Home > Press > Nanotubes pass acid test
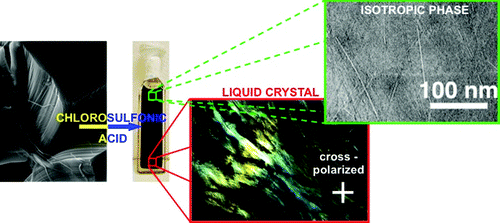 |
| We report that chlorosulfonic acid is a true solvent for a wide range of carbon nanotubes (CNTs), including single-walled (SWNTs), double-walled (DWNTs), multiwalled carbon nanotubes (MWNTs), and CNTs hundreds of micrometers long. The CNTs dissolve as individuals at low concentrations, as determined by cryo-TEM (cryogenic transmission electron microscopy), and form liquid-crystalline phases at high concentrations. The mechanism of dissolution is electrostatic stabilization through reversible protonation of the CNT side walls, as previously established for SWNTs. CNTs with highly defective side walls do not protonate sufficiently and, hence, do not dissolve. The dissolution and liquid-crystallinity of ultralong CNTs are critical advances in the liquid-phase processing of macroscopic CNT-based materials, such as fibers and films. |
Abstract:
Rice researchers' method untangles long tubes, clears hurdle toward armchair quantum wire
Nanotubes pass acid test
Houston, TX | Posted on July 15th, 2010Rice University scientists have found the "ultimate" solvent for all kinds of carbon nanotubes (CNTs), a breakthrough that brings the creation of a highly conductive quantum nanowire ever closer.
Nanotubes have the frustrating habit of bundling, making them less useful than when they're separated in a solution. Rice scientists led by Matteo Pasquali, a professor in chemical and biomolecular engineering and in chemistry, have been trying to untangle them for years as they look for scalable methods to make exceptionally strong, ultralight, highly conductive materials that could revolutionize power distribution, such as the armchair quantum wire.
The armchair quantum wire -- a macroscopic cable of well-aligned metallic nanotubes -- was envisioned by the late Richard Smalley, a Rice chemist who shared the Nobel Prize for his part in discovering the the family of molecules that includes the carbon nanotube. Rice is celebrating the 25th anniversary of that discovery this year.
Pasquali, primary author Nicholas Parra-Vasquez and their colleagues reported this month in the online journal ACS Nano that chlorosulfonic acid can dissolve half-millimeter-long nanotubes in solution, a critical step in spinning fibers from ultralong nanotubes.
Current methods to dissolve carbon nanotubes, which include surrounding the tubes with soap-like surfactants, doping them with alkali metals or attaching small chemical groups to the sidewalls, disperse nanotubes at relatively low concentrations. These techniques are not ideal for fiber spinning because they damage the properties of the nanotubes, either by attaching small molecules to their surfaces or by shortening them.
A few years ago, the Rice researchers discovered that chlorosulfonic acid, a "superacid," adds positive charges to the surface of the nanotubes without damaging them. This causes the nanotubes to spontaneously separate from each other in their natural bundled form.
This method is ideal for making nanotube solutions for fiber spinning because it produces fluid dopes that closely resemble those used in industrial spinning of high-performance fibers. Until recently, the researchers thought this dissolution method would be effective only for short single-walled nanotubes.
In the new paper, the Rice team reported that the acid dissolution method also works with any type of carbon nanotube, irrespective of length and type, as long as the nanotubes are relatively free of defects.
Parra-Vasquez described the process as "very easy."
"Just adding the nanotubes to chlorosulfonic acid results in dissolution, without even mixing," he said.
While earlier research had focused on single-walled carbon nanotubes, the team discovered chlorosulfonic acid is also adept at dissolving multiwalled nanotubes (MWNTs). "There are many processes that make multiwalled nanotubes at a cheaper cost, and there's a lot of research with them," said Parra-Vasquez, who earned his Rice doctorate last year. "We hope this will open up new areas of research."
They also observed for the first time that long SWNTs dispersed by superacid form liquid crystals. "We already knew that with shorter nanotubes, the liquid-crystalline phase is very different from traditional liquid crystals, so liquid crystals formed from ultralong nanotubes should be interesting to study," he said.
Parra-Vasquez, now a postdoctoral researcher at Centre de Physique Moleculaire Optique et Hertzienne, Universite' de Bordeaux, Talence, France, came to Rice in 2002 for graduate studies with Pasquali and Smalley.
Study co-author Micah Green, assistant professor of chemical engineering at Texas Tech and a former postdoctoral fellow in Pasquali's research group, said working with long nanotubes is key to attaining exceptional properties in fibers because both the mechanical and electrical properties depend on the length of the constituent nanotubes. Pasquali said that using long nanotubes in the fibers should improve their properties on the order of one to two magnitudes, and that similar enhanced properties are also expected in thin films of carbon nanotubes being investigated for flexible electronics applications.
An immediate goal for researchers, Parra-Vasquez said, will be to find "large quantities of ultralong single-walled nanotubes with low defects -- and then making that fiber we have been dreaming of making since I arrived at Rice, a dream that Rick Smalley had and that we have all shared since."
Co-authors of the paper are graduate students Natnael Behabtu, Colin Young, Anubha Goyal and Cary Pint; Pulickel Ajayan, the Benjamin M. and Mary Greenwood Anderson Professor in Mechanical Engineering and Materials Science and of chemistry, and Robert Hauge, a distinguished faculty fellow in chemistry, all at Rice; and Judith Schmidt, Ellina Kesselman, Yachin Cohen and Yeshayahu Talmon of the Department of Chemical Engineering, Technion-Israel Institute of Technology, Haifa, Israel.
