Home > Press > Biologists Capture Cell’s Elusive ‘Motor’ on Videotape
 |
Abstract:
In basic research with far-reaching impact, cell biologists Wei-Lih Lee and Steven Markus report in an article released today in Developmental Cell, with videos, that they have solved one of the fundamental questions in stem cell division: How dynein, the cell's two-part, nano-scale "mitotic motor," positions itself to direct the dividing process.
Biologists Capture Cell’s Elusive ‘Motor’ on Videotape
Amherst, MA | Posted on May 18th, 2011Their experiments can be likened to restoring never-before-seen footage to a classic film. What's more, Lee says that footage is crucial to the plot and helps to explain the entire production.
Further, because the same polarization processes as those Lee and colleagues observed in budding yeast occur during asymmetric division of analogous human stem cells, the significance of this discovery is enormous. It should advance understanding of such serious neurological disorders as human lissencephaly, a rare brain formation disorder resulting in a "smooth" brain without folds and grooves, he adds.
In neurons, dynein's major role is to carry waste products from the nerve terminal to the cell body. If this transport goes awry, neurons degenerate, leading to such diseases as Alzheimer's and ALS. "Our discovery solves a huge mystery about how dynein works during asymmetric cell divisions, such as those in stem cells, and confirms to us that the regulation we see for yeast dynein is common to mammalian dynein," Lee says.
In non-neurons, such as dividing stem cells, dynein performs dozens of division-related processes, so many in fact that it's said to "promiscuously" associate with many different cellular cargoes, Lee adds. He and Markus, with undergraduate Jesse Punch, are known for their previous discoveries of how dynein controls the early steps. They belong to the Morrill Motor & Mitosis group in the UMass Amherst biology department.
As they explain, asymmetric division in stem cells is specialized for generating different cell types that will develop into specific tissues such as skin, heart and kidney. Non-stem cell division is simpler and usually symmetrical.
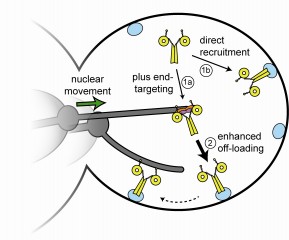
Dynein, the transport-directing two-part motor and cargo molecule, controls the first steps in asymmetric division, aligning and orienting the chromosome-separating spindle apparatus. When the cell is about to divide, dynein rides at the tips of track-like microtubules to a spot in the outer cell periphery, known as the cortex. Dynein's tail end binds and offloads the motor to the cortex, anchoring the molecule to the membrane at a point opposite the spindle apparatus.
With this, dynein acts like a tent stake to hold and pull the spindle apparatus as it moves to a new location to form two unequal daughter cells. Lee and colleagues also knew from earlier work that dynein's motor and tail parts inhibit or mask each other while moving along the microtubule, so no accidental interactions can take place. When the microtubule-guided dynein reaches the cortex and is ready to offload, it unmasks.
What have been completely hidden up to now are the events occurring between the microtubule ride and anchoring in the cell membrane. "This is a long-standing mystery," Lee says. "We could see the masked dynein traveling at the tips of the microtubules, but no one had ever seen it delivered to the cortex."
To trick dynein into revealing the steps in offloading, Lee and Markus engineered an 11-nanometer-long peptide spacer roughly equal to the motor section's diameter. Like a bolster pillow slipped between two halves of a clam shell, it increases the space between the motor and the cargo-attaching tail but still allows the parts to stay physically connected. The researchers then used a conventional fluorescence microscope to observe each part's separate activity with the spacer in place.
The setup worked better than they dared to hope. The mutants (those with an engineered peptide spacer) retained normal motor activity and allowed an enhanced view of offloading. Specifically, Lee says, "With that little forced physical distance between the motor and tail, we expected that both parts would react independently with respect to microtubule tips and the offloading sites, which is what we observed."
"What we didn't expect," he continues, "is the ability to actually observe the delivery process. It was a complete surprise and a eureka moment for us to witness a hypothesis supported by direct evidence for the first time. Now we know how it occurs."
The research team conducted further experiments to compare mutant with normal dynein under a Total Internal Reflection Fluorescence (TIRF) microscope, allowing them to visualize single dynein molecule behavior, Lee points out. These confirm that the elegantly designed peptide spacer disrupted just a single variable, its deployment in the cell, and did not globally disrupt the motor chemical activity.
