Home > Press > Liquid crystals line up to make self-healing photovoltaic device
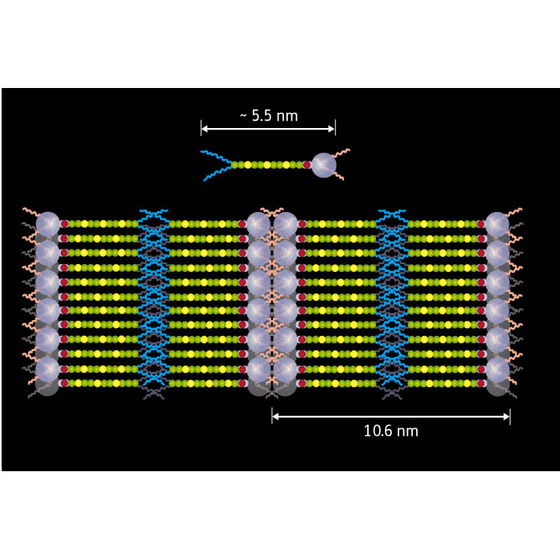 |
| Figure 1: Schematic of the liquid crystal molecule (top) and the formed liquid crystal photovoltaic device (bottom). Purple spheres represent the fullerene and yellow/green chains the oligothiophene, the hydrophobic/hydrophilic tails are represented by blue/red lines respectively. Image:Riken |
Abstract:
Molecules containing both electron donors and acceptors have been functionalized with tails that control their arrangement in a liquid-crystal photovoltaic device
Liquid crystals line up to make self-healing photovoltaic device
Japan | Posted on October 31st, 2008A huge market is developing for small disposable electronic devices, ranging from security tags to point-of-care diagnostics. Many of these devices require a power source, and photovoltaic devices (solar cells) are an attractive option. However, the expense of preparing and processing inorganic semiconductors used in traditional solar cells precludes their use in such applications. Organic photovoltaic devices, meanwhile have great potential in this area; they are relatively easy to prepare and can be processed by simple techniques such as inkjet printing.
Organic photovoltaic devices contain both electron donors, which release an electron when irradiated, and electron acceptors, which complete the circuit necessary to convert light energy into electrical energy. However, mixtures of typical electron donors such as π-conjugated oligomers�short chains of repeated, unsaturated, organic molecules, with alternating double and single bonds�and electron acceptors, such as C60 (buckminsterfullerene), have a tendency to form alternating stacks that results in lower efficiency. A partial solution is to directly attach the electron donor to the electron acceptor by a covalent bond and have both in a single molecule, but it is still important to have control over how the molecules pack together.
Now, a team of Japanese researchers including Takuzo Aida from the University of Tokyo and Masaki Takata from the RIKEN SPring-8 Center in Harima have designed liquid crystals�a phase that flows like a liquid but has short-range order between the molecules�that spontaneously assemble to form a donor-acceptor array1. "It's important to form separated columns or layers of the donors and acceptors, and to make a large contact area between them," explains Yohei Yamamoto, another member of the team from the Japan Science and Technology Agency in Tokyo.
The molecules they designed feature a fullerene�the electron acceptor�at one end and a thiophene oligomer�the electron donor�at the other. A hydrophobic, or water-repellent, tail is attached to the donor end and a hydrophilic, or water-loving, tail is attached to the acceptor end. This functionalization ensures that the molecules of the liquid crystal line up (Fig. 1) to produce ordered layers of donors and acceptors and results in efficient photovoltaic behavior. "The liquid characteristics are useful as well," notes Yamamoto, "the devices are self-healing as defects in the layer structure can be repaired by a simple heating and cooling process." The design principles developed in this work should lead to the development of high-efficiency organic photovoltaic devices.
Reference
1. Li, W.-S., Yamamoto, Y., Fukushima, T., Saeki, A., Seki, S., Tagawa, S., Masunaga, H., Sasaki, S., Takata, M. & Aida, T. Amphiphilic molecular design as a rational strategy for tailoring bicontinuous electron donor and acceptor arrays: photoconductive liquid crystalline oligothiophene-C60 dyads. Journal of the American Chemical Society 130, 8886-8887 (2008).
