Home > Press > Highly Active Enzymes on Graphene Oxide Surface
 |
Abstract:
Chemically reduced graphene oxide is a good substrate for high enzyme loads, and activities comparable with those found in solution can be achieved on this surface, as shown by Chinese scientists.
Highly Active Enzymes on Graphene Oxide Surface
Germany | Posted on November 10th, 2011Graphene oxide (GO) is an interesting substrate for absorption and immobilization of biological compounds because of its stability, low toxicity, and unique chemical, electrical, and mechanical properties. It has been used in biomolecule detection and as a drug carrier, and more recently as a substrate on which to immobilize enzymes for catalytic use. However until now, such attachment has had to be done either covalently, or via electrostatic interactions between the enzyme and the GO surface. Covalent bonding may alter the properties of the enzyme by changing its shape, but the electrostatic interactions can also affect enzyme activity.
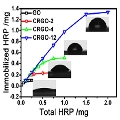
Scientists have started to experiment with a modified kind of GO called chemically reduced graphene oxide (CRGO) to overcome this problem of attachment. CRGO has fewer surface functional groups than GO, so should perturb the enzyme on it less. Now, a team led by Jingyan Zhang at East China University of Science and Technology and Shouwu Guo at Shanghai Jiao Tong University, China, has looked systematically at the loading and activity of enzymes on CRGO with varying degrees of reduction.
The team found that the more reduced the CRGO, the higher the possible loading of the enzyme, and that loadings of up to 60 times those possible on classically used materials were possible with CRGO. They found that the high loading was caused by hydrophobic interactions between the CRGO surface and the surface of the enzyme molecules, rather than the electrostatic interactions present with regular GO.
But when the team looked at the activity of the enzymes, they got quite a surprise. While one enzyme (horseradish peroxidase; HRP) was much less active than in solution, another (oxalate oxidase; OxOx) was nearly as active and retained up to 90% of its activity even after ten cycles of use, depending on the extent of CRGO reduction. Previous studies on immobilization of OxOx, e.g., on solid beads, have yielded much lower activities. The scientists attribute this finding to the particular structures of the two enzymes and how they bind to the surface. Some enzymes will change conformation on binding to the hydrophobic surface, which involves a dehydration step; this may affect the activity of the enzyme, while it appears that OxOx does not alter so much.
This new way to immobilize OxOx on CRGO while retaining high activity is very promising for clinical determinations of oxalate. The team believe that their method can be extended to use for other enzymes that retain their structure upon hydrophobic binding and dehydration, and that it has applications in detection and as a molecular carrier.
####
For more information, please click here
Copyright © Wiley-VCH Materials Science Journals
If you have a comment, please Contact us.Issuers of news releases, not 7th Wave, Inc. or Nanotechnology Now, are solely responsible for the accuracy of the content.
| Related Links |
![]() Y. Zhang et al., Small ; DOI: 10.1002/smll.201101695
Y. Zhang et al., Small ; DOI: 10.1002/smll.201101695
| Related News Press |
News and information
![]() Simulating magnetization in a Heisenberg quantum spin chain April 5th, 2024
Simulating magnetization in a Heisenberg quantum spin chain April 5th, 2024
![]() NRL charters Navy�s quantum inertial navigation path to reduce drift April 5th, 2024
NRL charters Navy�s quantum inertial navigation path to reduce drift April 5th, 2024
![]() Discovery points path to flash-like memory for storing qubits: Rice find could hasten development of nonvolatile quantum memory April 5th, 2024
Discovery points path to flash-like memory for storing qubits: Rice find could hasten development of nonvolatile quantum memory April 5th, 2024
Chemistry
![]() What heat can tell us about battery chemistry: using the Peltier effect to study lithium-ion cells March 8th, 2024
What heat can tell us about battery chemistry: using the Peltier effect to study lithium-ion cells March 8th, 2024
![]() Nanoscale CL thermometry with lanthanide-doped heavy-metal oxide in TEM March 8th, 2024
Nanoscale CL thermometry with lanthanide-doped heavy-metal oxide in TEM March 8th, 2024
Graphene/ Graphite
![]() NRL discovers two-dimensional waveguides February 16th, 2024
NRL discovers two-dimensional waveguides February 16th, 2024
Discoveries
![]() Chemical reactions can scramble quantum information as well as black holes April 5th, 2024
Chemical reactions can scramble quantum information as well as black holes April 5th, 2024
![]() New micromaterial releases nanoparticles that selectively destroy cancer cells April 5th, 2024
New micromaterial releases nanoparticles that selectively destroy cancer cells April 5th, 2024
![]() Utilizing palladium for addressing contact issues of buried oxide thin film transistors April 5th, 2024
Utilizing palladium for addressing contact issues of buried oxide thin film transistors April 5th, 2024
Announcements
![]() NRL charters Navy�s quantum inertial navigation path to reduce drift April 5th, 2024
NRL charters Navy�s quantum inertial navigation path to reduce drift April 5th, 2024
![]() Discovery points path to flash-like memory for storing qubits: Rice find could hasten development of nonvolatile quantum memory April 5th, 2024
Discovery points path to flash-like memory for storing qubits: Rice find could hasten development of nonvolatile quantum memory April 5th, 2024
|
|
||
|
|
||
| The latest news from around the world, FREE | ||
|
|
||
|
|
||
| Premium Products | ||
|
|
||
|
Only the news you want to read!
Learn More |
||
|
|
||
|
Full-service, expert consulting
Learn More |
||
|
|
||