Overall, results clearly show microtubule-mediated delivery is tightly controlled by a masking mechanism within the dynein molecule, Lee says, which now leads to a lot more questions. His team is already working on at least two: What molecule is doing the un-masking in the various microtubule-mediated polarization processes? And, once offloaded from the microtubule tip, how is the motor "turned on" at the cortex?
####
For more information, please click here
Contacts:
Wei-Lih Lee
413-545-2944
Copyright © Newswise
If you have a comment, please Contact us.Issuers of news releases, not 7th Wave, Inc. or Nanotechnology Now, are solely responsible for the accuracy of the content.
| Related Links |
| Related News Press |
News and information
![]() Simulating magnetization in a Heisenberg quantum spin chain April 5th, 2024
Simulating magnetization in a Heisenberg quantum spin chain April 5th, 2024
![]() NRL charters Navy’s quantum inertial navigation path to reduce drift April 5th, 2024
NRL charters Navy’s quantum inertial navigation path to reduce drift April 5th, 2024
![]() Discovery points path to flash-like memory for storing qubits: Rice find could hasten development of nonvolatile quantum memory April 5th, 2024
Discovery points path to flash-like memory for storing qubits: Rice find could hasten development of nonvolatile quantum memory April 5th, 2024
Videos/Movies
![]() New X-ray imaging technique to study the transient phases of quantum materials December 29th, 2022
New X-ray imaging technique to study the transient phases of quantum materials December 29th, 2022
![]() Solvent study solves solar cell durability puzzle: Rice-led project could make perovskite cells ready for prime time September 23rd, 2022
Solvent study solves solar cell durability puzzle: Rice-led project could make perovskite cells ready for prime time September 23rd, 2022
![]() Scientists prepare for the world’s smallest race: Nanocar Race II March 18th, 2022
Scientists prepare for the world’s smallest race: Nanocar Race II March 18th, 2022
![]() Visualizing the invisible: New fluorescent DNA label reveals nanoscopic cancer features March 4th, 2022
Visualizing the invisible: New fluorescent DNA label reveals nanoscopic cancer features March 4th, 2022
Molecular Machines
![]() First electric nanomotor made from DNA material: Synthetic rotary motors at the nanoscale perform mechanical work July 22nd, 2022
First electric nanomotor made from DNA material: Synthetic rotary motors at the nanoscale perform mechanical work July 22nd, 2022
![]() Nanotech scientists create world's smallest origami bird March 17th, 2021
Nanotech scientists create world's smallest origami bird March 17th, 2021
![]() Giant nanomachine aids the immune system: Theoretical chemistry August 28th, 2020
Giant nanomachine aids the immune system: Theoretical chemistry August 28th, 2020
Molecular Nanotechnology
![]() Scientists push the boundaries of manipulating light at the submicroscopic level March 3rd, 2023
Scientists push the boundaries of manipulating light at the submicroscopic level March 3rd, 2023
![]() First electric nanomotor made from DNA material: Synthetic rotary motors at the nanoscale perform mechanical work July 22nd, 2022
First electric nanomotor made from DNA material: Synthetic rotary motors at the nanoscale perform mechanical work July 22nd, 2022
![]() Nanotech scientists create world's smallest origami bird March 17th, 2021
Nanotech scientists create world's smallest origami bird March 17th, 2021
Discoveries
![]() Chemical reactions can scramble quantum information as well as black holes April 5th, 2024
Chemical reactions can scramble quantum information as well as black holes April 5th, 2024
![]() New micromaterial releases nanoparticles that selectively destroy cancer cells April 5th, 2024
New micromaterial releases nanoparticles that selectively destroy cancer cells April 5th, 2024
![]() Utilizing palladium for addressing contact issues of buried oxide thin film transistors April 5th, 2024
Utilizing palladium for addressing contact issues of buried oxide thin film transistors April 5th, 2024
Announcements
![]() NRL charters Navy’s quantum inertial navigation path to reduce drift April 5th, 2024
NRL charters Navy’s quantum inertial navigation path to reduce drift April 5th, 2024
![]() Discovery points path to flash-like memory for storing qubits: Rice find could hasten development of nonvolatile quantum memory April 5th, 2024
Discovery points path to flash-like memory for storing qubits: Rice find could hasten development of nonvolatile quantum memory April 5th, 2024
|
|
||
|
|
||
| The latest news from around the world, FREE | ||
|
|
||
|
|
||
| Premium Products | ||
|
|
||
|
Only the news you want to read!
Learn More |
||
|
|
||
|
Full-service, expert consulting
Learn More |
||
|
|
||








