Home > Press > Rare Coupling of Magnetic and Electric Properties in a Single Material: New multiferroic mechanism could lead to next-generation memory and sensing devices
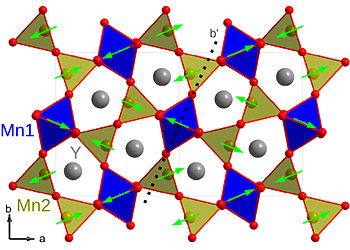 |
| The crystal structure of YMn2O5, which is made of yttrium, manganese, and oxygen. The oxygen atoms are shown in red and the yttrium atoms are gray. The magnetic moments on the manganese are shown as arrows. Ferroelectric polarization occurs between the oxygen and manganese atoms. |
Abstract:
Researchers at the U.S. Department of Energy's Brookhaven National Laboratory have observed a new way that magnetic and electric properties - which have a long history of ignoring and counteracting each other - can coexist in a special class of metals. These materials, known as multiferroics, could serve as the basis for the next generation of faster and energy-efficient logic, memory, and sensing technology.
Rare Coupling of Magnetic and Electric Properties in a Single Material: New multiferroic mechanism could lead to next-generation memory and sensing devices
Upton, NY | Posted on July 25th, 2011The researchers, who worked with colleagues at the Leibniz Institute for Solid State and Materials Research in Germany, published their findings online in Physical Review Letters on July 25, 2011.
Ferromagnets are materials that display a permanent magnetic moment, or magnetic direction, similar to how a compass needle always points north. They assist in a variety of daily tasks, from sticking a reminder to the fridge door to storing information on a computer's hard drive. Ferroelectrics are materials that display a permanent electric polarization - a set direction of charge - and respond to the application of an electric field by switching this direction. They are commonly used in applications like sonar, medical imaging, and sensors.
"In principle, the coupling of an ordered magnetic material with an ordered electric material could lead to very useful devices," said Brookhaven physicist Stuart Wilkins, one of the paper's authors. "For instance, one could imagine a device in which information is written by application of an electric field and read by detecting its magnetic state. This would make a faster and much more energy-efficient data storage device than is available today."
But multiferroics - magnetic materials with north and south poles that can be reversed with an electric field - are rare in nature. Ferroelectricity and magnetism tend to be mutually exclusive and interact weakly with each other when they coexist.
Most models used by physicists to describe this coupling are based on the idea of distorting the atomic arrangement, or crystal lattice, of a magnetic material, which can result in an electric polarization.
Now, scientists have found a new way that electric and magnetic properties can be coupled in a material. The group used extremely bright beams of x-rays at Brookhaven's National Synchrotron Light Source (NSLS) to examine the electronic structure of a particular metal oxide made of yttrium, manganese, and oxygen. They determined that the magnetic-electric coupling is caused by the outer cloud of electrons surrounding the atom.
"Previously, this mechanism had only been predicted theoretically and its existence was hotly debated," Wilkins said.
In this particular material, the manganese and oxygen electrons mix atomic orbitals in a process that creates atomic bonds and keeps the material together. The researchers' measurements show that this process is dependent upon the magnetic structure of the material, which in this case, causes the material to become ferroelectric, i.e. have an electric polarization. In other words, any change in the material's magnetic structure will result in a change in direction of its ferroelectric state. By definition, that makes the material a multiferroic.
"What is especially exciting is that this result proves the existence of a new coupling mechanism and provides a tool to study it," Wilkins said.
The researchers used a new instrument at NSLS designed to answer key questions about many intriguing classes of materials such as multiferroics and high-temperature superconductors, which conduct electricity without resistance. The instrument, developed by Wilkins and Brookhaven engineers D. Scott Coburn, William Leonhardt, and William Schoenig, will ultimately be moved to the National Synchrotron Light Source II (NSLS-II), a state-of-the-art machine currently under construction. NSLS-II will produce x-rays 10,000 times brighter than at NSLS, enabling studies of materials' properties at even higher resolution.
This work was supported by the U.S. Department of Energy Office of Science.
####
About Brookhaven National Laboratory
One of ten national laboratories overseen and primarily funded by the Office of Science of the U.S. Department of Energy (DOE), Brookhaven National Laboratory conducts research in the physical, biomedical, and environmental sciences, as well as in energy technologies and national security. Brookhaven Lab also builds and operates major scientific facilities available to university, industry and government researchers. Brookhaven is operated and managed for DOE's Office of Science by Brookhaven Science Associates, a limited-liability company founded by the Research Foundation of State University of New York on behalf of Stony Brook University, the largest academic user of Laboratory facilities, and Battelle, a nonprofit, applied science and technology organization.
For more information, please click here
Contacts:
Kendra Snyder
631-344-8191
or
Peter Genzer
631-344-3714
Copyright © Brookhaven National Laboratory
If you have a comment, please Contact us.Issuers of news releases, not 7th Wave, Inc. or Nanotechnology Now, are solely responsible for the accuracy of the content.
