Home > Press > Significant Property Improvement of Plastic Made From Carbon Dioxide
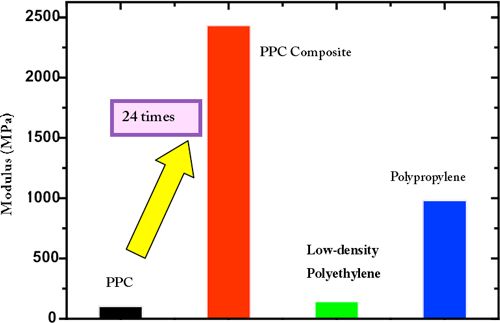 |
| Figure Modulus data for PPC, PPC composite, and general-purpose plastics |
Abstract:
- Poly(propylene carbonate) composite surpassing general-purpose polymers in elastic modulus -
Significant Property Improvement of Plastic Made From Carbon Dioxide
Japan | Posted on December 26th, 2008Points
* Development of high-performance material with 2.4 GPa elastic modulus by using a composite of plastic made from carbon dioxide
* Potential alternative to fossil resource derived plastics such as polyethylene and polypropylene
* Expected to contribute to global warming prevention as a technology for fixing and utilizing carbon dioxide massively emitted in and outside Japan
Summary
Hiroshi Shimizu (Leader) and Li Yongjin (Research Scientist), the Nanostructured Materials Group, the Nanotechnology Research Institute (Director: Nobutsugu Minami) of the National Institute of Advanced Industrial Science and Technology (AIST) (President: Hiroyuki Yoshikawa) succeeded in greatly improving the mechanical properties, including elastic modulus and strength, of plastic (aliphatic polycarbonate) made from carbon dioxide (CO2), previously far from practical use, by blending with other plastics. Plastic with excellent mechanical properties, that is 2.4 GPa of elastic modulus and 17.9 MPa of strength, has been developed by using a composite of poly(propylene carbonate) (PPC), a kind of aliphatic polycarbonate made from CO2 and propylene oxide, and other plastics (see Figure). The PPC composite is a high-performance material demonstrating not only improved mechanical properties but also increased heat resistance, and it is expected to be a viable alternative to petroleum-based general-purpose plastics, such as polyethylene, polypropylene, and polystyrene.
In PPC made from CO2, fixed CO2 accounts for 43% by weight. Although the ratio of fixed CO2 decreases to about 30% by weight by blending with other plastics to enhance its performance, this PPC composite has an advantage over general-purpose plastics in terms of reduced CO2 emission. In Japan, the amount of CO2 emitted from power plants and ironworks has reached a level of 500 million tons per year, calling for the urgent establishment of technologies for separation and fixation of CO2. Wide use of the newly developed PPC composite as an alternative to general-purpose plastics is expected to contribute to global warming prevention and lead to reduced dependence on petroleum resources.
Social Background for Research
A synthetic scheme in which plastics are produced from CO2, which was discovered 40 years ago by Prof. Shohei Inoue (currently Professor of Tokyo University of Science), is receiving renewed attention as a technology for fixing and utilizing CO2, which is one of the measures to prevent global warming. While technological development of aliphatic polycarbonate has been at the basic research level in Japan, pilot plants for PPC production were built by the national budget and commercial mass production of PPC has already started in China. However, the mechanical properties of the produced PPC is still far from practical use and its appearance is similar to soft, sticky rubber rather than plastic, and, therefore, extensive improvement is required.
History of Research
AIST has been developing techniques for mixing plastic materials at the nanometer level and dispersing nanometer-size particles and fillers such as carbon nanotubes into resins. On July 11, 2006, AIST released a polymer-blending technique that uses a high-shear flow field, which achieved nanometer-level mixing of immiscible polymers.
This time, we searched for effective ways to improve the mechanical properties of PPC. Taking into consideration the basic concept of improved material, we aimed at keeping the ratio of CO2 in the PPC composite at about 30 wt%. We hoped to greatly improve the mechanical properties by adding aliphatic polyesters as second and third ingredients, thus controlling the microstructure of the PPC composite.
Details of Research
While the weight-average molecular weight (Mw) of the PPC used in this study reached 447,000, the glass transition temperature (Tg) was as low as 30�C. In addition, poor heat resistance resulted in pyrolysis at 200�C. Its appearance was similar to clear rubber, and it was so sticky that the samples stuck together. It was an unsuitable polymer for practical use.