The Air Force Office of Scientific Research, the Air Force Research Laboratory, the National Science Foundation Division of Materials Research, the Robert A. Welch Foundation, the United States-Israel Binational Science Foundation and the Evans-Attwell Welch Postdoctoral Fellowship funded the research.
Read the abstract at: pubs.acs.org/doi/abs/10.1021/nn100864v
For more about Rice's 25th anniversary Year of Nano celebrations, visit:
buckyball.smalley.rice.edu/year_of_nano/
####
About Rice University
Who Knew? Located in Houston, Rice University is consistently ranked one of America's best teaching and research universities. Known for its "unconventional wisdom," Rice is distinguished by its: size -- 3,102 undergraduates and 2,237 graduate students; selectivity -- 12 applicants for each place in the freshman class; resources -- an undergraduate student-to-faculty ratio of 5-to-1; sixth largest endowment per student among American private research universities; residential college system, which builds communities that are both close-knit and diverse; and collaborative culture, which crosses disciplines, integrates teaching and research, and intermingles undergraduate and graduate work.
For more information, please click here
Contacts:
Jade Boyd
Science Editor
Office of Public Affairs/News & Media Relations
Rice University
(office) 713-348-6778
(cell) 713-302-2447
Copyright © Rice University
If you have a comment, please Contact us.Issuers of news releases, not 7th Wave, Inc. or Nanotechnology Now, are solely responsible for the accuracy of the content.
| Related News Press |
News and information
![]() Simulating magnetization in a Heisenberg quantum spin chain April 5th, 2024
Simulating magnetization in a Heisenberg quantum spin chain April 5th, 2024
![]() NRL charters Navy�s quantum inertial navigation path to reduce drift April 5th, 2024
NRL charters Navy�s quantum inertial navigation path to reduce drift April 5th, 2024
![]() Discovery points path to flash-like memory for storing qubits: Rice find could hasten development of nonvolatile quantum memory April 5th, 2024
Discovery points path to flash-like memory for storing qubits: Rice find could hasten development of nonvolatile quantum memory April 5th, 2024
Academic/Education
![]() Rice University launches Rice Synthetic Biology Institute to improve lives January 12th, 2024
Rice University launches Rice Synthetic Biology Institute to improve lives January 12th, 2024
![]() Multi-institution, $4.6 million NSF grant to fund nanotechnology training September 9th, 2022
Multi-institution, $4.6 million NSF grant to fund nanotechnology training September 9th, 2022
Nanotubes/Buckyballs/Fullerenes/Nanorods/Nanostrings
![]() Tests find no free-standing nanotubes released from tire tread wear September 8th, 2023
Tests find no free-standing nanotubes released from tire tread wear September 8th, 2023
![]() Detection of bacteria and viruses with fluorescent nanotubes July 21st, 2023
Detection of bacteria and viruses with fluorescent nanotubes July 21st, 2023
Discoveries
![]() Chemical reactions can scramble quantum information as well as black holes April 5th, 2024
Chemical reactions can scramble quantum information as well as black holes April 5th, 2024
![]() New micromaterial releases nanoparticles that selectively destroy cancer cells April 5th, 2024
New micromaterial releases nanoparticles that selectively destroy cancer cells April 5th, 2024
![]() Utilizing palladium for addressing contact issues of buried oxide thin film transistors April 5th, 2024
Utilizing palladium for addressing contact issues of buried oxide thin film transistors April 5th, 2024
Announcements
![]() NRL charters Navy�s quantum inertial navigation path to reduce drift April 5th, 2024
NRL charters Navy�s quantum inertial navigation path to reduce drift April 5th, 2024
![]() Discovery points path to flash-like memory for storing qubits: Rice find could hasten development of nonvolatile quantum memory April 5th, 2024
Discovery points path to flash-like memory for storing qubits: Rice find could hasten development of nonvolatile quantum memory April 5th, 2024
Energy
![]() Development of zinc oxide nanopagoda array photoelectrode: photoelectrochemical water-splitting hydrogen production January 12th, 2024
Development of zinc oxide nanopagoda array photoelectrode: photoelectrochemical water-splitting hydrogen production January 12th, 2024
![]() Shedding light on unique conduction mechanisms in a new type of perovskite oxide November 17th, 2023
Shedding light on unique conduction mechanisms in a new type of perovskite oxide November 17th, 2023
![]() Inverted perovskite solar cell breaks 25% efficiency record: Researchers improve cell efficiency using a combination of molecules to address different November 17th, 2023
Inverted perovskite solar cell breaks 25% efficiency record: Researchers improve cell efficiency using a combination of molecules to address different November 17th, 2023
![]() The efficient perovskite cells with a structured anti-reflective layer � another step towards commercialization on a wider scale October 6th, 2023
The efficient perovskite cells with a structured anti-reflective layer � another step towards commercialization on a wider scale October 6th, 2023
Research partnerships
![]() Discovery points path to flash-like memory for storing qubits: Rice find could hasten development of nonvolatile quantum memory April 5th, 2024
Discovery points path to flash-like memory for storing qubits: Rice find could hasten development of nonvolatile quantum memory April 5th, 2024
![]() Researchers� approach may protect quantum computers from attacks March 8th, 2024
Researchers� approach may protect quantum computers from attacks March 8th, 2024
![]() 'Sudden death' of quantum fluctuations defies current theories of superconductivity: Study challenges the conventional wisdom of superconducting quantum transitions January 12th, 2024
'Sudden death' of quantum fluctuations defies current theories of superconductivity: Study challenges the conventional wisdom of superconducting quantum transitions January 12th, 2024
|
|
||
|
|
||
| The latest news from around the world, FREE | ||
|
|
||
|
|
||
| Premium Products | ||
|
|
||
|
Only the news you want to read!
Learn More |
||
|
|
||
|
Full-service, expert consulting
Learn More |
||
|
|
||