| Related Links |
![]() The scientific paper is available at:
The scientific paper is available at:
![]() Visit Brookhaven Lab's electronic newsroom for links, news archives, graphics, and more at:
Visit Brookhaven Lab's electronic newsroom for links, news archives, graphics, and more at:
![]() Follow Brookhaven Lab on Twitter:
Follow Brookhaven Lab on Twitter:
| Related News Press |
Physics
![]() Simulating magnetization in a Heisenberg quantum spin chain April 5th, 2024
Simulating magnetization in a Heisenberg quantum spin chain April 5th, 2024
![]() 'Sudden death' of quantum fluctuations defies current theories of superconductivity: Study challenges the conventional wisdom of superconducting quantum transitions January 12th, 2024
'Sudden death' of quantum fluctuations defies current theories of superconductivity: Study challenges the conventional wisdom of superconducting quantum transitions January 12th, 2024
News and information
![]() Simulating magnetization in a Heisenberg quantum spin chain April 5th, 2024
Simulating magnetization in a Heisenberg quantum spin chain April 5th, 2024
![]() NRL charters Navy�s quantum inertial navigation path to reduce drift April 5th, 2024
NRL charters Navy�s quantum inertial navigation path to reduce drift April 5th, 2024
![]() Discovery points path to flash-like memory for storing qubits: Rice find could hasten development of nonvolatile quantum memory April 5th, 2024
Discovery points path to flash-like memory for storing qubits: Rice find could hasten development of nonvolatile quantum memory April 5th, 2024
Superconductivity
![]() Optically trapped quantum droplets of light can bind together to form macroscopic complexes March 8th, 2024
Optically trapped quantum droplets of light can bind together to form macroscopic complexes March 8th, 2024
Laboratories
![]() A battery�s hopping ions remember where they�ve been: Seen in atomic detail, the seemingly smooth flow of ions through a battery�s electrolyte is surprisingly complicated February 16th, 2024
A battery�s hopping ions remember where they�ve been: Seen in atomic detail, the seemingly smooth flow of ions through a battery�s electrolyte is surprisingly complicated February 16th, 2024
![]() NRL discovers two-dimensional waveguides February 16th, 2024
NRL discovers two-dimensional waveguides February 16th, 2024
Govt.-Legislation/Regulation/Funding/Policy
![]() NRL charters Navy�s quantum inertial navigation path to reduce drift April 5th, 2024
NRL charters Navy�s quantum inertial navigation path to reduce drift April 5th, 2024
![]() Discovery points path to flash-like memory for storing qubits: Rice find could hasten development of nonvolatile quantum memory April 5th, 2024
Discovery points path to flash-like memory for storing qubits: Rice find could hasten development of nonvolatile quantum memory April 5th, 2024
![]() Chemical reactions can scramble quantum information as well as black holes April 5th, 2024
Chemical reactions can scramble quantum information as well as black holes April 5th, 2024
Chip Technology
![]() Discovery points path to flash-like memory for storing qubits: Rice find could hasten development of nonvolatile quantum memory April 5th, 2024
Discovery points path to flash-like memory for storing qubits: Rice find could hasten development of nonvolatile quantum memory April 5th, 2024
![]() Utilizing palladium for addressing contact issues of buried oxide thin film transistors April 5th, 2024
Utilizing palladium for addressing contact issues of buried oxide thin film transistors April 5th, 2024
![]() HKUST researchers develop new integration technique for efficient coupling of III-V and silicon February 16th, 2024
HKUST researchers develop new integration technique for efficient coupling of III-V and silicon February 16th, 2024
Memory Technology
![]() Utilizing palladium for addressing contact issues of buried oxide thin film transistors April 5th, 2024
Utilizing palladium for addressing contact issues of buried oxide thin film transistors April 5th, 2024
![]() Interdisciplinary: Rice team tackles the future of semiconductors Multiferroics could be the key to ultralow-energy computing October 6th, 2023
Interdisciplinary: Rice team tackles the future of semiconductors Multiferroics could be the key to ultralow-energy computing October 6th, 2023
![]() Researchers discover materials exhibiting huge magnetoresistance June 9th, 2023
Researchers discover materials exhibiting huge magnetoresistance June 9th, 2023
Sensors
Discoveries
![]() Chemical reactions can scramble quantum information as well as black holes April 5th, 2024
Chemical reactions can scramble quantum information as well as black holes April 5th, 2024
![]() New micromaterial releases nanoparticles that selectively destroy cancer cells April 5th, 2024
New micromaterial releases nanoparticles that selectively destroy cancer cells April 5th, 2024
![]() Utilizing palladium for addressing contact issues of buried oxide thin film transistors April 5th, 2024
Utilizing palladium for addressing contact issues of buried oxide thin film transistors April 5th, 2024
Announcements
![]() NRL charters Navy�s quantum inertial navigation path to reduce drift April 5th, 2024
NRL charters Navy�s quantum inertial navigation path to reduce drift April 5th, 2024
![]() Discovery points path to flash-like memory for storing qubits: Rice find could hasten development of nonvolatile quantum memory April 5th, 2024
Discovery points path to flash-like memory for storing qubits: Rice find could hasten development of nonvolatile quantum memory April 5th, 2024
Tools
![]() Ferroelectrically modulate the Fermi level of graphene oxide to enhance SERS response November 3rd, 2023
Ferroelectrically modulate the Fermi level of graphene oxide to enhance SERS response November 3rd, 2023
![]() The USTC realizes In situ electron paramagnetic resonance spectroscopy using single nanodiamond sensors November 3rd, 2023
The USTC realizes In situ electron paramagnetic resonance spectroscopy using single nanodiamond sensors November 3rd, 2023
|
|
||
|
|
||
| The latest news from around the world, FREE | ||
|
|
||
|
|
||
| Premium Products | ||
|
|
||
|
Only the news you want to read!
Learn More |
||
|
|
||
|
Full-service, expert consulting
Learn More |
||
|
|
||








