "That's something we're exploring."
But the researchers made another interesting finding as well.
When particularly small nanoparticles form, they tend to form at very specific sizes, called discrete sizes. For instance, some types of nanoparticles may consist of 25 or 28 atoms - but never 26 or 27 atoms.
In this study, the researchers found that the bulkiness of the ligands also changed the discrete sizes of the nanoparticles. "This is interesting, in part, because each discrete size represents a different number of gold atoms and ligands," Tracy says, "which could influence the nanoparticle's chemical behavior. That question has yet to be addressed."
The paper, "Bulky Adamantanethiolate and Cyclohexanethiolate Ligands Favor Smaller Gold Nanoparticles with Altered Discrete Sizes," was published online June 15 in ACS Nano. The paper was co-authored by Dr. Junwei Wang, a former postdoctoral research associate at NC State; Dr. Nathaniel Hentz, of the Golden LEAF Biomanufacturing Training and Education Center at NC State; Krystian Kozek, a former undergraduate at NC State; Dr. Aaron Johnston-Peck, a postdoctoral researcher at Brookhaven National Laboratory; and Dr. Gregory Kalyuzhny of San Diego State University.
The research was supported by the National Science Foundation and the U.S. Department of Education.
####
For more information, please click here
Contacts:
Matt Shipman
News Services
919.515.6386
Dr. Joe Tracy
919.513.2623
Copyright © North Carolina State University
If you have a comment, please Contact us.Issuers of news releases, not 7th Wave, Inc. or Nanotechnology Now, are solely responsible for the accuracy of the content.
| Related Links |
| Related News Press |
News and information
![]() Simulating magnetization in a Heisenberg quantum spin chain April 5th, 2024
Simulating magnetization in a Heisenberg quantum spin chain April 5th, 2024
![]() NRL charters Navy�s quantum inertial navigation path to reduce drift April 5th, 2024
NRL charters Navy�s quantum inertial navigation path to reduce drift April 5th, 2024
![]() Discovery points path to flash-like memory for storing qubits: Rice find could hasten development of nonvolatile quantum memory April 5th, 2024
Discovery points path to flash-like memory for storing qubits: Rice find could hasten development of nonvolatile quantum memory April 5th, 2024
Chemistry
![]() What heat can tell us about battery chemistry: using the Peltier effect to study lithium-ion cells March 8th, 2024
What heat can tell us about battery chemistry: using the Peltier effect to study lithium-ion cells March 8th, 2024
![]() Nanoscale CL thermometry with lanthanide-doped heavy-metal oxide in TEM March 8th, 2024
Nanoscale CL thermometry with lanthanide-doped heavy-metal oxide in TEM March 8th, 2024
Govt.-Legislation/Regulation/Funding/Policy
![]() NRL charters Navy�s quantum inertial navigation path to reduce drift April 5th, 2024
NRL charters Navy�s quantum inertial navigation path to reduce drift April 5th, 2024
![]() Discovery points path to flash-like memory for storing qubits: Rice find could hasten development of nonvolatile quantum memory April 5th, 2024
Discovery points path to flash-like memory for storing qubits: Rice find could hasten development of nonvolatile quantum memory April 5th, 2024
![]() Chemical reactions can scramble quantum information as well as black holes April 5th, 2024
Chemical reactions can scramble quantum information as well as black holes April 5th, 2024
Chip Technology
![]() Discovery points path to flash-like memory for storing qubits: Rice find could hasten development of nonvolatile quantum memory April 5th, 2024
Discovery points path to flash-like memory for storing qubits: Rice find could hasten development of nonvolatile quantum memory April 5th, 2024
![]() Utilizing palladium for addressing contact issues of buried oxide thin film transistors April 5th, 2024
Utilizing palladium for addressing contact issues of buried oxide thin film transistors April 5th, 2024
![]() HKUST researchers develop new integration technique for efficient coupling of III-V and silicon February 16th, 2024
HKUST researchers develop new integration technique for efficient coupling of III-V and silicon February 16th, 2024
Nanomedicine
![]() New micromaterial releases nanoparticles that selectively destroy cancer cells April 5th, 2024
New micromaterial releases nanoparticles that selectively destroy cancer cells April 5th, 2024
![]() Good as gold - improving infectious disease testing with gold nanoparticles April 5th, 2024
Good as gold - improving infectious disease testing with gold nanoparticles April 5th, 2024
![]() Researchers develop artificial building blocks of life March 8th, 2024
Researchers develop artificial building blocks of life March 8th, 2024
Discoveries
![]() Chemical reactions can scramble quantum information as well as black holes April 5th, 2024
Chemical reactions can scramble quantum information as well as black holes April 5th, 2024
![]() New micromaterial releases nanoparticles that selectively destroy cancer cells April 5th, 2024
New micromaterial releases nanoparticles that selectively destroy cancer cells April 5th, 2024
![]() Utilizing palladium for addressing contact issues of buried oxide thin film transistors April 5th, 2024
Utilizing palladium for addressing contact issues of buried oxide thin film transistors April 5th, 2024
Announcements
![]() NRL charters Navy�s quantum inertial navigation path to reduce drift April 5th, 2024
NRL charters Navy�s quantum inertial navigation path to reduce drift April 5th, 2024
![]() Discovery points path to flash-like memory for storing qubits: Rice find could hasten development of nonvolatile quantum memory April 5th, 2024
Discovery points path to flash-like memory for storing qubits: Rice find could hasten development of nonvolatile quantum memory April 5th, 2024
|
|
||
|
|
||
| The latest news from around the world, FREE | ||
|
|
||
|
|
||
| Premium Products | ||
|
|
||
|
Only the news you want to read!
Learn More |
||
|
|
||
|
Full-service, expert consulting
Learn More |
||
|
|
||