The corresponding author for this highlight is based at the RIKEN Structural Materials Science Laboratory
####
For more information, please click here
Copyright © Riken
If you have a comment, please Contact us.Issuers of news releases, not 7th Wave, Inc. or Nanotechnology Now, are solely responsible for the accuracy of the content.
| Related Links |
| Related News Press |
News and information
![]() Simulating magnetization in a Heisenberg quantum spin chain April 5th, 2024
Simulating magnetization in a Heisenberg quantum spin chain April 5th, 2024
![]() NRL charters Navy�s quantum inertial navigation path to reduce drift April 5th, 2024
NRL charters Navy�s quantum inertial navigation path to reduce drift April 5th, 2024
![]() Discovery points path to flash-like memory for storing qubits: Rice find could hasten development of nonvolatile quantum memory April 5th, 2024
Discovery points path to flash-like memory for storing qubits: Rice find could hasten development of nonvolatile quantum memory April 5th, 2024
Discoveries
![]() Chemical reactions can scramble quantum information as well as black holes April 5th, 2024
Chemical reactions can scramble quantum information as well as black holes April 5th, 2024
![]() New micromaterial releases nanoparticles that selectively destroy cancer cells April 5th, 2024
New micromaterial releases nanoparticles that selectively destroy cancer cells April 5th, 2024
![]() Utilizing palladium for addressing contact issues of buried oxide thin film transistors April 5th, 2024
Utilizing palladium for addressing contact issues of buried oxide thin film transistors April 5th, 2024
Announcements
![]() NRL charters Navy�s quantum inertial navigation path to reduce drift April 5th, 2024
NRL charters Navy�s quantum inertial navigation path to reduce drift April 5th, 2024
![]() Discovery points path to flash-like memory for storing qubits: Rice find could hasten development of nonvolatile quantum memory April 5th, 2024
Discovery points path to flash-like memory for storing qubits: Rice find could hasten development of nonvolatile quantum memory April 5th, 2024
Energy
![]() Development of zinc oxide nanopagoda array photoelectrode: photoelectrochemical water-splitting hydrogen production January 12th, 2024
Development of zinc oxide nanopagoda array photoelectrode: photoelectrochemical water-splitting hydrogen production January 12th, 2024
![]() Shedding light on unique conduction mechanisms in a new type of perovskite oxide November 17th, 2023
Shedding light on unique conduction mechanisms in a new type of perovskite oxide November 17th, 2023
![]() Inverted perovskite solar cell breaks 25% efficiency record: Researchers improve cell efficiency using a combination of molecules to address different November 17th, 2023
Inverted perovskite solar cell breaks 25% efficiency record: Researchers improve cell efficiency using a combination of molecules to address different November 17th, 2023
![]() The efficient perovskite cells with a structured anti-reflective layer � another step towards commercialization on a wider scale October 6th, 2023
The efficient perovskite cells with a structured anti-reflective layer � another step towards commercialization on a wider scale October 6th, 2023
Solar/Photovoltaic
![]() Development of zinc oxide nanopagoda array photoelectrode: photoelectrochemical water-splitting hydrogen production January 12th, 2024
Development of zinc oxide nanopagoda array photoelectrode: photoelectrochemical water-splitting hydrogen production January 12th, 2024
![]() Shedding light on unique conduction mechanisms in a new type of perovskite oxide November 17th, 2023
Shedding light on unique conduction mechanisms in a new type of perovskite oxide November 17th, 2023
![]() Inverted perovskite solar cell breaks 25% efficiency record: Researchers improve cell efficiency using a combination of molecules to address different November 17th, 2023
Inverted perovskite solar cell breaks 25% efficiency record: Researchers improve cell efficiency using a combination of molecules to address different November 17th, 2023
![]() Charged �molecular beasts� the basis for new compounds: Researchers at Leipzig University use �aggressive� fragments of molecular ions for chemical synthesis November 3rd, 2023
Charged �molecular beasts� the basis for new compounds: Researchers at Leipzig University use �aggressive� fragments of molecular ions for chemical synthesis November 3rd, 2023
Printing/Lithography/Inkjet/Inks/Bio-printing/Dyes
![]() Presenting: Ultrasound-based printing of 3D materials�potentially inside the body December 8th, 2023
Presenting: Ultrasound-based printing of 3D materials�potentially inside the body December 8th, 2023
![]() Simple ballpoint pen can write custom LEDs August 11th, 2023
Simple ballpoint pen can write custom LEDs August 11th, 2023
![]() Disposable electronics on a simple sheet of paper October 7th, 2022
Disposable electronics on a simple sheet of paper October 7th, 2022
|
|
||
|
|
||
| The latest news from around the world, FREE | ||
|
|
||
|
|
||
| Premium Products | ||
|
|
||
|
Only the news you want to read!
Learn More |
||
|
|
||
|
Full-service, expert consulting
Learn More |
||
|
|
||








