Home > Press > Spider Silk Reveals a Paradox of Super-strength
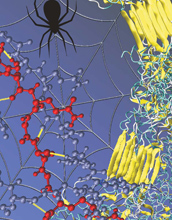 |
| Rendering of the nanoscale structure of silks with beta-sheet nanocrystals shown in yellowish color (right), including a detailed view of the semi-amophous domains between the beta-sheet nanocrystals (left). Credit: Figure courtesy M.J. Buehler (MIT) |
Abstract:
Research finds weakest chemical bonds produce materials stronger than steel
Spider Silk Reveals a Paradox of Super-strength
Arlington, VA | Posted on March 18th, 2010Since its development in China thousands of years ago, silk from silkworms, spiders and other insects has been used for high-end, luxury fabrics as well as for parachutes and medical sutures. Now, National Science Foundation-supported researchers are untangling some of its most closely guarded secrets, and explaining why silk is so super strong.
Researchers at the Massachusetts Institute of Technology's Center for Materials Science and Engineering say the key to silk's pound-for-pound toughness, which exceeds that of steel, is its beta-sheet crystals, the nano-sized cross-linking domains that hold the material together.
Markus Buehler, the Esther and Harold E. Edgerton Associate Professor in MIT's department of civil and environmental engineering, and his team recently used computer models to simulate exactly how the components of beta sheet crystals move and interact with each other. They found that an unusual arrangement of hydrogen bonds--the "glue" that stabilizes the beta-sheet crystals--play an important role in defining the strength of silk.
They found that hydrogen bonds, which are among the weakest types of chemical bonds, gain strength when confined to spaces on the order of a few nanometers in size. Once in close proximity, the hydrogen bonds work together and become extremely strong. Moreover, if a hydrogen bond breaks, there are still many hydrogen bonds left that can contribute to the material's overall strength, due to their ability to "self-heal" the beta-sheet crystals.
The researchers conclude that silk's strength and ductility--its ability to bend or stretch without breaking--results from this peculiar arrangement of atomic bonds. They say controlling the size of the area in which hydrogen or other chemical bonds act can lead to significantly enhanced properties for future materials, even when the initial chemical bonds are very weak.
The journal Nature Materials reported the findings online March 14.
####
About National Science Foundation
The National Science Foundation (NSF) is an independent federal agency that supports fundamental research and education across all fields of science and engineering. In fiscal year (FY) 2010, its budget is about $6.9 billion. NSF funds reach all 50 states through grants to nearly 2,000 universities and institutions. Each year, NSF receives over 45,000 competitive requests for funding, and makes over 11,500 new funding awards. NSF also awards over $400 million in professional and service contracts yearly.
For more information, please click here
Contacts:
Media Contacts
Bobbie Mixon
NSF
(703) 292-8485
Program Contacts
Jorn Larsen-Basse
NSF
(703) 292-7088
Principal Investigators
Markus Buehler
Massachusetts Institute of Technology
(617) 452-2750
Copyright © National Science Foundation
If you have a comment, please Contact us.Issuers of news releases, not 7th Wave, Inc. or Nanotechnology Now, are solely responsible for the accuracy of the content.
| Related News Press |
News and information
![]() Simulating magnetization in a Heisenberg quantum spin chain April 5th, 2024
Simulating magnetization in a Heisenberg quantum spin chain April 5th, 2024
![]() NRL charters Navy�s quantum inertial navigation path to reduce drift April 5th, 2024
NRL charters Navy�s quantum inertial navigation path to reduce drift April 5th, 2024
![]() Discovery points path to flash-like memory for storing qubits: Rice find could hasten development of nonvolatile quantum memory April 5th, 2024
Discovery points path to flash-like memory for storing qubits: Rice find could hasten development of nonvolatile quantum memory April 5th, 2024
Govt.-Legislation/Regulation/Funding/Policy
![]() NRL charters Navy�s quantum inertial navigation path to reduce drift April 5th, 2024
NRL charters Navy�s quantum inertial navigation path to reduce drift April 5th, 2024
![]() Discovery points path to flash-like memory for storing qubits: Rice find could hasten development of nonvolatile quantum memory April 5th, 2024
Discovery points path to flash-like memory for storing qubits: Rice find could hasten development of nonvolatile quantum memory April 5th, 2024
![]() Chemical reactions can scramble quantum information as well as black holes April 5th, 2024
Chemical reactions can scramble quantum information as well as black holes April 5th, 2024
Possible Futures
![]() Discovery points path to flash-like memory for storing qubits: Rice find could hasten development of nonvolatile quantum memory April 5th, 2024
Discovery points path to flash-like memory for storing qubits: Rice find could hasten development of nonvolatile quantum memory April 5th, 2024
![]() With VECSELs towards the quantum internet Fraunhofer: IAF achieves record output power with VECSEL for quantum frequency converters April 5th, 2024
With VECSELs towards the quantum internet Fraunhofer: IAF achieves record output power with VECSEL for quantum frequency converters April 5th, 2024
Academic/Education
![]() Rice University launches Rice Synthetic Biology Institute to improve lives January 12th, 2024
Rice University launches Rice Synthetic Biology Institute to improve lives January 12th, 2024
![]() Multi-institution, $4.6 million NSF grant to fund nanotechnology training September 9th, 2022
Multi-institution, $4.6 million NSF grant to fund nanotechnology training September 9th, 2022
Materials/Metamaterials/Magnetoresistance
![]() Nanoscale CL thermometry with lanthanide-doped heavy-metal oxide in TEM March 8th, 2024
Nanoscale CL thermometry with lanthanide-doped heavy-metal oxide in TEM March 8th, 2024
![]() Focused ion beam technology: A single tool for a wide range of applications January 12th, 2024
Focused ion beam technology: A single tool for a wide range of applications January 12th, 2024
Announcements
![]() NRL charters Navy�s quantum inertial navigation path to reduce drift April 5th, 2024
NRL charters Navy�s quantum inertial navigation path to reduce drift April 5th, 2024
![]() Discovery points path to flash-like memory for storing qubits: Rice find could hasten development of nonvolatile quantum memory April 5th, 2024
Discovery points path to flash-like memory for storing qubits: Rice find could hasten development of nonvolatile quantum memory April 5th, 2024
|
|
||
|
|
||
| The latest news from around the world, FREE | ||
|
|
||
|
|
||
| Premium Products | ||
|
|
||
|
Only the news you want to read!
Learn More |
||
|
|
||
|
Full-service, expert consulting
Learn More |
||
|
|
||