By adding an aliphatic polyester (X) to this PPC, a polymer blend (composite) was prepared. In addition, another polymer blend was also prepared by adding the second ingredient polymer X and a small amount (2-20 wt%) of another aliphatic polyester (Y), or the third ingredient.
Figure 1 shows the stress-strain curves of the developed PPC composites, which differ in composition. Properties, including elastic modulus, obtained from the stress-strain curves and dynamic viscoelasticity measurements are shown in Table 1. Low-density polyethylene (LDPE) and polypropylene (PP), which are general-purpose plastics, are also listed for comparison.
PPC without additives shows rubber-like features including extremely low elastic modulus and high elongation at break (%). A polymer blend prepared by mixing the PPC and aliphatic polyester X at a ratio of 70 to 30 shows about 15 times as high an elastic modulus, although the elongation at break decreased slightly. Another polymer blend prepared by adding aliphatic polyester Y in addition to the second ingredient X shows 24 times as high an elastic modulus at an optimum composition. By blending the polymers, the glass transition temperature (Tg) of composites increased by almost 10�C and the heat resistance was greatly improved.
Such enhancement of mechanical properties may stem from the inner structure of the PPC composite, and the finest inner structure may have formed at a composition of PPC/X/Y = 70/30/5.
Future Schedule
We continue to pursue further improvement of the properties of PPC composite and also plan to apply the PPC composite to not only general structural materials but also film and packaging materials by making the most of its transparency and gas-barrier property, and to evaluate its performance as a practical material. We intend, therefore, to conduct active technology transfer to companies with an interest in this technology.
####
About AIST
AIST conducts research that transcends the barriers between disciplines, to play an active role in developing advanced expertise for tomorrow's industries.
For more information, please click here
Contacts:
Copyright © AIST
If you have a comment, please Contact us.Issuers of news releases, not 7th Wave, Inc. or Nanotechnology Now, are solely responsible for the accuracy of the content.
| Related News Press |
News and information
![]() Simulating magnetization in a Heisenberg quantum spin chain April 5th, 2024
Simulating magnetization in a Heisenberg quantum spin chain April 5th, 2024
![]() NRL charters Navy�s quantum inertial navigation path to reduce drift April 5th, 2024
NRL charters Navy�s quantum inertial navigation path to reduce drift April 5th, 2024
![]() Discovery points path to flash-like memory for storing qubits: Rice find could hasten development of nonvolatile quantum memory April 5th, 2024
Discovery points path to flash-like memory for storing qubits: Rice find could hasten development of nonvolatile quantum memory April 5th, 2024
Discoveries
![]() Chemical reactions can scramble quantum information as well as black holes April 5th, 2024
Chemical reactions can scramble quantum information as well as black holes April 5th, 2024
![]() New micromaterial releases nanoparticles that selectively destroy cancer cells April 5th, 2024
New micromaterial releases nanoparticles that selectively destroy cancer cells April 5th, 2024
![]() Utilizing palladium for addressing contact issues of buried oxide thin film transistors April 5th, 2024
Utilizing palladium for addressing contact issues of buried oxide thin film transistors April 5th, 2024
Materials/Metamaterials/Magnetoresistance
![]() Nanoscale CL thermometry with lanthanide-doped heavy-metal oxide in TEM March 8th, 2024
Nanoscale CL thermometry with lanthanide-doped heavy-metal oxide in TEM March 8th, 2024
![]() Focused ion beam technology: A single tool for a wide range of applications January 12th, 2024
Focused ion beam technology: A single tool for a wide range of applications January 12th, 2024
Announcements
![]() NRL charters Navy�s quantum inertial navigation path to reduce drift April 5th, 2024
NRL charters Navy�s quantum inertial navigation path to reduce drift April 5th, 2024
![]() Discovery points path to flash-like memory for storing qubits: Rice find could hasten development of nonvolatile quantum memory April 5th, 2024
Discovery points path to flash-like memory for storing qubits: Rice find could hasten development of nonvolatile quantum memory April 5th, 2024
|
|
||
|
|
||
| The latest news from around the world, FREE | ||
|
|
||
|
|
||
| Premium Products | ||
|
|
||
|
Only the news you want to read!
Learn More |
||
|
|
||
|
Full-service, expert consulting
Learn More |
||
|
|
||








