Home > Press > Reaction Mechanisms during Plasma-assisted Atomic Layer Deposition
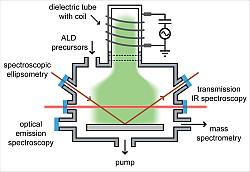 |
| Figure 1: Schematic concept of the different diagnostics implemented in situ to obtain a fundamental understanding of the reaction mechanism of plasma-assisted ALD |
Abstract:
Dr Erik Langereis and Prof Erwin Kessels
Department of Applied Physics, Eindhoven University of Technology, The Netherlands
Reaction Mechanisms during Plasma-assisted Atomic Layer Deposition
UK | Posted on June 14th, 2010Atomic layer deposition (ALD) is considered the primary candidate for growth of conformal films with thickness control on the atomic level. The technique derives its growth control by alternating (two) self-limiting adsorption reactions in order to ensure that a submonolayer of film is deposited per so-called ALD cycle. By selecting the appropriate amount of deposition cycles, the film thickness can be controlled with ultimate precision.
A novel ALD concept is to use a plasma to activate one of the reactants in the gas phase in order to provide additional reactivity to the surface chemistry. These plasma-assisted ALD processes are researched to allow for deposition at reduced temperatures, to realize improved and tunable film properties, and to increase the choice in chemistry and precursors. To obtain a fundamental understanding of the surface reactions taking place and to evaluate the merits of the use of a plasma, a systematic study on the reaction mechanism of plasma-assisted ALD has been carried out for the deposition of metaloxides, metal-nitrides, and noble metals (Fig. 1).
Especially at reduced substrate temperatures, plasma-assisted ALD of metal-oxides distinguishes itself by providing good quality films due to the reactivity delivered by an O2 plasma. By determination of the surface groups by transmission infrared (IR) spectroscopy [1] and the reaction by-products by the combination of mass spectrometry and optical emission spectroscopy (OES) [2], it has been established that the Al2O3 growth is driven by a combustion-like surface chemistry. This chemistry proceeds for depositions down to room temperature. At this low temperature, it is observed that the plasma exposure time is an effective means to optimize the film quality.
For plasma-assisted ALD of metal-nitrides, the reducing power of an H2 plasma is found to be key in depositing conductive films. Using a plasma provides, moreover, the opportunity to tailor the material properties of the film by controlling the plasma condition. For example, by varying the plasma exposure time and plasma gas composition, the TaNx film properties can be controlled from conductive TaN to semiconductive Ta3N5 as evident from spectroscopic ellipsometry [3,4].
Furthermore, for ALD of noble metals such as Pt and Ru, the nucleation and film closure can be improved considerably using a plasma, which can result in smoother and thinner applicable metal films [5]. The mechanisms and growth observations deduced from these studies are expected to be generic for equivalent plasma-assisted ALD processes.
[1] Langereis et al., Appl. Phys. Lett. 92, 231904 (2008).
[2] Heil et al., J. Appl. Phys. 103, 103302 (2008).
[3] Langereis et al., J. Appl. Phys. 102, 083517 (2007).
[4] Langereis et al., topical review in J. Phys. D.: Appl. Phys. 42, 073001 (2009)
[5] Knoops et al., Electrochem. Solid-State Lett. 12, G34 (2009).
####
For more information, please click here
Copyright © Oxford Instruments
If you have a comment, please Contact us.Issuers of news releases, not 7th Wave, Inc. or Nanotechnology Now, are solely responsible for the accuracy of the content.
| Related News Press |
News and information
![]() Simulating magnetization in a Heisenberg quantum spin chain April 5th, 2024
Simulating magnetization in a Heisenberg quantum spin chain April 5th, 2024
![]() NRL charters Navy�s quantum inertial navigation path to reduce drift April 5th, 2024
NRL charters Navy�s quantum inertial navigation path to reduce drift April 5th, 2024
![]() Discovery points path to flash-like memory for storing qubits: Rice find could hasten development of nonvolatile quantum memory April 5th, 2024
Discovery points path to flash-like memory for storing qubits: Rice find could hasten development of nonvolatile quantum memory April 5th, 2024
![]() Good as gold - improving infectious disease testing with gold nanoparticles April 5th, 2024
Good as gold - improving infectious disease testing with gold nanoparticles April 5th, 2024
Thin films
![]() Utilizing palladium for addressing contact issues of buried oxide thin film transistors April 5th, 2024
Utilizing palladium for addressing contact issues of buried oxide thin film transistors April 5th, 2024
![]() Understanding the mechanism of non-uniform formation of diamond film on tools: Paving the way to a dry process with less environmental impact March 24th, 2023
Understanding the mechanism of non-uniform formation of diamond film on tools: Paving the way to a dry process with less environmental impact March 24th, 2023
![]() New study introduces the best graphite films: The work by Distinguished Professor Feng Ding at UNIST has been published in the October 2022 issue of Nature Nanotechnology November 4th, 2022
New study introduces the best graphite films: The work by Distinguished Professor Feng Ding at UNIST has been published in the October 2022 issue of Nature Nanotechnology November 4th, 2022
Academic/Education
![]() Rice University launches Rice Synthetic Biology Institute to improve lives January 12th, 2024
Rice University launches Rice Synthetic Biology Institute to improve lives January 12th, 2024
![]() Multi-institution, $4.6 million NSF grant to fund nanotechnology training September 9th, 2022
Multi-institution, $4.6 million NSF grant to fund nanotechnology training September 9th, 2022
Announcements
![]() NRL charters Navy�s quantum inertial navigation path to reduce drift April 5th, 2024
NRL charters Navy�s quantum inertial navigation path to reduce drift April 5th, 2024
![]() Discovery points path to flash-like memory for storing qubits: Rice find could hasten development of nonvolatile quantum memory April 5th, 2024
Discovery points path to flash-like memory for storing qubits: Rice find could hasten development of nonvolatile quantum memory April 5th, 2024
|
|
||
|
|
||
| The latest news from around the world, FREE | ||
|
|
||
|
|
||
| Premium Products | ||
|
|
||
|
Only the news you want to read!
Learn More |
||
|
|
||
|
Full-service, expert consulting
Learn More |
||
|
|
||








