Home > Press > Researchers watch tiny living machines self-assemble
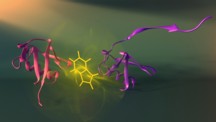 |
| Vall�e-B�lisle and Michnick have developed a new approach to visualize how proteins assemble, which may also significantly aid our understanding of diseases such as Alzheimer�s and Parkinson�s, which are caused by errors in assembly. Here shown are two different assembly stages (purple and red) of the protein ubiquitin and the fluorescent probe used to visualize these stage (tryptophan: see yellow). Credit: Peter Allen. Print resolution available on request. |
Abstract:
Enabling bioengineers to design new molecular machines for nanotechnology applications is one of the possible outcomes of a study by University of Montreal researchers that was published in Nature Structural and Molecular Biology today. The scientists have developed a new approach to visualize how proteins assemble, which may also significantly aid our understanding of diseases such as Alzheimer's and Parkinson's, which are caused by errors in assembly.
Researchers watch tiny living machines self-assemble
Montr�al, Canada | Posted on June 10th, 2012"In order to survive, all creatures, from bacteria to humans, monitor and transform their environments using small protein nanomachines made of thousands of atoms," explained the senior author of the study, Prof. Stephen Michnick of the university's department of biochemistry. "For example, in our sinuses, there are complex receptor proteins that are activated in the presence of different odor molecules. Some of those scents warn us of danger; others tell us that food is nearby." Proteins are made of long linear chains of amino acids, which have evolved over millions of years to self-assemble extremely rapidly - often within thousandths of a split second - into a working nanomachine. "One of the main challenges for biochemists is to understand how these linear chains assemble into their correct structure given an astronomically large number of other possible forms," Michnick said.
"To understand how a protein goes from a linear chain to a unique assembled structure, we need to capture snapshots of its shape at each stage of assembly said Dr. Alexis Vall�e-B�lisle, first author of the study. "The problem is that each step exists for a fleetingly short time and no available technique enables us to obtain precise structural information on these states within such a small time frame. We developed a strategy to monitor protein assembly by integrating fluorescent probes throughout the linear protein chain so that we could detect the structure of each stage of protein assembly, step by step to its final structure." The protein assembly process is not the end of its journey, as a protein can change, through chemical modifications or with age, to take on different forms and functions. "Understanding how a protein goes from being one thing to becoming another is the first step towards understanding and designing protein nanomachines for biotechnologies such as medical and environmental diagnostic sensors, drug synthesis of delivery," Vall�e-B�lisle said.
This research was supported by the Natural Sciences and Engineering Research Council of Canada and Le fond de recherch� du Qu�bec, Nature et Technologie. The article, "Visualizing transient protein folding intermediates by tryptophan scanning mutagenesis," published in Nature Structural & Molecular Biology, was coauthored by Alexis Vall�e-B�lisle and Stephen W. Michnick of the D�partement de Biochimie de l'Universit� de Montr�al. The University of Montreal is known officially as Universit� de Montr�al.
Full bibliographic information"Visualizing transient protein-folding intermediates by tryptophan-scanning mutagenesis" by Michnick has been scheduled for Advance Online Publication (AOP) on Nature Structural & Molecular Biology's website on 10 June at 1800 London time / 1300 US Eastern time, which is when the embargo will lift. If you wish to see the paper, the author(s) should be able to provide you with a copy.
The full listing of authors and their affiliations for this paper is as follows:
Alexis Vall�e-B�lisle1,3 & Stephen W Michnick1,2
1D�partement de Biochimie, Universit� de Montr�al, Montr�al, Qu�bec, Canada.
2Centre Robert-Cedergren en Bio-Informatique et G�nomique, Universit� de Montr�al, Montr�al, Qu�bec, Canada.
3Present address: Department of Chemistry and Biochemistry, University of California, Santa Barbara, California, USA.
####
About Universit� de Montr�al
Deeply rooted in Montreal and dedicated to its international mission, the Universit� de Montr�al (University of Montreal) is one of the top universities in the French-speaking world. Founded in 1878, the University today has 16 faculties and together with its two affiliated schools, HEC Montr�al and �cole Polytechnique, constitutes the largest centre of higher education and research in Qu�bec, the second largest in Canada, and one of the major centres in North America. It brings together 2,500 professors and researchers, accommodates more than 60,000 students, offers some 650 programs at all academic levels, and awards about 3,000 masters and doctorate diplomas each year.
