Home > Press > Mn-doped ZnS is unsuitable to act as a dilute magnetic semiconductor
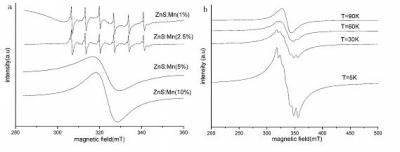 |
| This shows (a) Room temperature ESR spectra of ZnS:Mn; and (b) Low temperature ESR spectra of ZnS:Mn (20%).
Credit: �Science China Press |
Abstract:
Dilute magnetic semiconductors (DMS) have recently been a major focus of magnetic semiconductor research. A laboratory from the University of Science and Technology of China explored the feasibility of doping manganese (Mn) into zinc sulfide (ZnS) to obtain magnetic semiconductors.
Mn-doped ZnS is unsuitable to act as a dilute magnetic semiconductor
Hefei, PR China | Posted on February 20th, 2012Hideo Ohno and his group at the Tohoku University, Japan, were the first to measure ferromagnetism in transition metal-doped semiconductors such as indium arsenide and gallium arsenide doped with Mn. Ever since, researchers have attempted to obtain semiconductor hosts doped with different transition metals that exhibit ferromagnetic properties.
A team of researchers from Hefei National Laboratory for Physical Sciences at the Microscale, University of Science and Technology of China, discovered that Mn-doped ZnS (ZnS:Mn) shows paramagnetic behavior and is not suitable for use as a DMS. Their work entitled "Structure Characterization, Magnetic and Photoluminescence Properties of Mn-Doped ZnS Nanocrystalline" was published in SCIENCE CHINA Physics, Mechanics & Astronomy, 2012, Vol 55(2) .
Electron spin resonance (ESR) spectra (Figure 1a) of nanocrystalline ZnS:Mn show that at lower concentrations of Mn, a typical sextet centered at a g-value of 2 is associated with the allowed (Δms=�1, ΔmI=0) magnetic dipole transitions between the hyperfine-split Zeeman levels of the 6S5/2 ground state of the Mn2+ 3d electrons. The hyperfine structure arises from the interaction between the S=5/2 spin of the unpaired 3d electrons with I= 5/2 spin of the 55Mn nucleus. This indicates that Mn ions are distributed in the ZnS nanocrystalline lattice so that they are isolated from each other. At higher concentrations of Mn, the ions assemble together and are localized in the ZnS crystal lattice, decreasing the Mn-Mn atomic distance and increasing the dipole-dipole interaction. This causes the hyperfine structure to merge into one broad resonance. Further ESR experiments (Figure 1b) at low temperature also suggested that the sample was not ferromagnetic. All of the results indicated that ZnS:Mn is paramagnetic and not suitable for DMS.
See the article:
Zuo M, Tan S, Li G P, et al.Structure characterization,magnetic and photoluminescence properties of Mn doped ZnS nanocrystalline. SCIENCE CHINA Physics, Mechanics & Astronomy,2012,55: 219-223
####
For more information, please click here
Contacts:
Zhang Shuyuan
86-055-136-01503
Copyright © Science in China Press
If you have a comment, please Contact us.Issuers of news releases, not 7th Wave, Inc. or Nanotechnology Now, are solely responsible for the accuracy of the content.
| Related News Press |
News and information
![]() Quantum computer improves AI predictions April 17th, 2026
Quantum computer improves AI predictions April 17th, 2026
![]() Flexible sensor gains sensitivity under pressure April 17th, 2026
Flexible sensor gains sensitivity under pressure April 17th, 2026
![]() A reusable chip for particulate matter sensing April 17th, 2026
A reusable chip for particulate matter sensing April 17th, 2026
![]() Detecting vibrational quantum beating in the predissociation dynamics of SF6 using time-resolved photoelectron spectroscopy April 17th, 2026
Detecting vibrational quantum beating in the predissociation dynamics of SF6 using time-resolved photoelectron spectroscopy April 17th, 2026
Chip Technology
![]() A reusable chip for particulate matter sensing April 17th, 2026
A reusable chip for particulate matter sensing April 17th, 2026
![]() Metasurfaces smooth light to boost magnetic sensing precision January 30th, 2026
Metasurfaces smooth light to boost magnetic sensing precision January 30th, 2026
Nanoelectronics
![]() Lab to industry: InSe wafer-scale breakthrough for future electronics August 8th, 2025
Lab to industry: InSe wafer-scale breakthrough for future electronics August 8th, 2025
![]() Interdisciplinary: Rice team tackles the future of semiconductors Multiferroics could be the key to ultralow-energy computing October 6th, 2023
Interdisciplinary: Rice team tackles the future of semiconductors Multiferroics could be the key to ultralow-energy computing October 6th, 2023
![]() Key element for a scalable quantum computer: Physicists from Forschungszentrum J�lich and RWTH Aachen University demonstrate electron transport on a quantum chip September 23rd, 2022
Key element for a scalable quantum computer: Physicists from Forschungszentrum J�lich and RWTH Aachen University demonstrate electron transport on a quantum chip September 23rd, 2022
![]() Reduced power consumption in semiconductor devices September 23rd, 2022
Reduced power consumption in semiconductor devices September 23rd, 2022
Discoveries
![]() Quantum computer improves AI predictions April 17th, 2026
Quantum computer improves AI predictions April 17th, 2026
![]() Flexible sensor gains sensitivity under pressure April 17th, 2026
Flexible sensor gains sensitivity under pressure April 17th, 2026
![]() A reusable chip for particulate matter sensing April 17th, 2026
A reusable chip for particulate matter sensing April 17th, 2026
![]() Detecting vibrational quantum beating in the predissociation dynamics of SF6 using time-resolved photoelectron spectroscopy April 17th, 2026
Detecting vibrational quantum beating in the predissociation dynamics of SF6 using time-resolved photoelectron spectroscopy April 17th, 2026
Announcements
![]() A fundamentally new therapeutic approach to cystic fibrosis: Nanobody repairs cellular defect April 17th, 2026
A fundamentally new therapeutic approach to cystic fibrosis: Nanobody repairs cellular defect April 17th, 2026
![]() UC Irvine physicists discover method to reverse �quantum scrambling� : The work addresses the problem of information loss in quantum computing system April 17th, 2026
UC Irvine physicists discover method to reverse �quantum scrambling� : The work addresses the problem of information loss in quantum computing system April 17th, 2026
Research partnerships
![]() Lab to industry: InSe wafer-scale breakthrough for future electronics August 8th, 2025
Lab to industry: InSe wafer-scale breakthrough for future electronics August 8th, 2025
![]() HKU physicists uncover hidden order in the quantum world through deconfined quantum critical points April 25th, 2025
HKU physicists uncover hidden order in the quantum world through deconfined quantum critical points April 25th, 2025
|
|
||
|
|
||
| The latest news from around the world, FREE | ||
|
|
||
|
|
||
| Premium Products | ||
|
|
||
|
Only the news you want to read!
Learn More |
||
|
|
||
|
Full-service, expert consulting
Learn More |
||
|
|
||








