Home > Press > To make better fuel cells, study the defects
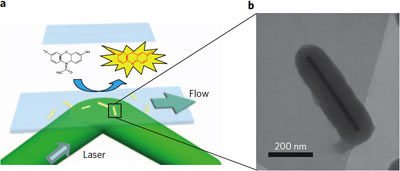 |
| Provided/Chen Lab When Amplex Red connects with a gold catalyst the structure is changed to make a fluorescent molecule that immediately emits a flash of light, showing where the catalytic event took place. Right, electron microphoto of a single gold nanorod, encased in a poirus silica shell. The shell keeps rods from clumping together and allows experimenters to use heat to clean away a coating that forms when the rods are created. |
Abstract:
Engineers trying to improve fuel-cell catalysts may be looking in the
wrong place, according to new research at Cornell.
To make better fuel cells, study the defects
Ithaca, NY | Posted on February 20th, 2012There is growing interest in forming the catalysts that break down fuel
to generate electricity into nanoparticles. Nanoparticles provide a
larger surface area to speed reactions, and in some cases, materials
that are not catalytic in bulk become so at the nanoscale.
These nanoparticles, typically just a few tens of nanometers (nm) wide,
are not neat little spheres, but rather jagged chunks, like microscale
gravel, and researchers have found that they can correlate catalytic
activity with information about the number and type of their surface
facets. But they may be looking at the forest and ignoring the trees.
"People measure the activity of a sample and then try to understand by
using facet information," said Peng Chen, associate professor of
chemistry and chemical biology. "The message we want to deliver is that
surface defects [on the facets] dominate the catalysis."
Chen's research is reported Feb. 19 in the online edition of the
journal Nature Nanotechnology.
Instead of particles, Chen's research group studied catalytic events on
gold "nanorods" up to 700 nm long, effectively letting them see how
activity varies over a single facet. Gold acts as a catalyst to convert
a chemical called Amplex Red into resorufin, which is fluorescent.
Each time a catalytic event occurs, the newly created molecule of
resorufin emits a flash of light that is detected by a digital camera
looking through a microscope. A flash typically appears as several
pixels, and additional computer processing averages their brightness to
pinpoint the actual event to within a few nanometers. The researchers
call the technique "super-resolution microscopy." After flooding a
field of nanorods with a solution of Amplex Red, they made a "movie"
with one frame every 25 milliseconds.
The researchers found more catalytic events near the middle of a rod,
tapering off toward the ends and a jump back up at the ends. They also
found variation in the amount of activity from one rod to another, even
though all the rods have the same types of facets.
To explain the results, they proposed that activity is higher in areas
where there are more surface defects. The nanorods are made by growing
gold crystals from a small "seed" crystal, growing outward from the
center to the ends, Chen explained, and more defects form at the
beginning of the process.
"Knowledge of the surface facets ... is insufficient to predict
reactivity," the researchers said in their paper. "Surface defects �
can also play a dominant role."
The findings with a gold catalyst and fluorescent molecules should be
equally applicable to other catalysts, including those used in fuel
cells and for pollution remediation, Chen said.
The research was supported in part by the Army Research Office, the
National Science Foundation (NSF), the Department of Energy and the
Alfred P. Sloan Foundation. Part of the work was carried out at the
Cornell Center for Materials Research and the Cornell Nanoscale Science
and Technology Facility, both supported by NSF.
####
For more information, please click here
Contacts:
Bill Steele
(607) 255-7164
Copyright © Cornell University
If you have a comment, please Contact us.Issuers of news releases, not 7th Wave, Inc. or Nanotechnology Now, are solely responsible for the accuracy of the content.
| Related News Press |
News and information
![]() Quantum computer improves AI predictions April 17th, 2026
Quantum computer improves AI predictions April 17th, 2026
![]() Flexible sensor gains sensitivity under pressure April 17th, 2026
Flexible sensor gains sensitivity under pressure April 17th, 2026
![]() A reusable chip for particulate matter sensing April 17th, 2026
A reusable chip for particulate matter sensing April 17th, 2026
![]() Detecting vibrational quantum beating in the predissociation dynamics of SF6 using time-resolved photoelectron spectroscopy April 17th, 2026
Detecting vibrational quantum beating in the predissociation dynamics of SF6 using time-resolved photoelectron spectroscopy April 17th, 2026
Govt.-Legislation/Regulation/Funding/Policy
![]() Quantum computer improves AI predictions April 17th, 2026
Quantum computer improves AI predictions April 17th, 2026
![]() Metasurfaces smooth light to boost magnetic sensing precision January 30th, 2026
Metasurfaces smooth light to boost magnetic sensing precision January 30th, 2026
![]() New imaging approach transforms study of bacterial biofilms August 8th, 2025
New imaging approach transforms study of bacterial biofilms August 8th, 2025
Discoveries
![]() Quantum computer improves AI predictions April 17th, 2026
Quantum computer improves AI predictions April 17th, 2026
![]() Flexible sensor gains sensitivity under pressure April 17th, 2026
Flexible sensor gains sensitivity under pressure April 17th, 2026
![]() A reusable chip for particulate matter sensing April 17th, 2026
A reusable chip for particulate matter sensing April 17th, 2026
![]() Detecting vibrational quantum beating in the predissociation dynamics of SF6 using time-resolved photoelectron spectroscopy April 17th, 2026
Detecting vibrational quantum beating in the predissociation dynamics of SF6 using time-resolved photoelectron spectroscopy April 17th, 2026
Announcements
![]() A fundamentally new therapeutic approach to cystic fibrosis: Nanobody repairs cellular defect April 17th, 2026
A fundamentally new therapeutic approach to cystic fibrosis: Nanobody repairs cellular defect April 17th, 2026
![]() UC Irvine physicists discover method to reverse �quantum scrambling� : The work addresses the problem of information loss in quantum computing system April 17th, 2026
UC Irvine physicists discover method to reverse �quantum scrambling� : The work addresses the problem of information loss in quantum computing system April 17th, 2026
Military
![]() Quantum engineers �squeeze� laser frequency combs to make more sensitive gas sensors January 17th, 2025
Quantum engineers �squeeze� laser frequency combs to make more sensitive gas sensors January 17th, 2025
![]() Chainmail-like material could be the future of armor: First 2D mechanically interlocked polymer exhibits exceptional flexibility and strength January 17th, 2025
Chainmail-like material could be the future of armor: First 2D mechanically interlocked polymer exhibits exceptional flexibility and strength January 17th, 2025
![]() Single atoms show their true color July 5th, 2024
Single atoms show their true color July 5th, 2024
![]() NRL charters Navy�s quantum inertial navigation path to reduce drift April 5th, 2024
NRL charters Navy�s quantum inertial navigation path to reduce drift April 5th, 2024
Fuel Cells
![]() Deciphering local microstrain-induced optimization of asymmetric Fe single atomic sites for efficient oxygen reduction August 8th, 2025
Deciphering local microstrain-induced optimization of asymmetric Fe single atomic sites for efficient oxygen reduction August 8th, 2025
![]() Current and Future Developments in Nanomaterials and Carbon Nanotubes: Applications of Nanomaterials in Energy Storage and Electronics October 28th, 2022
Current and Future Developments in Nanomaterials and Carbon Nanotubes: Applications of Nanomaterials in Energy Storage and Electronics October 28th, 2022
|
|
||
|
|
||
| The latest news from around the world, FREE | ||
|
|
||
|
|
||
| Premium Products | ||
|
|
||
|
Only the news you want to read!
Learn More |
||
|
|
||
|
Full-service, expert consulting
Learn More |
||
|
|
||








