Home > Press > New approach to wound healing may be easy on skin, but hard on bacteria
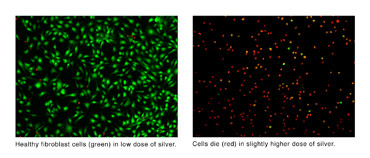 |
| Ankit Agarwal, a postdoctoral researcher at the University of Wisconsin�Madison, is developing polymer films with silver nanoparticles for use in wound treatments. As shown in these 10x microscopic images, Agarwal found that a low dose of silver allows healthy growth of fibroblast cells (shown at left in green), but still manages to kill the bacteria present. Using a slightly higher dose of silver caused the fibroblast cells to die (shown at right in red), which slows the healing. Photo: courtesy Ankit Agarwal/UW�Madison |
Abstract:
In a presentation today (Aug. 19) to the American Chemical Society meeting, Ankit Agarwal, a postdoctoral researcher at the University of Wisconsin-Madison, described an experimental approach to wound healing that could take advantage of silver's antibacterial properties, while sidestepping the damage silver can cause to cells needed for healing.
New approach to wound healing may be easy on skin, but hard on bacteria
Madison, WI | Posted on August 19th, 2009Silver is widely used to prevent bacterial contamination in wound dressings, says Agarwal, "but these dressings deliver a very large load of silver, and that can kill a lot of cells in the wound."
Wound healing is a particular problem in diabetes, where poor blood supply that inhibits healing can require amputations, and also in burn wards. Agarwal says some burn surgeons avoid silver dressings despite their constant concern with infection.
Using a new approach, Agarwal has crafted an ultra-thin material carrying a precise dose of silver. One square inch contains just 0.4 percent of the silver that is found in the silver-treated antibacterial bandages now used in medicine.
In tests in lab dishes, the low concentration of silver killed 99.9999 percent of the bacteria but did not damage cells called fibroblasts that are needed to repair a wound.
Agarwal builds the experimental material from polyelectrolyte multilayers � a sandwich of ultra-thin polymers that adhere through electrical attraction. To make the sandwich, Agarwal alternately dips a glass plate in two solutions of oppositely charged polymers and finally adds a precise dose of silver.
"This architecture is very easily tuned to different applications," Agarwal says, because it allows exact control of such factors as thickness, porosity and silver content. The final sandwich may range from a few nanometers to several hundred nanometers in thickness. (One nanometer is one-billionth of a meter; a human hair is about 60,000 nanometers in diameter.)
Nicholas Abbott, a professor of chemical and biological engineering who advises Agarwal, says during the past decade, "about a bazillion papers have been published on polyelectrolyte multilayers. It's been a tremendous investment by material scientists, and that investment is now ripe to be exploited."
The project was supported by seed funding from the Wisconsin Institutes of Discovery � a new unit devoted to advancing technology in five targeted areas, including tissue engineering � and benefited from contributions by Christopher Murphy, Jonathan McAnulty and Charles Czuprynski of UW-Madison's School of Veterinary Medicine; Ronald Raines of the Department of Biochemistry; and Michael Schurr, a burn surgeon at the School of Medicine and Public Health.
Although both mammalian cells and bacteria are sensitive to silver, bacteria are much more sensitive, leaving a sweet spot � a concentration of silver that can kill bacteria without harming cells needed for healing.
In tests using mouse cells and sample bacteria, Agarwal has tuned the dose to find the sweet spot where the silver bullet destroys 99.9999 percent of the bacteria, but does not harm fibroblasts.
Indeed, the system is so sensitive that increasing the silver dose from 0.4 percent to 1 percent of the level used in a commercial dressing severely damaged the fibroblasts.
To kill bacteria, silver must take the form of charged particles, or ions, and the tiny silver nanoparticles that Agarwal embeds in the sandwich can be designed to release ions for days or weeks as needed. In contrast, Agarwal says, commercial wound dressings contain a large dose of silver ions, which are released faster and with less control.
The required dose of silver can also be reduced because the new material would be designed to stay in close contact with the wound, Abbott says. "In a commercial dressing, the silver is part of the bandage that is placed on the wound surface. We envision this material becoming incorporated into the wound; the cells will grow over it and it will eventually decay and be absorbed into the body, much like an absorbable suture."