For more information, please click here
Contacts:
William Raillant-Clark
514-343-7593
Copyright © AlphaGalileo
If you have a comment, please Contact us.Issuers of news releases, not 7th Wave, Inc. or Nanotechnology Now, are solely responsible for the accuracy of the content.
| Related News Press |
News and information
![]() Simulating magnetization in a Heisenberg quantum spin chain April 5th, 2024
Simulating magnetization in a Heisenberg quantum spin chain April 5th, 2024
![]() NRL charters Navy�s quantum inertial navigation path to reduce drift April 5th, 2024
NRL charters Navy�s quantum inertial navigation path to reduce drift April 5th, 2024
![]() Discovery points path to flash-like memory for storing qubits: Rice find could hasten development of nonvolatile quantum memory April 5th, 2024
Discovery points path to flash-like memory for storing qubits: Rice find could hasten development of nonvolatile quantum memory April 5th, 2024
Molecular Machines
![]() First electric nanomotor made from DNA material: Synthetic rotary motors at the nanoscale perform mechanical work July 22nd, 2022
First electric nanomotor made from DNA material: Synthetic rotary motors at the nanoscale perform mechanical work July 22nd, 2022
![]() Nanotech scientists create world's smallest origami bird March 17th, 2021
Nanotech scientists create world's smallest origami bird March 17th, 2021
![]() Giant nanomachine aids the immune system: Theoretical chemistry August 28th, 2020
Giant nanomachine aids the immune system: Theoretical chemistry August 28th, 2020
Molecular Nanotechnology
![]() Scientists push the boundaries of manipulating light at the submicroscopic level March 3rd, 2023
Scientists push the boundaries of manipulating light at the submicroscopic level March 3rd, 2023
![]() First electric nanomotor made from DNA material: Synthetic rotary motors at the nanoscale perform mechanical work July 22nd, 2022
First electric nanomotor made from DNA material: Synthetic rotary motors at the nanoscale perform mechanical work July 22nd, 2022
![]() Nanotech scientists create world's smallest origami bird March 17th, 2021
Nanotech scientists create world's smallest origami bird March 17th, 2021
Self Assembly
![]() Liquid crystal templated chiral nanomaterials October 14th, 2022
Liquid crystal templated chiral nanomaterials October 14th, 2022
![]() Nanoclusters self-organize into centimeter-scale hierarchical assemblies April 22nd, 2022
Nanoclusters self-organize into centimeter-scale hierarchical assemblies April 22nd, 2022
![]() Atom by atom: building precise smaller nanoparticles with templates March 4th, 2022
Atom by atom: building precise smaller nanoparticles with templates March 4th, 2022
![]() Nanostructures get complex with electron equivalents: Nanoparticles of two different sizes break away from symmetrical designs January 14th, 2022
Nanostructures get complex with electron equivalents: Nanoparticles of two different sizes break away from symmetrical designs January 14th, 2022
Nanomedicine
![]() New micromaterial releases nanoparticles that selectively destroy cancer cells April 5th, 2024
New micromaterial releases nanoparticles that selectively destroy cancer cells April 5th, 2024
![]() Good as gold - improving infectious disease testing with gold nanoparticles April 5th, 2024
Good as gold - improving infectious disease testing with gold nanoparticles April 5th, 2024
![]() Researchers develop artificial building blocks of life March 8th, 2024
Researchers develop artificial building blocks of life March 8th, 2024
Discoveries
![]() Chemical reactions can scramble quantum information as well as black holes April 5th, 2024
Chemical reactions can scramble quantum information as well as black holes April 5th, 2024
![]() New micromaterial releases nanoparticles that selectively destroy cancer cells April 5th, 2024
New micromaterial releases nanoparticles that selectively destroy cancer cells April 5th, 2024
![]() Utilizing palladium for addressing contact issues of buried oxide thin film transistors April 5th, 2024
Utilizing palladium for addressing contact issues of buried oxide thin film transistors April 5th, 2024
|
|
||
|
|
||
| The latest news from around the world, FREE | ||
|
|
||
|
|
||
| Premium Products | ||
|
|
||
|
Only the news you want to read!
Learn More |
||
|
|
||
|
Full-service, expert consulting
Learn More |
||
|
|
||