Tests on animals will be needed to before the new material can be tested on humans, says Abbott. "A commercial dressing needs to have a large quantity of silver so it can diffuse to the wound bed, and that quantity turns out to be toxic to mammalian cells in lab dishes. We are putting the silver where we need it, so we can use a small loading of silver, which does not exhibit toxicity to mammalian cells because the silver is precisely targeted."
####
About University of Wisconsin-Madison
UW�Madison ranks as one of the most prolific research universities in the world, placing third among American public universities for research expenditures. Driven by a desire to both explore new worlds and to apply new ideas to real-world problems, research at UW�Madison isn't conducted only by faculty, staff and graduate students. Undergraduate research opportunities also are fostered, making research a truly campuswide enterprise.
For more information, please click here
Contacts:
Ankit Agarwal
515-708-1330
Nicholas L. Abbott
608-265-5278
Copyright © University of Wisconsin-Madison
If you have a comment, please Contact us.Issuers of news releases, not 7th Wave, Inc. or Nanotechnology Now, are solely responsible for the accuracy of the content.
| Related News Press |
News and information
![]() Simulating magnetization in a Heisenberg quantum spin chain April 5th, 2024
Simulating magnetization in a Heisenberg quantum spin chain April 5th, 2024
![]() NRL charters Navy�s quantum inertial navigation path to reduce drift April 5th, 2024
NRL charters Navy�s quantum inertial navigation path to reduce drift April 5th, 2024
![]() Discovery points path to flash-like memory for storing qubits: Rice find could hasten development of nonvolatile quantum memory April 5th, 2024
Discovery points path to flash-like memory for storing qubits: Rice find could hasten development of nonvolatile quantum memory April 5th, 2024
Possible Futures
![]() Discovery points path to flash-like memory for storing qubits: Rice find could hasten development of nonvolatile quantum memory April 5th, 2024
Discovery points path to flash-like memory for storing qubits: Rice find could hasten development of nonvolatile quantum memory April 5th, 2024
![]() With VECSELs towards the quantum internet Fraunhofer: IAF achieves record output power with VECSEL for quantum frequency converters April 5th, 2024
With VECSELs towards the quantum internet Fraunhofer: IAF achieves record output power with VECSEL for quantum frequency converters April 5th, 2024
Nanomedicine
![]() New micromaterial releases nanoparticles that selectively destroy cancer cells April 5th, 2024
New micromaterial releases nanoparticles that selectively destroy cancer cells April 5th, 2024
![]() Good as gold - improving infectious disease testing with gold nanoparticles April 5th, 2024
Good as gold - improving infectious disease testing with gold nanoparticles April 5th, 2024
![]() Researchers develop artificial building blocks of life March 8th, 2024
Researchers develop artificial building blocks of life March 8th, 2024
Discoveries
![]() Chemical reactions can scramble quantum information as well as black holes April 5th, 2024
Chemical reactions can scramble quantum information as well as black holes April 5th, 2024
![]() New micromaterial releases nanoparticles that selectively destroy cancer cells April 5th, 2024
New micromaterial releases nanoparticles that selectively destroy cancer cells April 5th, 2024
![]() Utilizing palladium for addressing contact issues of buried oxide thin film transistors April 5th, 2024
Utilizing palladium for addressing contact issues of buried oxide thin film transistors April 5th, 2024
Announcements
![]() NRL charters Navy�s quantum inertial navigation path to reduce drift April 5th, 2024
NRL charters Navy�s quantum inertial navigation path to reduce drift April 5th, 2024
![]() Discovery points path to flash-like memory for storing qubits: Rice find could hasten development of nonvolatile quantum memory April 5th, 2024
Discovery points path to flash-like memory for storing qubits: Rice find could hasten development of nonvolatile quantum memory April 5th, 2024
Nanobiotechnology
![]() New micromaterial releases nanoparticles that selectively destroy cancer cells April 5th, 2024
New micromaterial releases nanoparticles that selectively destroy cancer cells April 5th, 2024
![]() Good as gold - improving infectious disease testing with gold nanoparticles April 5th, 2024
Good as gold - improving infectious disease testing with gold nanoparticles April 5th, 2024
![]() Researchers develop artificial building blocks of life March 8th, 2024
Researchers develop artificial building blocks of life March 8th, 2024
|
|
||
|
|
||
| The latest news from around the world, FREE | ||
|
|
||
|
|
||
| Premium Products | ||
|
|
||
|
Only the news you want to read!
Learn More |
||
|
|
||
|
Full-service, expert consulting
Learn More |
||
|
|
||








